Translate this page into:
Percutaneous Direct Needle Puncture and Transcatheter N-butyl Cyanoacrylate Injection Techniques for the Embolization of Pseudoaneurysms and Aneurysms of Arteries Supplying the Hepato-pancreato-biliary System and Gastrointestinal Tract
Address for correspondence: Dr. Rajanikant R Yadav, Department of Radiodiagnosis, Sanjay Gandhi Postgraduate Institute of Medical Sciences, Raibareli Road, Lucknow - 226 014, Uttar Pradesh, India. E-mail: rajani24478@gmail.com
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Aims:
The aim of this study was to evaluate the safety and clinical efficacy of percutaneous direct needle puncture and transcatheter N-butyl cyanoacrylate (NBCA) injection techniques for the embolization of pseudoaneurysms and aneurysms of arteries supplying the hepato-pancreato-biliary (HPB) system and gastrointestinal (GI) tract.
Subjects and Methods:
A hospital-based cross-sectional retrospective study was conducted, where the study group comprised 11 patients with pseudoaneurysms/aneurysms of arteries supplying the HPB system and GI tract presenting to a tertiary care center from January 2015 to June 2016. Four patients (36.4%) underwent percutaneous direct needle puncture of pseudoaneurysms with NBCA injection, 3 patients (27.3%) underwent transcatheter embolization with NBCA as sole embolic agent, and in 4 patients (36.4%), transcatheter NBCA injection was done along with coil embolization.
Results:
This retrospective study comprised 11 patients (8 males and 3 females) with mean age of 35.8 years ± 1.6 (standard deviation [SD]). The mean volume of NBCA: ethiodized oil (lipiodol) mixture injected by percutaneous direct needle puncture was 0.62 ml ± 0.25 (SD) (range = 0.5–1 ml), and by transcatheter injection, it was 0.62 ml ± 0.37 (SD) (range = 0.3–1.4 ml). Embolization with NBCA was technically and clinically successful in all patients (100%). No recurrence of bleeding or recurrence of pseudoaneurysm/aneurysm was noted in our study.
Conclusions:
Percutaneous direct needle puncture of visceral artery pseudoaneurysms and NBCA glue injection and transcatheter NBCA injection for embolization of visceral artery pseudoaneurysms and aneurysms are cost-effective techniques that can be used when coil embolization is not feasible or has failed.
Keywords
Gastrointestinal bleed
N-butyl cyanoacrylate
pseudoaneurysms
transcatheter

INTRODUCTION
Visceral artery aneurysms are uncommon with a reported incidence of 0.01%–0.2% in autopsies.[1] Symptomatic visceral artery aneurysms are clinically important as rupture of these aneurysms is a potentially life-threatening situation. Twenty-two percent of all visceral artery aneurysms present as acute clinical emergency and about 8.5% of visceral artery aneurysm results in death because of rupture.[1] Even though visceral artery aneurysms have all three layers of arterial wall, they may undergo progressive dilatation with thinning of their walls and eventually form pseudoaneurysms which are prone for rupture.[1]
Pseudoaneurysms of arteries supplying the HPB system and gastrointestinal (GI) tract are prone to rupture and may present with life-threatening acute GI or internal bleeding requiring immediate management. These pseudoaneurysms develop as a result of inflammation, infection, trauma, neoplasm, or as a complication of surgical, endoscopic, and radiological interventions. Surgical ligation of the culprit artery proximal and distal to the pseudoaneurysm or resection of the pseudoaneurysm with or without partial resection of the end organ involved was the conventional treatment for these pseudoaneurysms. This has now largely been replaced by transcatheter embolization which has a high success rate and lower morbidity and mortality rates.[12] Metallic coils are conventionally the safe and preferred embolic materials for the management of GI bleed due to visceral artery pseudoaneurysms with reported technical success rates of 63%–100%.[34] However, a high incidence of rebleeding (3%–37%) has been reported with coil embolization.[3456]
Liquid embolic materials such as N-butyl cyanoacrylate (NBCA) are now increasingly being used as additional embolic material in cases of failure of coil embolization and also as the sole embolic agent for GI bleeding due to visceral artery pseudoaneurysms with better clinical success rates and lower recurrence rates.[267] NBCA is usually injected through an intravascular microcatheter into the pseudoaneurysm or into the culprit arteries. NBCA polymerizes immediately on contact with anions in the blood and immediately occludes the vessel. Mixed with iodized oil (Lipiodol; Guerbet, Villepinte, France) which confers radio-opacity and increases polymerization time of NBCA, it can be delivered into distal vessels/collaterals which are inaccessible with the microcatheter. NBCA is effective even in cases with coagulopathy because it does not depend on the normal coagulation process for its therapeutic effect. However, use of NBCA as an embolic agent by transcatheter injection requires practice and experience in terms of volume, dilution, and rate of injection to avoid complications such as nontarget distal embolization, proximal reflux into nontarget vessels, and adhesion of catheter to the embolized vessel.[89]
Direct percutaneous puncture with a needle and injection of NBCA for the management of visceral artery pseudoaneurysms has rarely been described in few studies involving a small number of cases or in case reports.[10111213]
We evaluate the safety and clinical efficacy of percutaneous direct needle puncture and transcatheter NBCA injection techniques for the embolization of pseudoaneurysms and aneurysms of arteries supplying the hepato-pancreato-biliary (HPB) system and GI tract.
SUBJECTS AND METHODS
After the approval from the Institutional Ethics Committee, a hospital-based cross-sectional retrospective study was conducted. The study group comprised 11 patients with pseudoaneurysms or aneurysms of the HPB system or GI tract, presenting to the Departments of Radiodiagnosis, Medical Gastroenterology, and Surgical Gastroenterology at a tertiary care center from January 2015 to June 2016.
The diagnosis of visceral artery pseudoaneurysm or aneurysm was made by multidetector computed tomography (CT) angiography in all cases using a Brilliance 64 slice CT scanner (Philips, Netherlands). Written informed consent was obtained from each patient or their relatives before embolization.
The indications for using NBCA as an embolic agent were:
-
Inability to access a pseudoaneurysm and the artery distal to it with a microcatheter due to
-
Narrow caliber of feeding artery proximal to the pseudoaneurysm
-
Previously coil-embolized artery proximal to a pseudoaneurysm
-
Difficult catheterization of feeding artery.
-
-
Inability to access the distal artery across a large aneurysm
-
Failed coil embolization, and
-
Multiple collaterals distal to a pseudoaneurysm.
Arterial access was obtained in all patients through the femoral route using a 5 Fr vascular access sheath. Using a 0.35" hydrophilic guidewire (Radiofocus; Terumo Inc., Japan), and a Simmons 1 guiding catheter (Cook Inc., USA), the celiac trunk and superior mesenteric arteries (SMAs) were canulated under fluoroscopy. Where required, a 2.7 Fr Progreat microcatheter (Terumo Inc., Japan) was used to super-selectively access the culprit arteries. Angiograms were obtained to delineate the pseudoaneurysms/aneurysms and determine the treatment plan.
Four patients (36.4%) underwent percutaneous embolization of pseudoaneurysms [Figures 1-3]. The pseudoaneurysms were accessed under ultrasound guidance with a 20-gauge Chiba needle (Cook Incorporation, USA). Few contrast injections were made into the pseudoaneurysms under fluoroscopy to determine the rate of injection and volume of NBCA: lipiodol mixture required for embolization of the pseudoaneurysm and to just cause minimal reflux into the culprit artery. The Chiba needle was then flushed a few times slowly with 1 ml of 5% dextrose, and then, under fluoroscopy guidance, the required amount and ratio of NBCA: lipiodol mixture were injected into the pseudoaneurysm. The Chiba needle was then removed. Catheter angiograms were taken to check for adequacy of embolization.

- A 19-year-old male patient of extrahepatic portal venous obstruction with portal biliopathy presented with pericatheter bleeding 5 days after percutaneous cholecystostomy. Digital subtraction angiography images (a and b) show a peripheral left hepatic artery pseudoaneurysm (↗black arrow) with a tortuous and narrow caliber proximal artery. (c) 0.5 ml N-butyl cyanoacrylate: lipiodol (1:1) injection after percutaneous direct puncture of the pseudoaneurysm (↗black arrow) with a 20-gauge Chiba needle. Post N-butyl cyanoacrylate injection catheter angiogram in image (d) shows glue cast in the pseudoaneurysm (↗black arrow) with no residual filling of pseudoaneurysm.
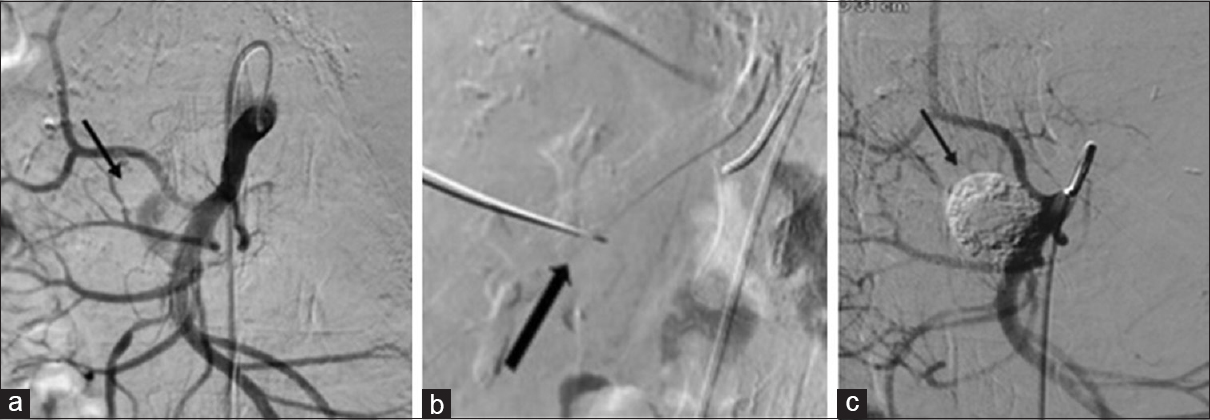
- 34 year old male patient with acute on chronic pancreatitis presented with pain abdomen, (a) DSA image shows a large SMA branch pseudoaneurysm (↓black block arrow) arising close to its origin from the SMA. Image (b) shows percutaneous direct puncture of the pseudoaneurysm with a 20 Gauge Chiba needle (↑black block arrow). Image (c) post NBCA injection angiogram shows glue cast in the pseudoaneurysm (↓black block arrow) with no residual filling of the pseudoaneurysm and preserved lumen of SMA and its branches.
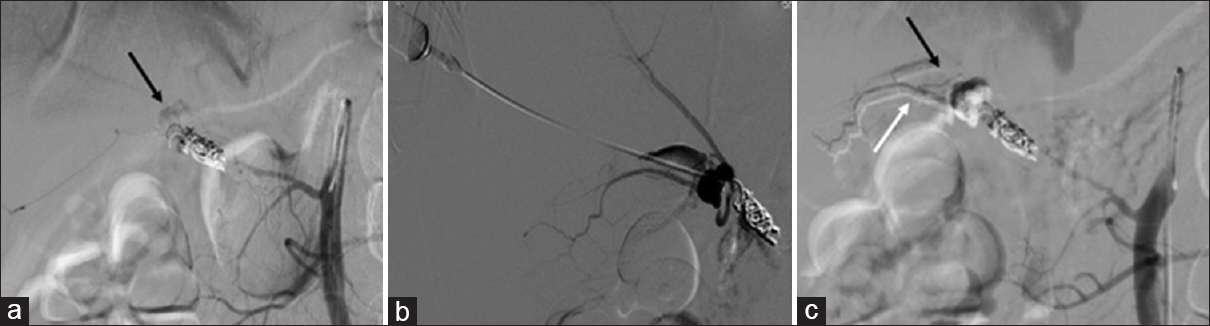
- A 35-year-old male patient presented with massive upper gastrointestinal bleed 1 month after coil embolization of the replaced right hepatic artery proximal to a cystic artery pseudoaneurysm. (a) Superior mesenteric artery angiogram shows a persistent cystic artery pseudoaneurysm (↘black arrow) arising from a replaced right hepatic artery which was inadequately coiled initially. (b) Angiogram obtained after percutaneous needle puncture shows filling of the pseudoaneurysm. (c) Post N-butyl cyanoacrylate injection superior mesenteric artery angiogram shows glue cast in the pseudoaneurysm (↘black arrow) and minimal distal glue reflux into the cystic artery (↗white arrow).
Seven patients (63.6%) underwent transcatheter embolization with NBCA: lipiodol mixture. Of these, NBCA was used as the sole embolic agent in 3 patients (27.3%) [Figures 4 and 5] and was used in addition to coils in 4 patients (36.4%) [Figures 6 and 7]. The pseudoaneurysm or its feeding proximal artery or distal collaterals were accessed with a microcatheter. The microcatheter was flushed a few times with 5% dextrose, and then, NBCA: lipiodol mixture was injected. The microcatheter was immediately pulled in all cases to prevent attachment to the culprit artery. NBCA: lipiodol was used in 1:3 ratio and 0.4–0.7 ml of the mixture was injected at a time. If the initial injection did not achieve occlusion of the vessel, further injection of similar concentration and volume was done with a new microcatheter. The rate and volume of NBCA: lipiodol mixture injected (inclusive of ≈ 0.3 ml dead space of microcatheter lumen) was determined by repeated contrast injections so as to fill the pseudoaneurysm with minimal reflux into the culprit artery.

- A 18-year-old female with acute on chronic pancreatitis presented with severe upper gastrointestinal bleed with shock. Digital subtraction angiography image (a) common hepatic artery angiogram shows a small right gastric artery pseudoaneurysm (↙black arrow). (b) Superselective microcatheter angiogram after shows the right gastric artery pseudoaneurysm (↙black arrow). (c) Angiogram obtained after transcatheter N-butyl cyanoacrylate injection shows glue cast (↙black arrow) in the pseudoaneurysm and the right gastric artery proximal and distal to the pseudoaneurysm with minimal glue reflux into the hepatic arteries (↗ and ← white arrows) causing partial luminal occlusion.
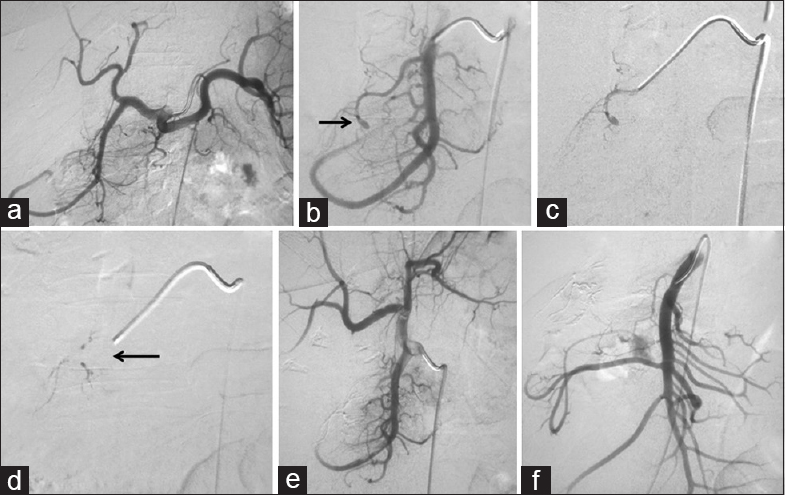
- A 35-year-old female with persistent upper gastrointestinal bleed after endoscopic sphincterotomy and surgery for common bile duct stone clearance. (a) Celiac and (b) gastroduodenal artery angiograms show a pseudoaneurysm arising from a proximal branch of gastroduodenal artery (→black arrow). (c) Super selective angiogram shows the pseudoaneurysm. (d) N-butyl cyanoacrylate injection with glue cast in the pseudoaneurysm and the gastroduodenal artery branch proximal and distal to the pseudoaneurysm (←black arrow). (e and f) Check celiac and superior mesenteric angiograms show glue cast in the pseudoaneurysm with preservation of the lumen of the gastroduodenal artery and nonfilling of the pseudoaneurysm.
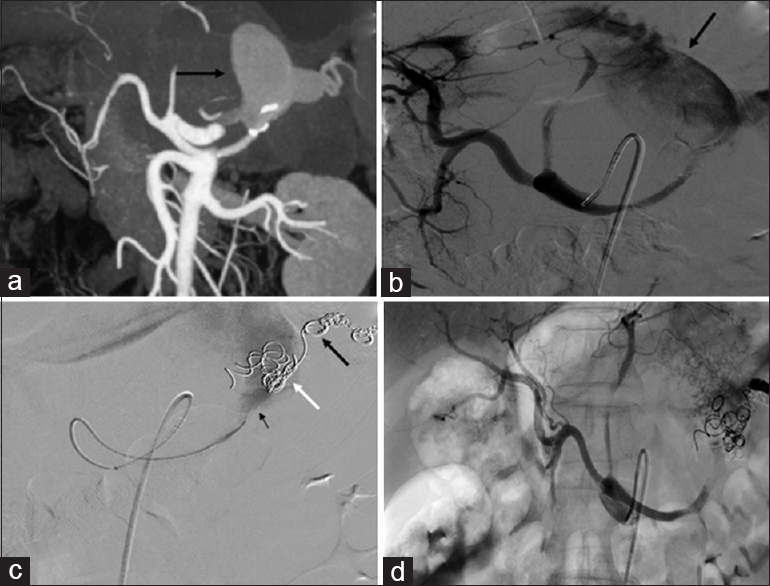
- A 56-year-old male with hypertension. (a) Computed tomography angiogram and (b) digital subtraction angiography celiac angiogram shows a large aneurysm (↙black arrow) arising from splenic artery. (c) Splenic artery angiogram shows occlusion of the splenic artery distal to the aneurysm with coils (↖black arrow) with multiple migrated coils (↖white arrow) in the aneurysm with residual aneurysm sac (small ↖ black arrow). (d) Celiac angiogram obtained after transcatheter N-butyl cyanoacrylate: lipiodol (1:3) injection into the proximal splenic artery shows no filling of the aneurysm and occlusion of proximal splenic artery.
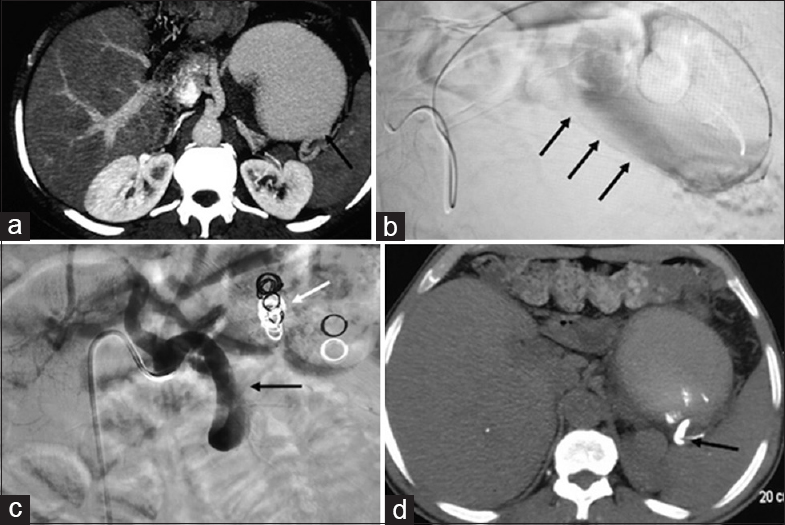
- A 56-year-male with abdominal pain. (a) Axial computed tomography angiogram shows a large splenic artery aneurysm (↖black arrow). (b) Microcatheter angiogram through microcatheter in the aneurysm (↗black arrows) with inability to access the artery distal to the aneurysm. (c) Celiac angiogram after failed coiled embolization shows migration of coils (←white arrow) and after N-butyl cyanoacrylate shows glue cast in the midsplenic artery (←black arrow) with nonvisualization of pseudoaneurysm. (d) Postprocedure noncontrast axial computed tomography section shows glue cast (←black arrow) in the splenic artery distal to the pseudoaneurysm.
Statistical analysis
Data were presented in terms of percentage and mean. Calculations were done using Microsoft Excel and SPSS programs (Statistical Package for the Social Science version 16, SPSS Inc., Chicago, USA).
RESULTS
This retrospective study included 11 patients (8 males and 3 females; mean age of 35.8 years ± 1.6 [standard deviation (SD)], age range = 18–63 years) with visceral artery pseudoaneurysm or aneurysm of arteries supplying the HPB system or GI tract with variable clinical presentation and severity. Pertinent patient's information is summarized in Table 1. The mean volume of NBCA: ethiodized oil (lipiodol) mixture injected by percutaneous direct needle puncture was 0.62 ml ± 0.25 (SD) (range = 0.5-1 ml) and by transcatheter injection, it was 0.62 ml ± 0.37 (SD) (range = 0.3–1.4 ml).
Technical and clinical success was seen in all 11 patients (100%) with no rebleeding or recurrence of pseudoaneurysm/aneurysm in any of the patients. No complications were seen in patients who underwent percutaneous direct puncture of pseudoaneurysms with NBCA injection. Complications were seen in 1 out of 7 (14.3%) patients who underwent transcatheter glue embolization. Inconsequential reflux of glue into the hepatic arteries occurred in 1 patient with the right gastric artery (RGA) pseudoaneurysm [Figure 4]. The mean duration of follow-up of patients in this study was 13.5 months ± 11.5 (SD). No recurrence of pseudoaneurysms occurred in the follow-up period. Table 2 shows frequency and sizes of aneurysms and pseudoaneurysms.

DISCUSSION
In this retrospective study, we studied percutaneous direct needle puncture and NBCA injection and transcatheter NBCA injection techniques for the embolization of pseudoaneurysms and aneurysms of arteries supplying the HPB system and GI tract.
Technical and clinical success rate in our study with direct needle puncture of pseudoaneurysms and NBCA glue injection were 100%, and no recurrences or complications were observed.
Vyas et al., embolized four blunt abdominal trauma-related hepatic artery pseudoaneurysms with percutaneous direct needle puncture of pseudoaneurysms and injection of NBCA: lipiodol mixture and observed similar results. In one of their cases, NBCA: lipiodol mixture had to be injected after failure of direct puncture and thrombin injection into the pseudoaneurysm.[10]
Kulkarni et al., used the same method for the management of a pancreatitis-related gastroduodenal artery (GDA) pseudoaneurysm which showed refilling from the inferior pancreaticoduodenal artery (IPDA) collaterals after coiling of the GDA proximal to the pseudoaneurysm had been done.[11] In our study, one patient was referred with a persistent bleeding cystic artery pseudoaneurysm due to refilling from inadequately coiled feeding replaced right hepatic artery (RHA) and from distal collaterals. As the pseudoaneurysm could not be accessed through the replaced RHA or through collaterals, direct puncture of the pseudoaneurysm and injection of NBCA: lipiodol mixture were done [Figure 3]. Minimal intended glue reflux was observed in the distal cystic artery.
Gulati et al., performed percutaneous direct puncture and injection of NBCA: lipiodol in 1:3 ratio into an IPDA pseudoaneurysm arising close to the IPDA origin with a balloon inflated in the SMA. They observed small glue reflux into the jejunal branches of the SMA and delayed occurrence of jejunal stricture after 8 months, for which the patient had to be operated.[12] In our two cases of jejunal branch of SMA pseudoaneurysms (one with short safe landing zone [Figure 2] and the other a peripheral jejunal branch pseudoaneurysm), calculated volume of NBCA: lipiodol was injected in 1:1 ratio to completely embolize the pseudoaneurysm and no reflux of glue was observed. We also did not require placing a balloon catheter in the SMA to stop reflux. Significant reflux did not occur in our cases probably due to higher concentration and calculated volume of NBCA used which caused immediate polymerization of glue in the pseudoaneurysm. Direct injection of NBCA into the pseudoaneurysm may lead to rupture of a friable-walled pseudoaneurysm due to increased pressure within the pseudoaneurysm.[11] We did not observe pseudoaneurysm rupture in any of our cases as a calculated minimal volume of NBCA: lipiodol mixture was injected.
The present study had a technical success rate and clinical success rate of 100% for transcatheter embolization using NBCA as was seen by Izaki et al., (n = 12),[3] Madhusudhan et al., (n = 31),[6] Won et al., (n = 10)[14] in their studies on use of NBCA for embolization of visceral artery pseudoaneurysms. Song et al.,[2] reported 92.3% (12 of 13 patients) technical success rate for transcatheter embolization of GI arterial, splenic artery, and hepatic arterial pseudoaneurysms using NBCA. Madhusudhan et al.,[6] reported 9.7% recurrences (3 out of 31 patients) within 30 days of transcatheter embolization which were managed by repeat embolization. Song et al.,[2] Izaki et al.,[3] and Won et al.,[14] reported no recurrences. No recurrence of bleeding was seen in our study.
Complication rates varied from 0% to 16.6%[23614] in earlier studies that employed transcatheter NBCA injection for the management of visceral artery aneurysm and pseudoaneurysms. Song et al.,[2] and Won et al.,[14] did not experience any complication in their cases. Madhusudhan et al.,[6] observed complications in 3 out of 31 (9.7%) cases including proximal glue reflux with hepatic arterial occlusion and raised transaminases, distal reflux with splenic infarct requiring splenectomy, and catheter adhesion in 1 case each. Izaki et al.,[3] reported complications in 2 out of 12 procedures (16.6%) which included distal overflow with inconsequential partial embolization of the middle colic artery in 1 patient and proximal reflux with inconsequential partial occlusion of the middle colic and first jejunal arteries in 1 patient.
We observed minor complications in 1 out of our 7 (14.3%) patients of transcatheter embolization in the form of inconsequential hepatic arterial reflux in 1 patient with RGA pseudoaneurysm [Figure 4]. The higher incidence of complications in our study was related to small number of patients in our study. Jae et al.,[9] advocated trial injections with iodinated contrast material to understand the flow dynamics before embolization so that nontarget embolization can be avoided. They also suggested that only a small amount (0.2–0.6 ml) of NBCA: lipiodol mixture was sufficient to achieve occlusion of bleeding site, feeding, and collateral arteries.[9]
Madhusudan et al., advocated a modified technique of sequential slow injection of 0.1–0.3 ml NBCA injection followed by flushing the microcatheter with dextrose. This is repeated if required without having to use a new microcatheter.[6] Won et al., advocated a packing technique using NBCA: lipiodol in 1:1 or 1:2 ratio when access to the pseudoaneurysm and stable catheter position was achievable to avoid prolonged polymerization of glue and prevent migration of embolic material. When access to the pseudoaneurysm was not possible, they advocated an isolation technique using NBCA: lipiodol in 1:3 ratio.[14]
Limitation
Our study included only 11 patients. We look forward to doing a prospective study in future including a larger sample size to assess the safety, efficacy, indications, and technique for embolization of visceral artery pseudoaneurysms and aneurysms using NBCA.
CONCLUSIONS
We advocate that embolization of visceral artery pseudoaneurysms by percutaneous direct needle puncture and NBCA injection is a cost-effective technique in cases with inability to reach a microcatheter to the pseudoaneurysm or beyond it, inability to catheterize the artery feeding the pseudoaneurysm with a microcatheter or in cases of persistent/collateralized pseudoaneurysms when coil embolization has been done only proximal to the pseudoaneurysm. Transcatheter NBCA embolization for visceral artery pseudoaneurysms and aneurysms requires practice and experience to avoid complications. Small volumes of NBCA (preferably <0.6 ml) should be injected through the microcatheter at a time. It should be used in situations where coil embolization is not possible or has failed. It is useful when a pseudoaneurysm/aneurysm and/or the artery distal to it are inaccessible with a microcatheter (due to narrow proximal artery or a large pseudoaneurysm/aneurysm), when a pseudoaneurysm has multiple distal collaterals, or when rapid flow causes loss of coils into large aneurysms or pseudoaneurysms.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/48/196278
REFERENCES
- Visceral artery aneurysms: Diagnosis and percutaneous management. Semin Intervent Radiol. 2009;26:196-206.
- [Google Scholar]
- Transcatheter N-butyl cyanoacrylate embolization of pseudoaneurysms. J Vasc Interv Radiol. 2010;21:1508-11.
- [Google Scholar]
- N-butyl cyanoacrylate embolization for pseudoaneurysms complicating pancreatitis or pancreatectomy. J Vasc Interv Radiol. 2011;22:302-8.
- [Google Scholar]
- Transcatheter microcoil embolotherapy for ruptured pseudoaneurysm following pancreatic and biliary surgery. Cardiovasc Intervent Radiol. 2002;25:180-5.
- [Google Scholar]
- Improved outcomes in postoperative and pancreatitis-related visceral pseudoaneurysms. J Gastrointest Surg. 2007;11:50-5.
- [Google Scholar]
- Endovascular embolization of visceral artery pseudoaneurysms using modified injection technique with N-butyl cyanoacrylate glue. J Vasc Interv Radiol. 2015;26:1718-25.
- [Google Scholar]
- Endovascular management of pseudoaneurysms of the splenic artery: Experience with six patients. Australas Radiol. 2005;49:283-8.
- [Google Scholar]
- Properties of N-butyl cyanoacrylate-iodized oil mixtures for arterial embolization: In vitro and in vivo experiments. J Vasc Interv Radiol. 2012;23:1215-21.e1.
- [Google Scholar]
- Transcatheter arterial embolization of nonvariceal upper gastrointestinal bleeding with N-butyl cyanoacrylate. Korean J Radiol. 2007;8:48-56.
- [Google Scholar]
- Minimally invasive image-guided interventional management of hepatic artery pseudoaneurysms. Trop Gastroenterol. 2014;35:238-45.
- [Google Scholar]
- Endovascular management of pancreatitis related pseudoaneurysms. A single center experience. Gastrointest Interv. 2014;3:46-53.
- [Google Scholar]
- Percutaneous glue embolization of a visceral artery pseudoaneurysm in a case of sickle cell anemia. Cardiovasc Intervent Radiol. 2006;29:665-8.
- [Google Scholar]
- Direct puncture embolization using N-butyl cyanoacrylate for a hepatic artery pseudoaneurysm. Minim Invasive Ther Allied Technol. 2014;23:110-4.
- [Google Scholar]
- Clinical efficacy of transcatheter embolization of visceral artery pseudoaneurysms using N-butyl cyanoacrylate (NBCA) Diagn Interv Imaging. 2015;96:563-9.
- [Google Scholar]






