Translate this page into:
Embolized fibrin sheath material presenting as intraluminal calcifications within the pulmonary arteries on CT

*Corresponding author: Mihir Patel, Department of Radiology, New Jersey Medical School, Newark, New Jersey, United States. mihirpa@njms.rutgers.edu
-
Received: ,
Accepted: ,
How to cite this article: Patel M, Maldjian PD. Embolized fibrin sheath material presenting as intraluminal calcifications within the pulmonary arteries on CT. J Clin Imaging Sci 2022;12:39.
Abstract
Fibrin sheath formation is a well-described sequela of an indwelling central venous catheter. Fibrin sheaths may also develop around other foreign bodies within the venous system. We describe a case of fibrin sheath formation within the left brachiocephalic vein secondary to automatic implantable cardioverter-defibrillator (AICD) leads with subsequent embolization of sheath material presenting as calcifications within the pulmonary vasculature on computed tomography (CT). Most of the relevant literature focuses on catheter-related sheath formation and associated complications while reports on fibrin sheaths from other foreign bodies are sparse. We advise that radiologists who encounter intraluminal calcifications within the pulmonary arteries on CT should consider the possibility of a fibrin sheath as the source and search for its remnants in the central venous system for confirmation.
Keywords
Fibrin sheath
Fibrin sheath embolization
Pulmonary artery calcifications
Intraluminal calcifications
CT pulmonary arteries
INTRODUCTION
Fibrin sheaths are a patchwork of proteinaceous materials which arise from endothelial cell irritation and damage usually caused by long-term catheter placement.[1] Most fibrin sheaths form around central venous catheters which are used for a variety of reasons ranging from hemodialysis to fluid resuscitation.[2] On a computed tomography (CT) scan, a fibrin sheath usually appears as an irregular linear or tubular filling defect in the venous system with or without linear calcifications.[1] Histologic analysis shows that fibrin sheaths can have several different appearances ranging from a mesh-like thrombus that bridges venous walls and catheters to a mural thrombus. In addition, sheaths may remain in veins even after catheter removal. In such cases, retained sheaths can remain affixed to the venous walls. A retained fibrin sheath can lead to venous stenosis, thrombus formation, or pulmonary embolism and can serve as a nidus for infection. Most literature discussing fibrin sheaths focuses on vascular catheters as the etiology, however, other indwelling foreign bodies in the venous system can also result in fibrin sheath formation. Pacemaker leads within veins are known to cause fibrosis and venous thrombosis increasing the risk of vessel occlusion.[3] We present a case of embolization of material from a fibrin sheath that formed around a pacemaker wire manifesting as intraluminal calcifications within pulmonary artery branches on CT. We also discuss the differential diagnosis for this appearance.
CASE REPORT
Our patient is a 61-year-old man who presented to the emergency department with shortness of breath. His medical history was significant for congestive heart failure, coronary artery bypass graft surgery, hypertension, chronic obstructive pulmonary disease, diabetes mellitus type II, atrial fibrillation, and a right chest wall automatic implantable cardioverter-defibrillator (AICD). Three months prior to this admission, the patient had complete removal of a left chest wall AICD including the leads due to bacterial endocarditis with the formation of vegetations around the atrial portion of the ventricular lead. At that time a new right chest wall AICD with new leads was placed.
On presentation in the emergency department the patient was hypertensive (156/108 mmHg) and tachycardic (132 beats/min) with a regular rhythm. Physical examination showed jugular venous distention, pitting edema in both lower extremities, and bilateral rales. Laboratory tests were significant for leukocytosis, hyponatremia, hyperkalemia, elevated brain natriuretic peptide, and normal troponins. A chest radiograph showed cardiomegaly, parenchymal opacities consistent with pulmonary edema, and a right chest wall AICD with pacer lead projecting over the right atrium and shock coil projecting over the right ventricle. A subsequent CT scan of the chest was performed due to concern for pulmonary embolism. The CT scan showed bilateral ground-glass attenuation opacities in a perihilar and basilar distribution consistent with pulmonary edema. There was no evidence of acute pulmonary embolism. A calcified fibrin sheath was demonstrated in the left brachiocephalic vein. In addition, there were linear calcified opacities within the right interlobar and the left lower lobe segmental pulmonary arteries consistent with embolized fibrin sheath material [Figure 1]. The linear calcifications within the pulmonary arteries were not present on a chest CT scan performed 9 months prior to the current admission. The left brachiocephalic vein could not be adequately assessed for the presence of a fibrin sheath on the prior CT scan due to extensive artifacts surrounding the AICD wires which had not yet been removed at the time of that study. The patient was admitted to the telemetry service with a diagnosis of acute decompensated heart failure. After successful treatment with a course of diuretics, the patient was discharged 2 days later without incident.
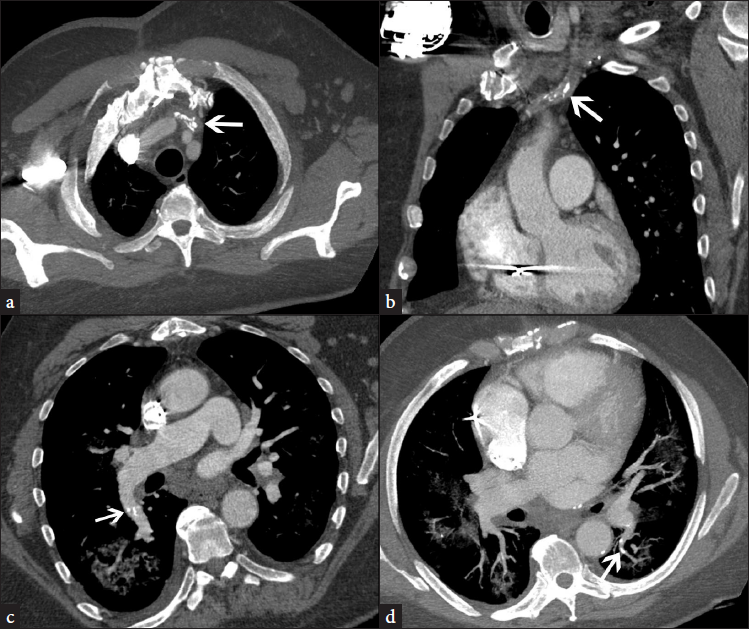
- A 61-year-old man presenting with heart failure found to have embolized fibrin sheath material on CT. Axial image from contrast-enhanced CT of the chest shows linear calcifications in left brachiocephalic vein (arrow) from the formation of a fibrin sheath. Figure 1b: Coronal reformatted image depicts linear calcifications from fibrin sheath in the left brachiocephalic vein (arrow). Figure 1c: Oblique reformatted image shows linear intraluminal calcifications within the right interlobar pulmonary artery (arrow). Figure 1d: Axial maximum intensity projection image shows intraluminal linear calcifications in segmental branch of left lower lobe pulmonary artery (arrow).
DISCUSSION
Fibrin sheaths are a patchwork of proteinaceous materials which arise from endothelial cell irritation and damage caused by long-term catheter placement.[1] Sheath formation involves endothelial cell damage leading to thrombus formation with smooth muscle activation, collagen, and calcium deposition.[2] The sheath is essentially a sleeve of thrombus that forms around the indwelling catheter. The sheath often remains in place after the removal of the catheter. One study revealed the prevalence of retained fibrin sheaths of 13.6% after removal of central venous catheters with nearly half of the sheaths demonstrating calcification on CT. Sheaths can vary in length from 1.5 to 16 cm and can be continuous or discontinuous in structure.[1]
Most literature regarding fibrin sheaths focuses on catheter-related sheath formation. Although the description of fibrin sheath formation from other indwelling sources is unusual, it has been reported to occur with AICD leads as in our case.[4] While fibrin sheath formation can occur around intravascular pacemaker leads, they are difficult to discern on CT due to the artifacts produced by the metallic pacemaker wires. The fibrin sheath would only be visible after the removal of the pacemaker lead in its entirety (as in our case) which is rarely necessary. We believe that manipulation of the previous AICD leads in the left brachiocephalic vein during removal of the device resulted in disruption of portions of the fibrin sheath with embolization of fragments to the pulmonary arteries.
The differential diagnosis for high attenuation within pulmonary arteries includes pulmonary artery calcification, calcification of chronic pulmonary thrombus, and embolization of foreign material such as catheter fragments, polymethacrylate cement, and iodinated oils used for therapeutic embolization of liver neoplasms. Pulmonary arterial calcifications occur within the vessel wall and are almost always due to pulmonary hypertension.[5] In our case, the calcified material is intraluminal rather than within the vessel wall. Chronic pulmonary artery thrombi from pulmonary thromboembolic disease can also calcify but these usually appear as soft tissue attenuation filling defects within the pulmonary arteries containing foci of calcification. Affected patients often also demonstrate enlarged central pulmonary arteries and dilated right cardiac chambers due to pulmonary hypertension.[6] Embolization of catheter fragments and iatrogenic embolization of foreign substances can be excluded if there is no corroborative history.
Fibrin sheaths are usually asymptomatic but can be associated with complications such as pulmonary embolism, infection, and thrombus formation. Pulmonary embolism during or after catheter removal can occur although the overall incidence is quite low (reportedly only up to 6%). Fibrin sheaths can form a biofilm that may serve as a nidus for the growth of microorganisms. Microorganism growth on fibrin sheaths can be asymptomatic, can result in catheter-related infection, or cause the formation of thrombus. Treatment for fibrin sheaths is usually reserved for symptomatic patients. Treatment options include percutaneous angioplasty, thrombolytic therapy, and fibrin sheath stripping. Surgical intervention may be appropriate in cases where fibrin sheaths have formed dense calcifications that are difficult to remove via percutaneous methods. Prophylactic treatment to prevent fibrin sheath formation includes an early catheter or device removal with the maintenance of serum calcium and phosphorus levels.[2]
CONCLUSION
In summary, we have presented a case of embolized fibrin sheath material presenting as intraluminal calcifications within the pulmonary arteries on CT. We advise that radiologists who encounter intraluminal calcifications within the pulmonary arteries on CT should consider the possibility of a fibrin sheath as the source and search for its remnants in the central venous system for confirmation.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Retained fibrin sheaths: Chest computed tomography findings and clinical associations. J Thorac Imaging. 2014;29:118-24.
- [CrossRef] [PubMed] [Google Scholar]
- Presence of retained calcified fibrin sheath after central venous catheter removal: A systematic literature review. J Vasc Access. 2020;23:644-52.
- [CrossRef] [PubMed] [Google Scholar]
- Neointimal fibrotic lead encapsulation—Clinical challenges and demands for implantable cardiac electronic devices. J Cardiol. 2017;70:7-17.
- [CrossRef] [PubMed] [Google Scholar]
- An unusual appearance of fibrin sheaths after implantable cardioverter-defibrillator removal because of infective endocarditis. Echocardiography. 2014;31:E257-8.
- [CrossRef] [PubMed] [Google Scholar]
- CT diagnosis of chronic pulmonary thromboembolism. RadioGraphics. 2009;29:31-50.
- [CrossRef] [PubMed] [Google Scholar]
- Approach to pulmonary hypertension: From CT to clinical diagnosis. RadioGraphics. 2018;38:357-73.
- [CrossRef] [PubMed] [Google Scholar]






