Translate this page into:
A Case of Metastatic Rectal Squamous Cell Carcinoma Initially Diagnosed as Lung Cancer
Address for correspondence: Dr. Aung Zaw Win, Department of Radiology, San Francisco VA Medical Center, 4150 Clement Street, San Francisco, California - 94121, USA. E-mail: aungzwin@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Squamous cell carcinoma (SCC) of the rectum is extremely rare with a reported incidence of only 0.025–0.1% of all colorectal tumors. The patient was a 68-year-old man who presented with fatigue, dry cough, shortness of breath, and unintentional weight loss. A chest CT revealed a left suprahilar mass suspected to be lung cancer and an initial diagnosis of primary lung cancer was made. However, fluorodeoxyglucose positron emission tomography and computed tomography (FDG-PET/CT) exam revealed an intensely hypermetabolic rectal mass which turned out to be rectal squamous cell carcinoma. This is the first report that shows FDG-PET/CT images of rectal squamous cell carcinoma metastasis to the skin, muscle, bone, and lung. Use of PET/CT in the initial diagnosis of non-resectable rectal cancers may avoid unnecessary surgery.
Keywords
Bone
fluorodeoxyglucose positron emission tomography and computed tomography
metastasis
muscle
rectal squamous cell carcinoma
skin nodule
INTRODUCTION

Rectal cancers are typically more aggressive than colon cancers.[1] In World Health Organization (WHO) classification, a number of histologic variants of colorectal carcinomas are listed, such as mucinous, signet ring cell, medullary, micropapillary, serrated, cribriform comedo-type, adenosquamous, spindle cell, and undifferentiated.[1] Other rare types of colorectal carcinomas include neuroendocrine and squamous cell carcinomas.[1] More than 98% of colorectal cancers are adenocarcinomas.[2] Squamous cell carcinoma (SCC) of the rectum is extremely rare with a reported incidence of only 0.025–0.1% of all colorectal tumors.[3] Smoking and alcohol consumption are more strongly associated with rectal cancer than colon cancer.[3] The liver and regional lymph nodes are the most common sites of metastasis for colorectal cancers.[2] Moreover, rectal carcinomas are more prone to metastasize to the lymph nodes than colon carcinomas.[2] Rectal SCC is more aggressive than adenocarcinoma and has a worse prognosis.[3] There is currently no effective and satisfactory treatment for this disease. Fluorodeoxyglucose positron emission tomography and computed tomography (FDG-PET/CT) imaging can provide both functional and anatomical data.
CASE REPORT
The patient was a 68-year-old man with a history of smoking, alcohol use, and emphysema, who presented with a 3-month history of fatigue, dry cough, shortness of breath, and unintentional weight loss. He had a family history of lung cancer. A chest CT found a left suprahilar mass suspected to be lung cancer. Laboratory values were: Urine sodium 73 mEq/l (normal 20 mEq/l), urine osmolality 1012 mOsm/kg (normal 500–800 mOsm/kg), serum sodium 128 mmol/l (normal 136–145 mmol/l), and serum potassium 3.1 mmol/l (normal 3.5–5.0 mmol/l). FDG-PET/CT exam revealed an intensely hypermetabolic rectal mass and perirectal lymphadenopathy, raising the suspicion for primary malignancy [Figure 1]. In addition, a large liver mass and a skin nodule on the forehead, both with high FDG uptake, were observed [Figure 2]. A CT-guided fine needle aspiration (FNA) of the liver revealed CK7-, p63-, p16-positive, poorly differentiated SCC. A punch biopsy of the solitary skin nodule on the left upper forehead showed metastatic SCC. The mass in distal rectum was biopsied by flexible sigmoidoscopy and pathology confirmed it as SCC. Lesions in the left iliac wing and left gluteus medius muscle were also discovered by FDG-PET/CT [Figure 2] and biopsy confirmed them as rectal SCC. The patient was referred for hospice care.
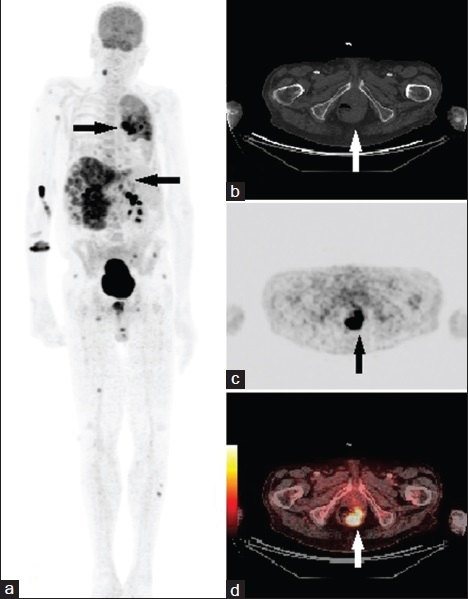
- 68-year-old man presented with fatigue, dry cough, shortness of breath, and unintentional weight loss. He was finally diagnosed with rectal squamous cell carcinoma. (a) Whole body maximum intensity projection (MIP) fluorodeoxyglucose positron emission tomography and computed tomography (FDG-PET/CT) image shows widespread metastasis (arrows) of rectal SCC. (b–d) FDG-PET/CT axial view of rectum show a 4-cm left eccentric intensely hypermetabolic rectal mass (arrows) with maximum standardized uptake value (SUVmax) of 10.6.
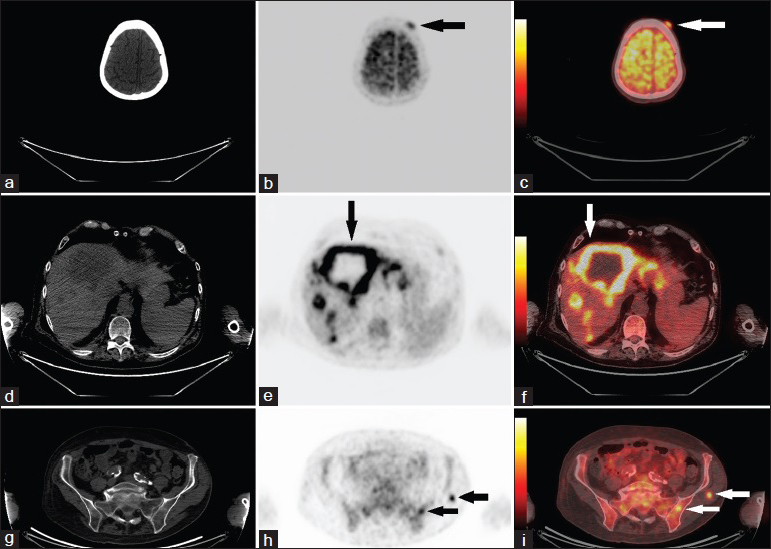
- 68-year-old man presented with fatigue, dry cough, shortness of breath, and unintentional weight loss. He was finally diagnosed with rectal squamous cell carcinoma. (a–c) FDG-PET/CT axial view of skull shows a hypermetabolic 12-mm soft tissue nodule (arrows) within the left frontal scalp, with an SUVmax of 3.7. (d–f) FDG-PET/CT axial view of enlarged liver show innumerable hypermetabolic metastatic masses (arrows), many of which demonstrate central necrosis. (g–i) FDG-PET/CT axial view of pelvis show metastasis (arrows) in the left medial iliac wing and in the left gluteus medius muscle (SUVmax 5.2).
DISCUSSION
To our knowledge, this is the first report that shows FDG-PET/CT images of rectal SCC metastasis to the skin, muscle, bone, and lung. There is one report of rectal SCC metastasis to the lung, brain, spine, and skin (lower chin).[4] There is also another case of rectal SCC metastasis to the lumbar vertebrae.[5] Breast cancer is the most common primary cancer followed by lung cancer with cutaneous metastasis.[6] The chest is the most common site of cutaneous metastasis. In general, adenocarcinomas give rise to cutaneous metastases at a higher frequency, compared with other histologic subtypes.[6] In this case, the skin metastasis presented as a non-tender mobile skin nodule on the left forehead. The presentation of rectal SCC can be nonspecific and patients tend to present with advanced stage disease. Our patient presented with nonspecific complaints of fatigue and unintentional weight loss. Poorly differentiated feature carries a bad prognosis.
In rectal cancer, distant metastasis can occur by both lymphatogenous and vascular drainage routes.[7] The tumor in the present case was located in the distal rectum. The distal rectum is drained by the inferior rectal vein, which in turn drains into the common iliac vein and then into the inferior vena cava. So, systemic metastasis can occur via this route. Moreover, compared to upper rectal cancers, distal rectal and mid-rectal cancers are more likely to metastasize to the lungs and bone.[7] In fact, Chiang et al., reported that distant metastasis occurs in the following sequential order: Liver, lungs, bone, brain, and other systemic sites.[7]
At first, hyponatremia and hypo-osmolality in our patient raised the suspicion of inappropriate antidiuretic hormone hypersecretion (SIADH). Small cell lung cancers can have ectopic production of antidiuretic hormone (ADH) and this can lead to SIADH. Our patient had an extensive history of smoking and alcohol consumption, and his family history was positive for lung cancer. This, in addition to the CT findings and physical complaints (dry cough, shortness of breath, and unintentional weight loss), further supported the diagnosis of lung cancer. Only after the FDG-PET/CT exam, the true diagnosis of rectal SCC metastasis was made.
PET/CT is more accurate than CT and MRI in staging rectal tumors and assessing response to therapy.[8] Restaging of rectal cancer with MRI can be challenging due in part to post-treatment fibrosis and reaction, whereas FDG-PET is not affected by scar tissue.[9] Both CT and MRI use size as the main criterion in the evaluation of nodal involvement, although the lymph node size is not an ideal indicator of metastasis and lacks sufficient accuracy for clinical decision-making. Moreover, both CT and MRI have low accuracy for identifying the depth of tumor infiltration.[10] A prospective study reported an improved survival after preoperative staging with FDG-PET.[9] FDG-PET can detect recurrent disease earlier than conventional imaging and as a result, those lesions can be curatively resected.[9] Similar to this case, Llamas-Elvira et al., found that pre-surgical staging with FDG-PET can reveal unsuspected metastases.[10]
CONCLUSION
FDG-PET/CT can scan the whole body for metastases in one exam. On the other hand, chest/abdomen/pelvis CT exams are required to accomplish the same task. In the present case, thoracic CT missed the primary rectal cancer and only detected pulmonary metastasis. Use of PET/CT in the initial diagnosis of non-resectable rectal cancers may avoid unnecessary surgery. Thus, PET/CT can be a valuable tool in the initial staging of rectal cancers.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2015/5/1/22/156125
Source of Support: No funding was received for this work
Conflict of Interest: The authors declare that they have no conflict of interest.
REFERENCES
- Treatment of primary rectal squamous cell carcinoma by primary chemoradiotherapy: Should surgery still be considered a standard of care? Eur J Cancer. 2008;44:2340-3.
- [Google Scholar]
- Role of human papillomavirus in squamous cell metaplasia-dysplasia-carcinoma of the rectum. Am J Surg Pathol. 2007;31:919-25.
- [Google Scholar]
- Metastatic cystic nodule of rectal SCC with basaloid features mimicking a BCC of the face. J Am Acad Dermatol. 2012;67:e160-2.
- [Google Scholar]
- Rates of cutaneous metastases from different internal malignancies: Experience from a Taiwanese medical center. J Am Acad Dermatol. 2009;60:379-87.
- [Google Scholar]
- Rectal cancer level significantly affects rates and patterns of distant metastases among rectal cancer patients post curative-intent surgery without neoadjuvant therapy. World J Surg Oncol. 2014;12:197.
- [Google Scholar]
- Comparison of CT, MRI and FDG-PET in response prediction of patients with locally advanced rectal cancer after multimodal preoperative therapy: Is there a benefit in using functional imaging? Eur Radiol. 2005;15:1658-66.
- [Google Scholar]
- Controversies in the management of colorectal liver metastases: Role of PET and PET/CT. Dig Surg. 2008;25:413-20.
- [Google Scholar]
- Fluorine-18 fluorodeoxyglucose PET in the preoperative staging of colorectal cancer. Eur J Nucl Med Mol Imaging. 2007;34:859-67.
- [Google Scholar]






