Translate this page into:
Unusual Benign Tumors of the Breast
Address for correspondence: Dr. Gary J Whitman, Department of Diagnostic Radiology, University of Texas MD Anderson Cancer Center, Unit 1350, PO Box 301439, Houston, Texas, 77230-1439, USA. E-mail: gwhitman@mdanderson.org
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The purpose of this article is to describe the imaging characteristics of a variety of benign breast tumors that may be encountered in daily practice, in order to formulate an appropriate differential diagnosis and to establish concordance between the imaging and the pathologic findings, and to assist the clinician with appropriate management.
Keywords
Benign breast masses
breast masses
breast tumors
INTRODUCTION

- Dr. Beatriz E. Adrada
There are a number of benign tumors of the breast with a wide range of imaging appearances. Benign breast tumors may appear suspicious for malignancy. In this article, we discuss the imaging and the pathologic appearances of several rare benign tumors in order to help radiologists to make appropriate differential diagnoses.
Pseudoangiomatous stromal hyperplasia
Pseudoangiomatous stromal hyperplasia (PASH) is a mesenchymal tumor of the breast with a broad range of clinical and imaging presentations.[1] Clinically, PASH varies from completely asymptomatic to a palpable mass, affecting premenopausal and perimenopausal women.[2] On mammography, PASH can be depicted as oval or round non-calcified masses [Figure 1], focal asymmetries, or it can be mammographically occult. On ultrasound, PASH appears as oval, circumscribed masses with homogeneous or heterogeneous echotexture.[1] Often, it is difficult to distinguish PASH and fibroadenomas based on imaging. On magnetic resonance imaging (MRI), PASH can be seen as masses with variable enhancement or areas of non-mass enhancement, often with Type I curves on kinetic assessment.[3] Histologically, PASH is characterized by a proliferation of fibroblasts and myofibroblasts with slit-like spaces lined by spindle cells and dense collagenous stroma. Pathologically, angiosarcoma is a differential consideration. Hormonal factors have been implied in the development of PASH. Biopsy is necessary for diagnosing PASH. Close follow-up for interval growth is recommended.

- 49-year-old woman with pseudoangiomatous stromal hyperplasia (PASH). (a) Right craniocaudal mammogram shows an oval mass with obscured margins at the 12 o’clock position (arrows). (b) Longitudinal ultrasound shows that the mass is isoechoic with indistinct margins (arrows). (c) Power Doppler ultrasound shows minimal vascularity (arrow). (d) Photomicrograph of the core biopsy specimen (hematoxylin and eosin stain, ×20) shows benign breast tissue with slit-like spaces lined by spindle cells representing myofibroblasts, mimicking vascular spaces. Note that the spaces are empty and do not contain red blood cells as in true vascular spaces.
Diabetic mastopathy
Diabetic mastopathy is a rare fibrous condition in the breast that occurs with either long-standing Type I or Type II diabetes.[4] Histologically, diabetic mastopathy comprises lymphocyte proliferation and dense stromal fibrosis. Clinically, single or multiple non-tender, palpable masses are present. On mammography, features of diabetic mastopathy include asymmetries, architectural distortions, or masses [Figure 2].[5] On sonography, diabetic mastopathy manifests as hypoechoic masses with irregular margins and marked posterior acoustic shadowing.[4] Breast MRI can help to resolve equivocal findings on mammography or ultrasound. The enhancement of these lesions has been reported as patchy with persistent curves on kinetic assessment.[6]
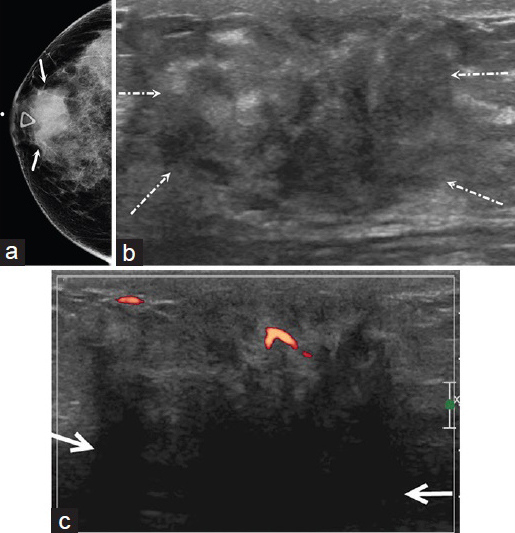
- 30-year-old woman with diabetic mastopathy. The patient had a history of Type I diabetes and presented with a palpable abnormality in the right breast. (a) Right craniocaudal mammogram demonstrates a round mass with obscured margins (arrows) in the retroareolar region. (b) Transverse ultrasound shows a heterogeneous mass with irregular margins (arrows). (c) Power Doppler ultrasound shows minimal vascularity and marked posterior acoustic shadowing (arrows).
Granulomatous mastitis
Granulomatous mastitis is a rare benign inflammatory condition seen in the breast. It is usually related to pregnancy, lactation, or the use of contraceptives.[7] The etiology is not clear and several infectious, hormonal and autoimmune factors have been implicated. The most commonly accepted hypothesis is that granulomatous mastitis is an autoimmune disease.[7] It is a histologic diagnosis of exclusion. Clinically, the most common finding is a breast mass. Inflammatory signs, including redness, skin thickening, and peau d’orange, may be identified.[7] Sinus tracts and abscesses are sometimes present. The mammographic findings are not specific, which include focal asymmetries, ill-defined masses, or regions of diffuse increased density.[8] Sonographic features include hypoechoic masses with associated tubular and finger-like structures[Figure 3], irregular heterogeneous hypoechoic masses, and areas of architectural distortion.[8]

- 31-year-old woman with chronic granulomatous mastitis in the left breast. She presented for evaluation of redness in the skin and a palpable abnormality in the upper outer quadrant of the left breast. (a) Extended field of view ultrasound shows a hypoechoic mass associated with a tubular structure (arrows) at the 2 o’clock position, corresponding to the area of the palpable abnormality. (b) Power Doppler ultrasound shows peripheral hypervascularity (arrows).
Biopsy is necessary to confirm the clinical suspicion of granulomatous mastitis. The treatment of choice remains controversial. Some investigators advocate surgical excision, whereas others favor medical treatment with steroid therapy and close surveillance.[78]
Sarcoidosis
Sarcoidosis is a granulomatous disease of unknown etiology. The breasts are involved in less than 1% of cases when the sarcoidosis is usually systemic.[9] Sarcoidosis typically affects Afro-American women in their third and fourth decades of life. Histologically, non-caseating granulomas are characteristic of sarcoidosis. Palpable masses are common clinical presentations of sarcoidosis. Mammographically, irregular breast masses and/or axillary lymphadenopathy have been described in sarcoidosis.[9] On ultrasound, sarcoidosis presents as irregular and spiculated masses, mimicking malignancy. Core biopsy is essential to establish the diagnosis of sarcoidosis. Breast MRI is not beneficial, since sarcoidosis cannot be distinguished from breast cancer.[10]
Tubular adenoma
Tubular adenomas are rare benign tumors, accounting for about 4% of all benign breast benign lesions. Tubular adenomas are commonly seen in young women.[11] Histologically, tubular adenomas differ from fibroadenomas, as tubular adenomas are formed from packed acini with sparse stroma. On mammography, tubular adenomas are oval or lobulated masses with circumscribed margins. Ultrasound often demonstrates the benign nature of tubular adenomas[Figure 4]. On sonography, tubular adenomas are seen as oval or lobulated masses, and the echotexture is usually homogeneous.[11] Biopsy of these lesions is necessary to exclude malignancy. If there is imaging–pathologic concordance, no further work-up is needed.

- 29-year-old woman with a tubular adenoma. The patient had a history of metastatic carcinoid tumor. Multiple masses had been documented in both breasts. (a) Axial post-contrast T1-weighted MRI shows several enhancing masses in both breasts. The most suspicious mass is noted in the medial posterior left breast, along the chest wall (arrow). (b) Longitudinal ultrasound shows a 2.5 cm lobular hypoechoic mass (arrows) at the 9 o’clock position, correlating with the mass seen on MRI. (c) Photomicrograph of the core biopsy specimen (hematoxylin and eosin stain, ×100) shows a well-circumscribed, benign, fibroepithelial tumor composed of glands and mildly cellular stroma (arrows). The glands are benign-appearing with an inner luminal layer and an outer myoepithelial cell layer. The spindle cells in the stroma did not exhibit any pleomorphism or mitotic activity.
Fibromatosis
Fibromatosis is an infiltrative proliferation of fibroblasts and myofibroblasts from musculo-aponeurotic tissue. Fibromatosis is extremely rare, constituting 0.2% of all breast tumors.[12] Surgical procedures such as reductions and augmentations have been shown to be associated with fibromatosis. Fibromatosis tends to be solitary and unilateral, with the condition found in the age range of 14–83 years.[13] Fibromatosis of the breast usually presents as a painless, hard mass. Because of its infiltrative nature, fibromatosis may demonstrate tethering of the skin, nipple retraction, or it may be inseparable from the chest wall. On mammography, fibromatosis is characterized as an irregular, spiculated mass.[1213] On sonography, fibromatosis is depicted as an irregular solid mass with angular margins and posterior acoustic shadowing[Figure 5]. Since fibromatosis is a locally aggressive neoplasm, breast MRI has been shown to be a useful diagnostic modality in the evaluation of the extent of disease, specifically regarding possible involvement of the pectoralis muscle and the chest wall.[12] Fibromatosis is usually diagnosed with imaging-guided core biopsy. Surgical treatment with grossly negative margins is the recommended treatment, since fibromatosis may recur in up to 29% of cases.[13]

- 69-year old woman with fibromatosis in the right breast. (a) Right craniocaudal mammogram demonstrates an irregular, non-calcified mass (arrows) at the 1 o’clock position. (b) Transverse ultrasound shows a 1 cm, irregular, hypoechoic mass (arrows). (c) Power Doppler ultrasound shows marked vascularity. (d) Photomicrograph of the surgical specimen (hematoxylin and eosin stain, ×50) shows spindle cell proliferation with invasive edges (arrows). There are relatively bland spindle cells with inconspicuous mitosis and no nuclear pleomorphism.
Myofibroblastoma
Myofibroblastomas are spindle cell tumors composed of a uniform population of spindle cells, scattered multinucleated cells, and interspersed bands of collagen.[12] Myofibroblastomas occur frequently in older men and in post-menopausal women.[14] These masses usually present as slow-growing, palpable, painless, movable masses. They range from 1 to 3 cm in diameter. On mammography, myofibroblastomas are found as circumscribed, non-calcified tumors[Figure 6]. On ultrasound, myofibroblastomas are usually seen as oval, well-marginated masses.[15] The internal echotexture is commonly hypoechoic, but heterogeneous and hyperechoic masses have been described. The diagnosis can be made by image-guided biopsy. The treatment is surgical excision.[1415]

- 78-year-old man with a myofibroblastoma in the left breast. The patient had a history of renal and lung cancers and presented for evaluation of an incidental left breast mass seen on chest CT. (a) Axial contrast-enhanced chest CT shows a circumscribed enhancing mass (arrow) in the left breast. (b) Left mediolateral oblique mammogram demonstrates a 1 cm, well-circumscribed, non-calcified, homogeneously dense mass (arrow) at the 11 o’clock position. (c) Transverse ultrasound of the left breast shows a well-circumscribed, hypoechoic mass (arrows) at the 11 o’clock position. (d) Longitudinal power Doppler ultrasound shows marked vascularity (arrows). (e) Photomicrograph of the surgical specimen (hematoxylin and eosin stain, ×100) demonstrates a relatively well-circumscribed mass (arrows) with predominantly epithelioid cells.
Schwannoma
Schwannomas are benign tumors arising from the Schwann cells of the nerve sheaths. Schwannomas in the breast are extremely rare, with a reported incidence of 2.4% of all schwannomas.[16] The reported age range is from 18 to 83 years, with a mean age of 47 years.[16] Schwannomas may present as slow-growing, palpable masses, or they may be asymptomatic. On mammography, schwannomas have been described as well-circumscribed masses without calcifications. Ill-defined densities have also been reported.[17] On ultrasound, the appearance of schwannomas is variable. The masses are generally circumscribed. The internal echotexture varies from homogeneous hypoechoic to partially cystic[Figure 7]. Differential diagnosis includes fibroadenomas, papillary lesions, and carcinomas. The definitive diagnosis is established by fine needle aspiration (FNA) or core biopsy. If FNA reveals a spindle cell neoplasm, schwannoma should be considered in the differential diagnosis. The treatment is local excision with complete enucleation of the tumor including the capsule.[1617] Recurrence is rare.
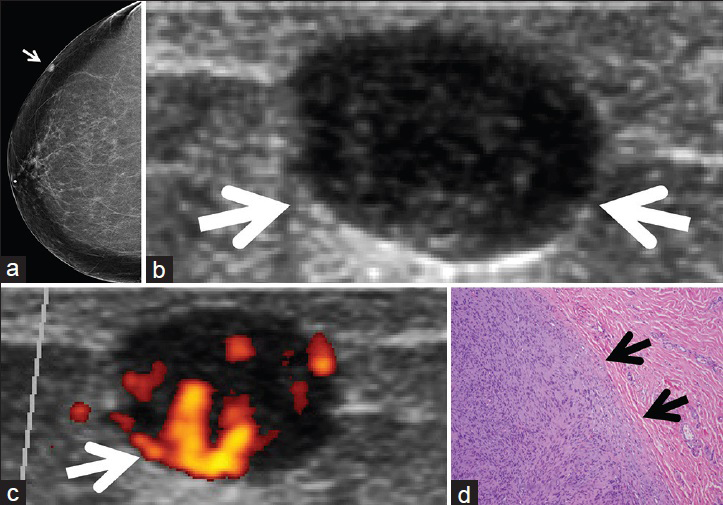
- 64-year-old-woman with a schwannoma in the right breast. (a) Craniocaudal mammogram shows a 0.8 cm, circumscribed, non-calcified, superficial, oval mass (arrow) in the lateral aspect of the right breast. (b) Ultrasound of the right breast shows a well-circumscribed, hypoechoic mass (arrows), correlating with the mass seen on mammography. (c) Power Doppler ultrasound of the right breast mass shows high vascularity (arrow). Ultrasound-guided fine needle aspiration was performed, showing a spindle cell neoplasm. (d) Photomicrograph of the surgical specimen (hematoxylin and eosin stain, ×100) shows a well-circumscribed tumor composed of spindle cells with moderate pleomorphism and inconspicuous mitotic activity (black arrows point to the well-circumscribed edge of the tumor).
Granular cell tumors
Granular cell tumors are extremely rare tumors of neural origin. Granular cell tumors have been associated with premenopausal African-American women. The mammographic findings are variable, usually with masses with indistinct or spiculated margins.[18] On sonography, granular cell tumors mimic malignant tumors. Granular cell tumors may be hypoechoic or heterogeneous with irregular or circumscribed margins, with or without posterior acoustic shadowing[Figure 8]. A few cases have been reported showing the MRI characteristics of granular cell tumors. On MRI, granular cell tumors have been reported as enhancing masses with morphologic features suspicious of malignancy. The dynamic contrast enhancement pattern may show wash-in and wash-out or a progressive curve.[19] Breast MRI is a valuable adjunctive imaging tool to evaluate the extent of the disease and to rule out involvement of the pectoralis muscle. The imaging findings are often indistinguishable from breast carcinoma; therefore, image-guided biopsy is required for diagnosis. Because granular cell tumors are locally aggressive, wide excision is recommended.[1819]

- 46 year-old African-American woman with a granular cell tumor of the right breast. The patient was a BRCA1 carrier and she presented for evaluation of a palpable abnormality in the right axilla. (a) Lateromedial mammogram shows a spiculated mass (arrow) in the right axillary region. (b) Longitudinal right breast ultrasound shows a 6-cm spiculated mass (arrows) with marked posterior acoustic shadowing. (c) Axial T1-weighted MRI with fat saturation demonstrates the enhancing tumor (arrows) involving the pectoralis muscle. (d) Photomicrograph (S100 immunostain, ×100) shows that the tumor cells are strongly positive for S100 immunostain.
CONCLUSION
This review has emphasized the importance of recognizing the characteristics of rare benign tumors in the breast, as some rare tumors may be included in differential diagnoses in daily clinical practice. Knowledge of the histopathologic findings related to these rare tumors is also necessary to evaluate concordance between the imaging and the pathologic findings.
ACKNOWLEDGMENT
The University of Texas MD Anderson Cancer Center is supported in part by the National Institutes of Health through Cancer Center Support Grant CA16672.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2015/5/1/27/157603
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Pseudoangiomatous stromal hyperplasia (PASH) of the breast: A series of 24 patients. Breast J. 2012;18:242-7.
- [Google Scholar]
- Pseudoangiomatous stromal hyperplasia (PASH) of the breast: A clinicopathological study of 79 cases. Int J Surg Pathol. 2012;20:54-8.
- [Google Scholar]
- Pseudoangiomatous stromal hyperplasia: Imaging findings with pathologic and clinical correlation. AJR Am J Roentgenol. 2010;195:1036-42.
- [Google Scholar]
- Diabetic mastopathy: A diagnostic challenge in breast sonography. J Clin Ultrasound. 2015;43:113-7.
- [Google Scholar]
- Fibrous lesions of the breast: Imaging-pathologic correlation. Radiographics. 2005;25:1547-59.
- [Google Scholar]
- Dynamic contrast enhanced magnetic resonance imaging and magnetic resonance spectroscopy in diabetic mastopathy. Breast. 2005;14:68-70.
- [Google Scholar]
- Granulomatous mastitis: Clinical, pathological features, and management. Breast J. 2010;16:176-82.
- [Google Scholar]
- Granulomatous lobular mastitis: Imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193:574-81.
- [Google Scholar]
- Radiologic evaluation of uncommon inflammatory and reactive breast disorders. Radiographics. 2005;25:411-24.
- [Google Scholar]
- Primary sarcoid of the breast with incidental malignancy. J Clin Imaging Sci. 2012;2:46.
- [Google Scholar]
- Tubular adenomas of the breast: Imaging findings with histologic correlation. AJR Am J Roentgenol. 2000;174:757-61.
- [Google Scholar]
- Mammary fibromatosis.A benign spindle-cell tumor with significant risk for local recurrence. Cancer. 1989;63:1363-69.
- [Google Scholar]
- Mammary myofibroblastoma: A tumor with a wide morphologic spectrum. Arch Pathol Lab Med. 2008;132:1813-20.
- [Google Scholar]
- Sonographic findings in solitary schwannoma of the breast. J Clin Ultrasound. 2001;29:99-101.
- [Google Scholar]
- Sonographic and mammographic appearances of granular cell tumors of the breast with pathological correlation. J Clin Ultrasound. 2006;34:153-60.
- [Google Scholar]
- Magnetic resonance appearance of granular cell tumor of the breast. Clin Imaging. 2009;33:395-7.
- [Google Scholar]






