Translate this page into:
Thoracic Presentations of Small Round Blue Cell Tumors
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The term “small round blue cell” is frequently used as a cursory radiologic pathological correlation of aggressive tumors throughout the body. We present a pictorial essay of common and uncommon subtypes of small round blue cell tumors in the chest illustrating the characteristic radiologic findings of each lesion. In addition, we review the pathologic findings of each tumor subtype with characteristic hematoxylin- and eosin-stained photomicrographs and immunohistochemical and molecular studies. Represented tumors include small cell carcinoma, Ewing sarcoma, extranodal marginal zone B-cell lymphoma, embryonal rhabdomyosarcoma, desmoplastic small round cell tumor, and posttransplant lymphoproliferative disorder. Understanding and ability to recognize these lesions are essential to broaden the radiologist's differential diagnosis and help guide patient care.
Keywords
Radiologic-pathologic correlation
small round blue cell tumors
thoracic small round blue cell tumors

INTRODUCTION
The term small round blue cell tumors (SRBCTs) refers to similar appearing neoplasms composed of small, round, basophilic cells on hematoxylin and eosin (H and E) staining. Within the thorax, many SRBCTs appear as nonspecific pulmonary nodules or masses on imaging. Therefore, accurate diagnosis relies on a combination of clinical presentation, radiographic studies, and pathologic specimens. In this pictorial essay, we present a radiologic and pathologic review of SRBCTs in the thorax highlighting the characteristic radiologic images, histomorphology, immunohistochemistry, and molecular studies utilized to arrive at the diagnosis. Represented tumors include small cell carcinoma, Ewing sarcoma, extranodal marginal zone B-cell lymphoma, embryonal rhabdomyosarcoma, desmoplastic small round cell tumor, and posttransplant lymphoproliferative disorder (PTLDs).
SMALL CELL LUNG CARCINOMA
Small cell lung carcinoma (SCLC) is a common, rapidly growing neoplasm that has a strong correlation with cigarette smoking.[1] Patients present clinically with a cough, hemoptysis, or postobstructive pneumonia.[1] Radiographically, SCLC presents as a centrally located bulky hilar/perihilar mass with associated adenopathy. Complications include displacement or narrowing of the tracheobronchial tree or vessels, pleural effusions, and/or metastases [Figure 1]. Histologically, SCLC is characterized by densely packed neuroendocrine cells with high mitotic activity. Positive immunostaining for chromogranin A or synaptophysin is confirmatory [Figure 2].
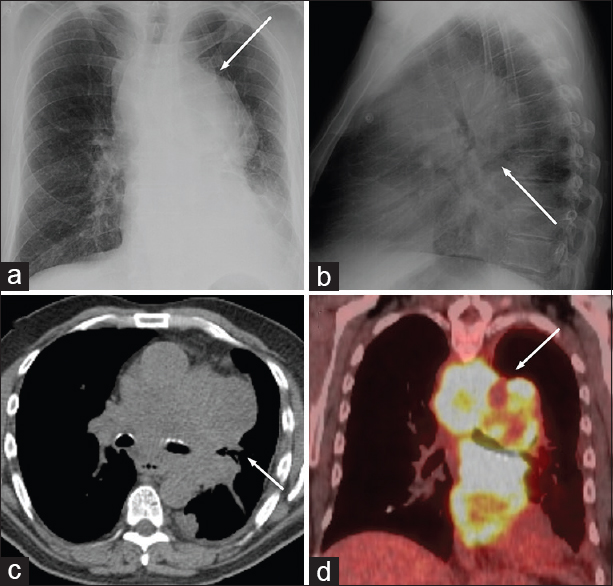
- 59-year-old female smoker with shortness of breath diagnosed with small cell carcinoma. (a and b) Frontal and lateral chest radiographs show a large right mediastinal mass (arrow). (c) Axial computed tomography image on soft tissue windows demonstrates a multilobulated mass encasing the hilar structures (arrow). (d) Subsequent positron emission tomography scan shows an intensely fludeoxyglucose avid mass (arrow).

- Pathology from patient in Figure 1. (a) Gross pathology shows white nodules compressing the bronchioles (arrow). (b) Densely packed, haphazardly arranged, monotonous sheets of small tumor cells interspersed with areas of necrosis (H and E, ×100). (c) Positive cytoplasmic immunoreactivity for synaptophysin supports neuroendocrine differentiation (brown, arrow).
EWING SARCOMA
Ewing sarcoma commonly presents with nonspecific localized bony pain in children and adolescents. The most common locations include the pelvis, femur, spine, ribs, and skull.[2] Radiographically, they present as an osseous lesion with aggressive periosteal reaction and soft tissue mass.[23] Extra-osseous Ewing sarcoma can present in the thorax and arise from chest wall structures or soft tissues (Askin tumor), presenting as a pleural-based mass with an associated effusion.[3] Histologically, they are characterized by sheets of small round cells (or rarely spindled cells) [Figure 3]. Positive immunostaining for CD99 [Figure 4] and t(11;22)(q24;q12) chromosomal translocation are distinguishing features.

- A 7-year-old female presenting with a cough with concern for pneumonia refractory to antibiotic treatment, diagnosed with Ewing sarcoma. (a) Axial computed tomography image on soft tissue windows shows a large necrotic soft tissue mass involving the right eighth rib (arrow). (b) There is increased radiotracer uptake in the same region on bone scan (arrow).
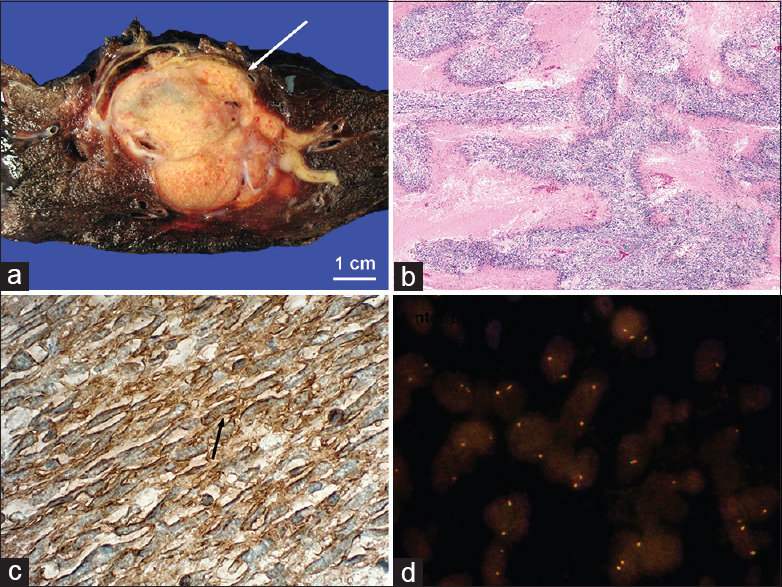
- Pathology from patient in Figure 3. (a) Gross pathology shows a multilobular, yellow tumor arising from the chest wall (arrow). (b) Extensive geographic zones of necrosis with tumor cells around blood vessels imparting a striking “filigree pattern” (H and E, ×40). (c) Strong immunoreactivity for CD99 confirms primitive neuroectodermal differentiation (brown, arrow). (d) Fluorescent in situ hybridization demonstrates negative SYT-SSX fusion gene which rules out the translocation found in synovial sarcomas and indirectly supports Ewing sarcoma.
EMBRYONAL RHABDOMYOSARCOMA
Rhabdomyosarcoma is the most common childhood soft tissue tumor and often arises from the head, neck, and genitourinary system.[4] Isolated thoracic rhabdomyosarcomas are unusual but can arise from the heart, lungs, pleura, airways, or mediastinum.[4] Radiographically, they often present as a soft tissue mass, which erodes into the adjacent bone. Metastatic disease can present as pulmonary/pleural nodules [Figure 5]. The unifying histomorphologic feature is neoplastic cells that resemble embryonic skeletal myoblasts, which display positive immunostaining for myogenin and/or MyoD1 [Figure 6].
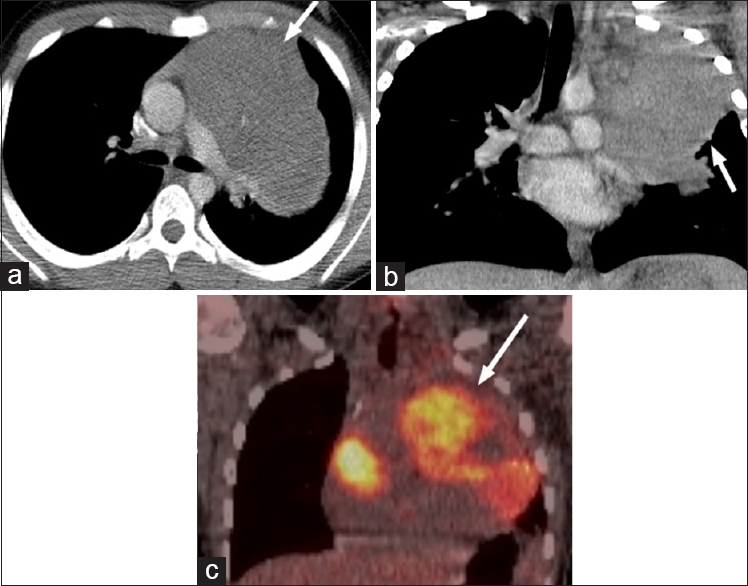
- 13-year-old male presenting with a cough diagnosed with embryonal rhabdomyosarcoma. (a and b) Axial and coronal computed tomography images on soft tissue windows demonstrate a multilobulated left anterior mediastinal mass (arrows). (c) Subsequent PET scan demonstrates that this FDG avid lesion is contiguous with the pericardium (arrow).
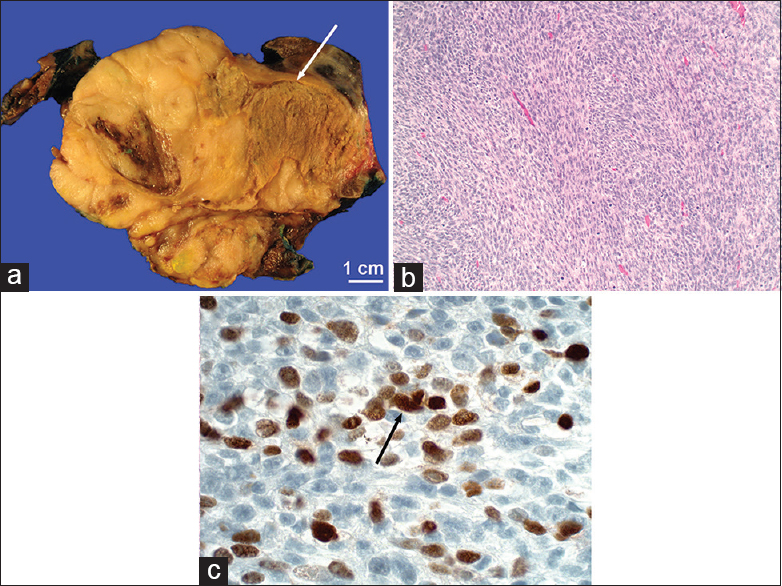
- Pathology from patient in Figure 5. (a) Gross pathology shows a tan, poorly circumscribed tumor (arrow). (b) Fascicles of fusiform cells (H and E, ×100). (c) Positive immunoreactivity for myogenin confirms primitive skeletal muscle differentiation (brown, arrow).
DESMOPLASTIC SMALL ROUND CELL TUMOR
Desmoplastic small round cell tumors are rare neoplasms which typically arise in the abdomen and present with abdominal pain.[5] Radiologic imaging demonstrates multiple abdominopelvic masses with a dominant mass arising from the retrovesical/retrouterine space.[5] Thoracic dissemination may manifest as pulmonary nodules or pleural involvement [Figure 7].[5] The defining histologic feature is that of desmoplastic stroma enveloping irregular nests of cells [Figure 8]. Positive immunostaining for vimentin [Figure 8] and t(11;22)(p13;q12) chromosomal translocation are distinguishing features.[6]

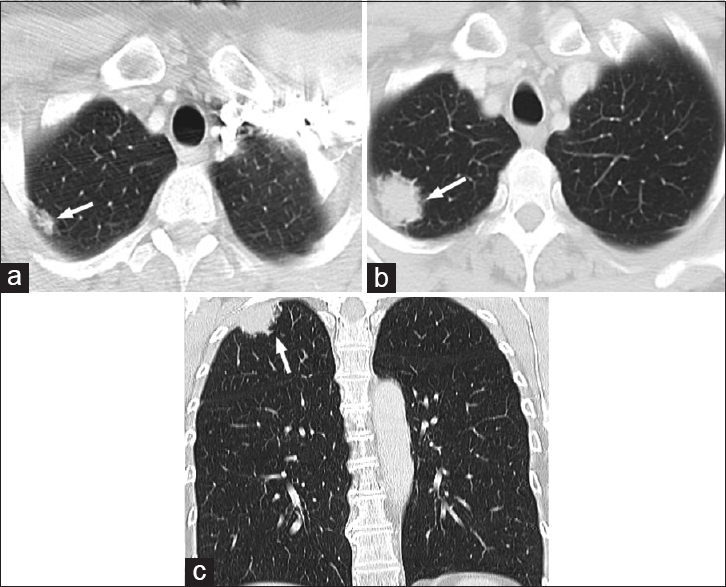
- 12-year-old female with left shoulder pain diagnosed with desmoplastic small round tumor. (a) Axial computed tomography section on lung windows demonstrate a pulmonary nodule in the right lower lobe, which progressively enlarged over a year (b) (arrows).
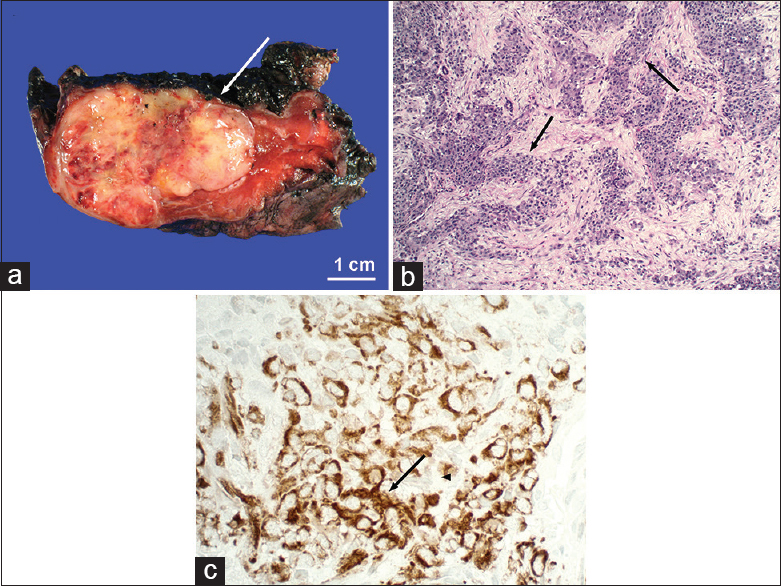
- Pathology from patient in Figure 7. (a) Gross pathology shows grey-white pulmonary nodules with foci of necrosis and hemorrhage (arrow). (b) Multiple sharply demarcated nests surrounded by prominent desmoplastic stroma (arrows) (H and E, ×100). (c) Positive immunoreactivity for vimentin confirms mesenchymal differentiation (brown, arrow).
EXTRANODAL MARGINAL B-CELL LYMPHOMA
Extranodal marginal B-cell lymphoma or mucosa-associated lymphoid tissue, often arises from the gastrointestinal tract.[7] Within the lungs, they can present as metastatic disease or primary bronchial-associated lymphoid tissue lymphoma. Radiographically, they appear as lung nodules/masses or airspace consolidations [Figure 9].[8] Immunohistochemically, these neoplastic cells typically express IgM and stain strongly for CD20 [Figure 10].
- A 56-year-old female with abdominal pain diagnosed with gastric extranodal marginal B-cell lymphoma. (a) Four years after diagnosis, computed tomography images on lung windows demonstrate a spiculated lesion at the right lung apex (arrow) with subsequent growth over a year (b and c) (arrows).
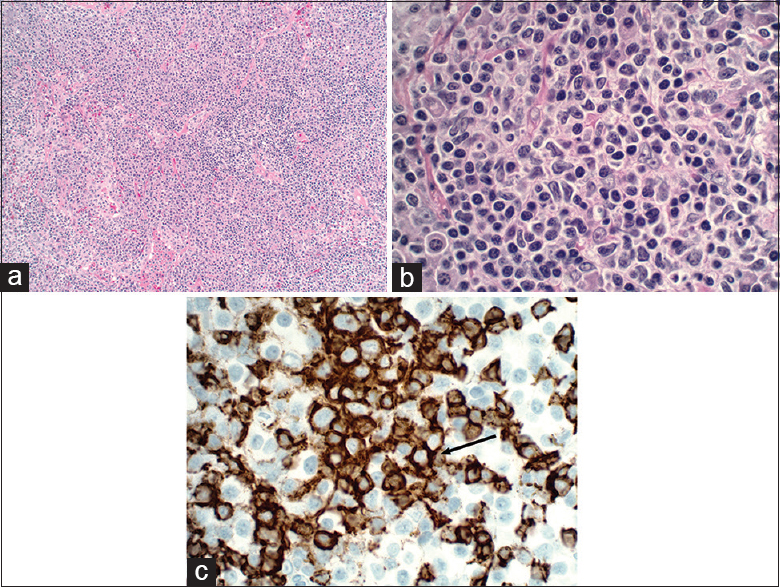
- Pathology from patient in Figure 9. (a) Architectural effacement by the proliferation of atypical lymphoid cells, which show centrocyte, monocytoid, and plasmacytoid features on × 400 (H and E, ×100) (b). (c) Strong membranous immunoreactivity for CD20 confirms B-cell lineage (brown, arrow).
POSTTRANSPLANT LYMPHOPROLIFERATIVE DISORDER
PTLDs are lymphoid or plasmacytic proliferations that develop as a consequence of immunosuppression following organ transplantation. The abdomen is most frequently involved. Thoracic manifestations are less common, presenting as randomly distributed pulmonary nodules [Figure 11].[9] A characteristic feature of PTLD is its association with Epstein–Barr virus infection, which can be detected by Epstein–Barr encoding region in situ hybridization [Figure 12].
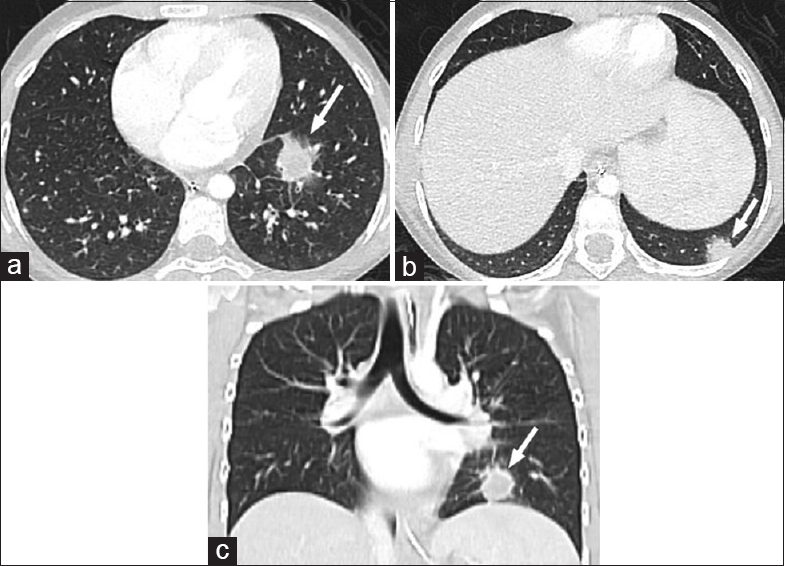
- A 10-year-old female status postliver transplant for Wilson disease diagnosed with the posttransplant lymphoproliferative disease. (a-c) Axial and coronal computed tomography sections on lung windows show two pulmonary nodules in the left lower lobe ten months after transplant (arrows).
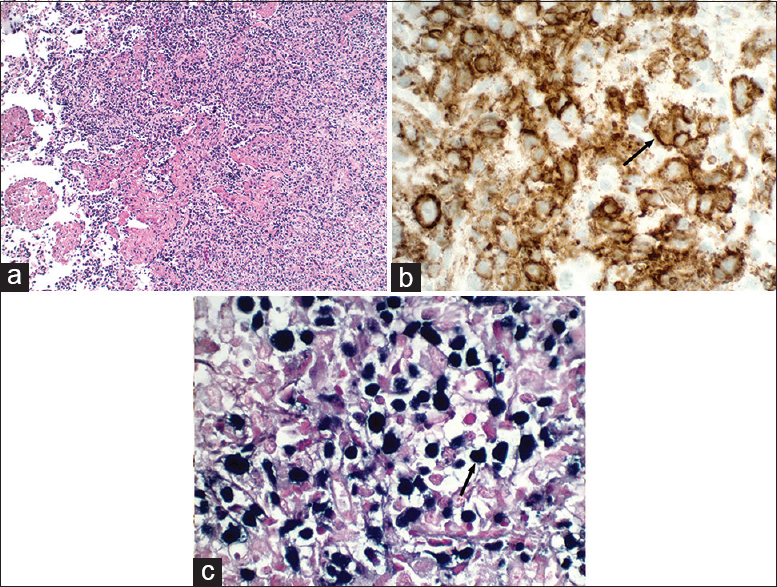
- Pathology from patient in Figure 11. (a) Effacement of normal architecture by the proliferation of large atypical lymphoid cells (H and E, ×100). (c) Strong reactivity for CD20 confirms B-cell lineage (brown, arrow). (d) Positive Epstein–Barr encoding region in situ hybridization detects Epstein–Barr virus in neoplastic B-cells (dark blue, arrow).
DISCUSSION/CONCLUSION
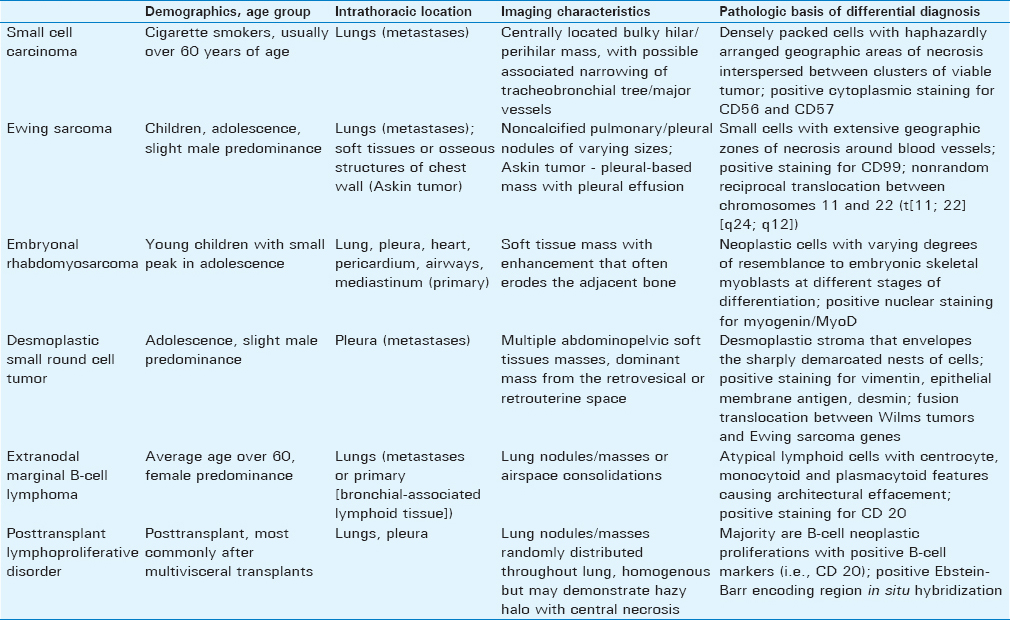
SRBCTs are a heterogeneous group of aggressive neoplasms [Table 1]. Within the thorax, many of these lesions appear as nonspecific pulmonary nodules/masses radiographically and can be difficult to diagnose definitively without soft tissue sampling. On H and E staining, SRBCTs often appear similar, and immunohistochemistry and cytogenetics help distinguish between the entities. Understanding these lesions can broaden the radiologist's differential diagnosis and help guide patient care.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/25/184539
REFERENCES
- Small cell lung carcinoma: Staging, imaging, and treatment considerations. Radiographics. 2014;34:1707-21.
- [Google Scholar]
- Ewing sarcoma: Historical perspectives, current state-of-the-art, and opportunities for targeted therapy in the future. Curr Opin Oncol. 2008;20:412-8.
- [Google Scholar]
- From the radiologic pathology archives: Ewing sarcoma family of tumors: Radiologic-pathologic correlation. Radiographics. 2013;33:803-31.
- [Google Scholar]
- Rhabdomyosarcoma in pediatric patients: The good, the bad, and the unusual. AJR Am J Roentgenol. 2001;176:1563-9.
- [Google Scholar]
- Desmoplastic small round cell tumor in the abdomen and pelvis: Report of CT findings in 11 affected children and young adults. AJR Am J Roentgenol. 2005;184:1910-4.
- [Google Scholar]
- Desmoplastic small round cell tumor: Imaging findings associated with clinicopathologic features. J Comput Assist Tomogr. 2002;26:579-83.
- [Google Scholar]
- Mucosa-associated lymphoid tissue (MALT) lymphoma arising in the esophagus, stomach, and lung. Gen Thorac Cardiovasc Surg. 2011;59:826-30.
- [Google Scholar]
- Bronchial-associated lymphoid tissue lymphoma: A clinical study of a rare disease. Eur J Cancer. 2004;40:1320-6.
- [Google Scholar]
- Imaging of posttransplantation lymphoproliferative disorder after solid organ transplantation. Radiographics. 2009;29:981-1000.
- [Google Scholar]






