Translate this page into:
Fungal Infections of the Central Nervous System: A Pictorial Review
Address for correspondence: Dr. Carola Birgit Mullins, Department of Radiology, Texas Tech University Health Science Center, 4800 Alberta Avenue, El Paso, TX 79905, USA. E-mail: carola.mullins@yahoo.com
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Fungal infections of the central nervous system (CNS) pose a threat to especially immunocompromised patients and their development is primarily determined by the immune status of the host. With an increasing number of organ transplants, chemotherapy, and human immunodeficiency virus infections, the number of immunocompromised patients as susceptible hosts is growing and fungal infections of the CNS are more frequently encountered. They may result in meningitis, cerebritis, abscess formation, cryptococcoma, and meningeal vasculitis with rapid disease progression and often overlapping symptoms. Although radiological characteristics are often nonspecific, unique imaging patterns can be identified through computer tomography as a first imaging modality and further refined by magnetic resonance imaging. A rapid diagnosis and the institution of the appropriate therapy are crucial in helping prevent an often fatal outcome.
Keywords
Brain abscess
cryptococcoma
fungal cerebritis
fungal meningitis
vascular cerebral fungal infections
human immune deficiency virus

- Jose Gavito-Higuera
INTRODUCTION
Fungal infections of the central nervous system (CNS) are rare clinical entities and their development largely depends on the interplay between the host's immune system and fungal virulence factors. Due to a growing number of organ transplants, chemotherapies, and human immunodeficiency virus infections, the incidence of susceptible hosts is increasing. Infections can develop via hematogenous dissemination from a distant focus such as lung, through direct implantation after trauma or secondary to the local extension from sinonasal, orbital, or spinal infections.[1] An aggressive diagnostic approach and timely initiation of antifungal therapy is imperative to reduce morbidity and mortality.
Based on their morphology, fungi can be categorized as yeast (small, unicellular organisms) or hyphae (growth in branchlike colonies). The most common yeast pathogens are Cryptococcus and Candida, and as their small size allows them access to the microcirculation; they can cause diffuse leptomeningitis or manifest as parenchymal granulomas or abscesses. Aspergillus and fungi of the order Mucorales are the most common hyphae forms. Their larger morphology precludes access to the meningeal microcirculation causing a more focal disease leading to cerebritis, abscess formation, or involvement of larger vessels resulting in vasculitis, vascular occlusion, cerebral infarctions, or formation of mycotic aneurysms.[2]
NEUROIMAGING EVALUATION
The immune status and inflammatory response of the host determines the determine of CNS involvement. Although radiological characteristics are often nonspecific, characteristic imaging patterns can help refine the diagnosis.
Because of its broad availability, computed tomography (CT) is the first imaging modality in the evaluation of CNS infections allowing rapid assessment of possible complications such as hydrocephalus, mass effect, and hemorrhage.[3]
Magnetic resonance imaging (MRI) is the most sensitive imaging modality for further characterization of leptomeningitis, empyema, ventriculitis, or cerebral infarction. The sequences employed in the evaluation of infectious processes include diffusion-weighted imaging (DWI), fluid-attenuated inversion recovery (FLAIR), T2-weighted imaging (T2WI), T1 postcontrast (T1 + C), and spectroscopy.[3]
FUNGAL MENINGITIS
Acute or subacute fungal meningitis is often caused by yeast organisms, and cerebrospinal fluid (CSF) analysis serves as the diagnostic gold standard. Since only 50% of all patients diagnosed with meningitis show corresponding radiographic findings, neuroimaging can help confirm suspected meningitis, rule out meningitis mimics, or evaluate for increased intracranial pressure before lumbar puncture.[1] Typical enhancement of the meninges is thin, symmetric, linear, and discontinuous.[1] Leptomeningeal enhancement due to a fungal infection may be smooth or thick, nodular and irregular, long and continuous, poorly demarcated or asymmetric, and may extend into the base of the sulci. All patterns should be confirmed on more than three contiguous MRI.[1] Viral and bacterial meningitis tend to produce a smooth, linear enhancement, which has also been described in fungal meningitis in immunocompetent patients [Figures 1 and 2].
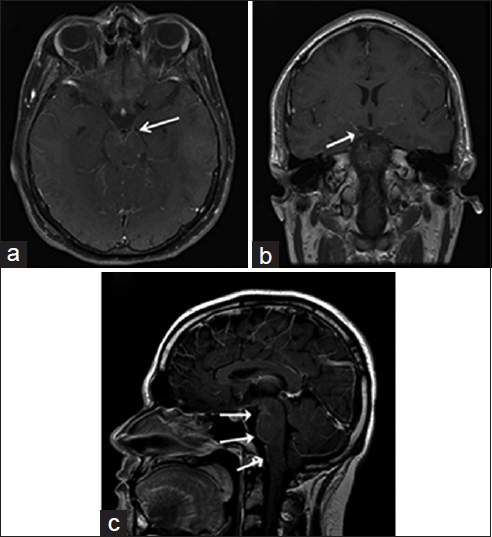
- A 22-year-old immunocompetent man, who presented with headaches, is diagnosed with coccidioidal meningitis. Magnetic resonance imaging T1-weighted postcontrast imaging of the brain in axial (a), coronal (b) and sagittal (c) view shows marked supra- and infra-tentorial smooth linear leptomeningeal enhancement (white arrows) is consistent with meningitis.
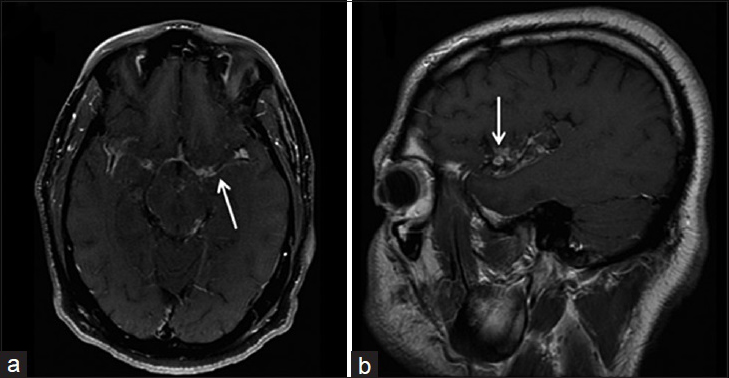
- A 40-year-old immunocompetent man, who presented with headaches and had a history of disseminated coccidioidomycosis, is diagnosed with granulomatous leptomeningitis. Magnetic resonance imaging T1-weighted postcontrast imaging of the brain in axial (a) and sagittal (b) view shows nodular, irregular and intense leptomeningeal enhancement (arrows) involving the sylvian fissure. These findings are consistent with granulomatous leptomeningitis.
The inflammatory response in fungal disease results in deposits of thick gelatinous exudates containing inflammatory cells, fibrin, and hemorrhage leading to arachnoiditis.[1] The increase of proteins in the subarachnoid space results in a shortened T1 relaxation time and hyperintensity on FLAIR sequences.[3] Meningeal adhesion can result in arachnoid granulation occlusion and secondary hydrocephalus [Figures 3 and 4]. As fungal meningitis invites many differentials such as tuberculosis, sarcoid, or meningeal carcinomatosis, we emphasize that the imaging appearance of smooth and nodular leptomeningeal enhancement is often encountered in fungal infections but is nonspecific and needs to be correlated with CSF analysis and lab findings.
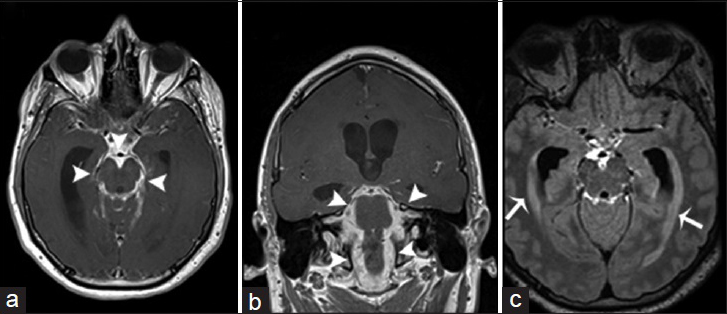
- A 25-year-old man, who presented with a history of acute daily headaches over the past 6 months and had a history of chronic coccidioidal meningitis (treated), is diagnosed with chronic hypertrophic basal arachnoiditis and secondary hydrocephalus. Magnetic resonance imaging T1-weighted postcontrast imaging of the brain in axial (a) and coronal (b) view demonstrates marked leptomeningeal hypertrophy with diffuse enhancement involving predominantly the basal cisterns (arrowheads). (c) Magnetic resonance imaging fluid-attenuated inversion recovery postcontrast imaging of the brain in axial view shows a significant enlargement of the lateral ventricles with transependymal migration of cerebrospinal fluid (arrows). These findings are consistent with a hydrocephalus secondary to chronic hypertrophic basal arachnoiditis.
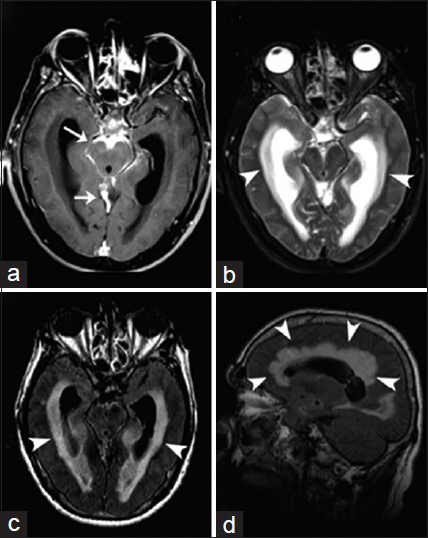
- A 60-year-old woman, who presented with headaches, is diagnosed with coccidioidal basal arachnoiditis and acute communicating hydrocephalus. (a) Magnetic resonance imaging T1-weighted postcontrast imaging of the brain in axial view shows abnormal leptomeningeal thickening and enhancement along the perimesencephalic and superior cerebellar cisterns (arrows). (b) Magnetic resonance imaging T2-weighted imaging of the brain in axial view and magnetic resonance imaging fluid-attenuated inversion recovery (c and d) of the brain in axial and sagittal view demonstrate supratentorial ventriculomegaly associated with effacement of the cortical sulci and transependymal edema (arrowheads). These findings are consistent with basal arachnoiditis and acute communicating hydrocephalus.
FUNGAL CEREBRITIS
Cerebritis is the earliest manifestation of brain infections and the precursor of abscess development.[4] Although almost any fungus may cause encephalitis, Cryptococcus is most frequently identified, followed by Aspergillus and Candida.[5] The brain parenchyma is reached by fungi penetrating the Virchow-Robin spaces adjacent to the meninges.[2]
On CT imaging, ill-defined intraparenchymal hypodense lesions resulting from edema and scattered areas of increased attenuation due to either hemorrhage or increased concentration of metal ions are indicative of fungal cerebritis.[6] Metal ions are necessary for fungal growth and represent metabolic waste products resulting in increased attenuation on CT and paramagnetic effects on MRI. On T1WI, fungal cerebritis appears as an iso- or hypo-intense area with subtle mass effect, shift and minimal to no enhancement.[6] T2WI and FLAIR sequences show a hyperintense lesion, which may present areas of decreased signal intensity within the lesion due to the paramagnetic effects of metal ions. These lesions typically present with restricted diffusion on DWI.[46]
Britt and Enzmann defined early and late cerebritis, followed by early and late capsulitis as the four stages of abscess formation. Late cerebritis (4–5 days to 2 weeks) can be differentiated from early cerebritis (3–5 days) through the presence of a thick nodular enhancement on postcontrast MRI [Figure 5].[7] Early stages of capsulitis (2 weeks) are characteristic for minimal collagen content, progressing to a mature collagen capsule with perilesional gliosis in late capsulitis. On CT imaging, both stages demonstrate a low attenuation core with a contrast enhancing capsule.[7]
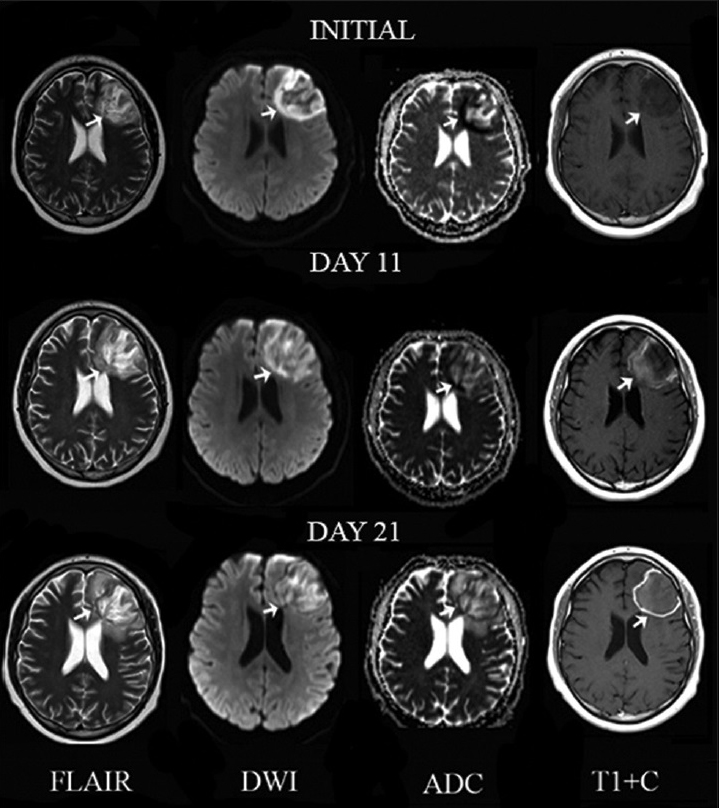
- A 17-year-old man with headaches and lymphoma diagnosed with Aspergillus fumigatus infection of the central nervous system. Serial magnetic resonance imaging of a left frontal lobe lesion (arrows) on T2, diffusion-weighted imaging, apparent diffusion coefficient map and T1 postcontrast (T1 + C) from left to right in axial view. Initially, the lesion appears ill-defined with increased signal intensity with foci of signal void on T2, restricted diffusion on diffusion-weighted imaging and apparent diffusion coefficient map and no abnormal enhancement on T1 post contrast suggesting late cerebritis. An 11-day follow-up shows decrease of signal voids on T2, restricted diffusion on diffusion-weighted imaging and faint peripheral enhancement on T1 post contrast consistent with early capsulitis. A 21-day follow-up demonstrates development of well-defined borders and linear peripheral enhancement on T1 post contrast consistent with late capsulitis.
BRAIN ABSCESS
Clinical symptoms include specific focal neurologic deficits and an increased intracranial pressure. It has an estimated mortality of 85–100% and characteristic imaging features may help differentiate a fungal from a bacterial cause. While fungal abscesses are more likely to be multiple and can involve the basal ganglia, bacterial abscesses are often solitary lesions sparing the basal ganglia.[2] On DWI, fungal abscesses show restricted diffusion in their intracavitary projections[2] and the abscess wall while sparing the core of the lesion [Figure 6].[8]
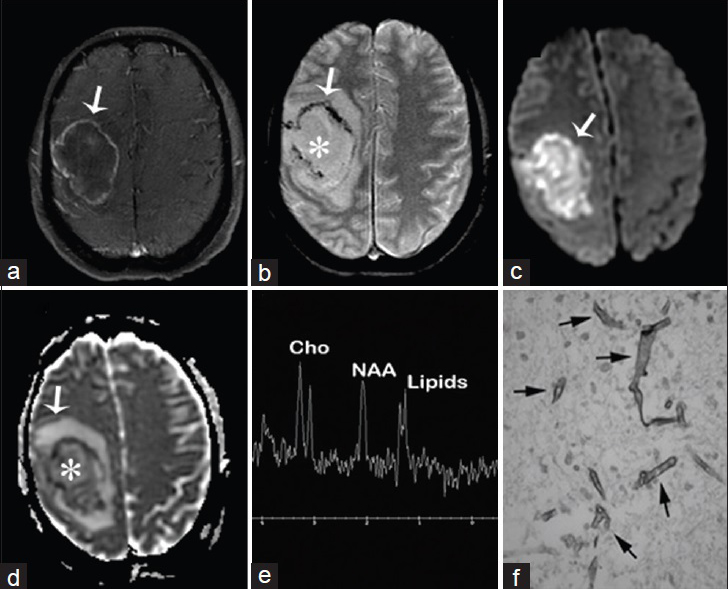
- A 50-year-old woman with acute myeloid leukemia, fever, headaches, and neurologic deficits diagnosed with brain abscess due to Rhizomucor pusillus (a) Magnetic resonance imaging T1 fast spin echo/fat sat postcontrast of the brain, axial view shows right sided mass with enhancing wall (arrow) (b) magnetic resonance imaging T2* GRE, axial view shows nonenhancing liquefied core (asterisk) and hypointense wall with susceptibility artifact (arrow) (c and d) diffusion-weighted imaging and apparent diffusion coefficient map of the brain, axial view show restricted diffusion of the wall and intracavitary projections (arrows) and no restriction inside the lesion (asterisk d) (e) MR spectroscopy of the brain shows inverted NAA/Cho ratio due to NAA depletion and increased lipids due to cell destruction (f) Gomori methenamine silver nitrate stain shows Zygomycetes (arrows).
On MR T1WI, fungal abscesses demonstrate a hypointense core with a surrounding iso- to mildly hyper-intense rim. T2WI shows increased signal intensity of the core of the lesion with a surrounding rim of hypointensity. Peripheral enhancement is seen on T1 weighted post contrast enhancement sequence. A dual-rim sign on DWI is a distinctive feature of a pyogenic abscess.[9]
Fungal abscesses may contain lipids (1.2–1.3 ppm), lactate (1.3 ppm), alanine (1.5 ppm), acetate (1.9 ppm), succinate (2.4 ppm), and choline (3.2 ppm) which can be visualized on MR spectroscopy.[6] A typical feature of fungal infections is the disaccharide trehalose (3.6 ppm) as a distinctive component of the fungal wall.[2]
CRYPTOCOCCOMA
CNS involvement in a Cryptococcus infection usually leads to inflammation of the meninges, but abscess and granuloma formation may also occur [Figure 7]. Metabolites released by Cryptococcus can inhibit the migration and function of leukocytes and promote survival as well as localized replication of the pathogen, facilitating chronic granulomatous inflammation and cryptococcoma formation. Typical imaging findings include multiple T2 hyperintense masses with peripheral enhancement on T1 weighted post contrast enhanced images predominately found in the basal ganglia of immunocompromised individuals [Figure 8], which may be mistaken for metastatic disease.[2]
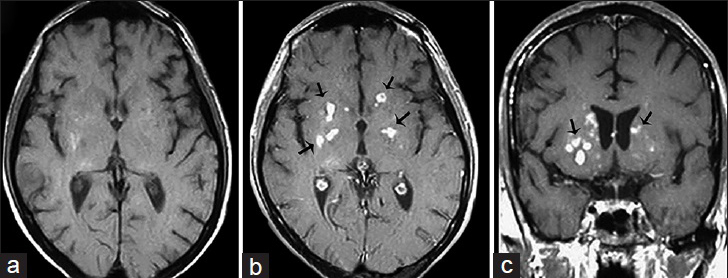
- A 38-year-old immunocompromised male presenting with headache and altered mental status diagnosed with Cryptococcosis (a) Magnetic resonance imaging T1-weighted imaging of the head in axial view, T1 postcontrast-weighted imaging of the head in axial (b) and coronal view (c) reveal multiple small enhancing nodules in the basal ganglia bilaterally within perivascular spaces consistent with cryptococcomas (arrows).
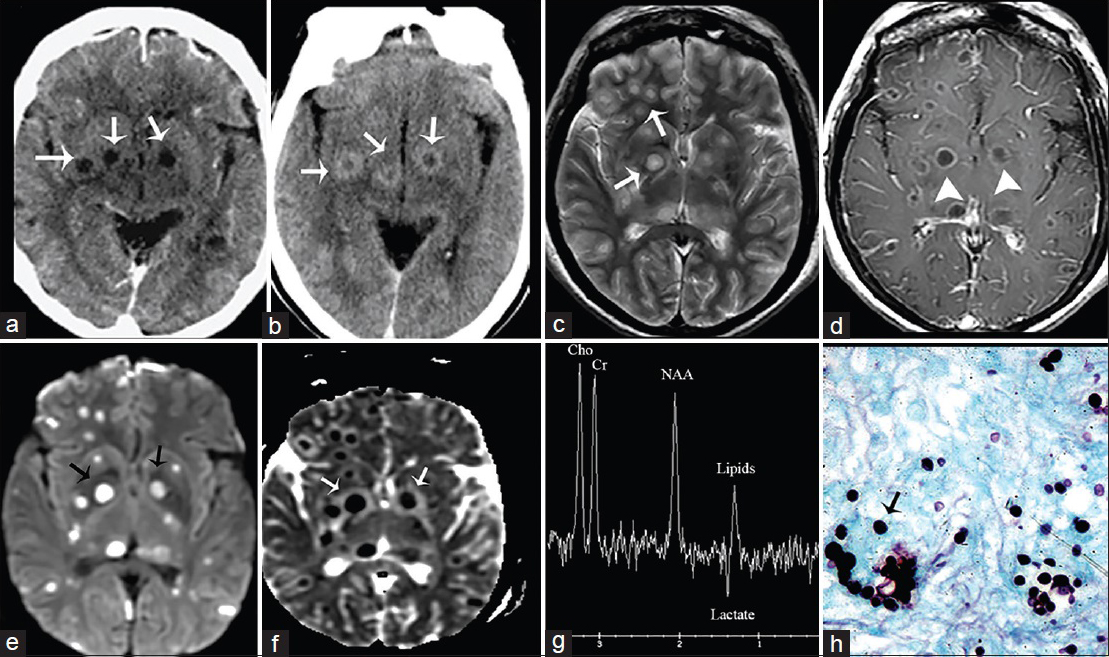
- A 52-year-old woman, human immunodeficiency virus-positive with neurological deficits diagnosed with central nervous system cryptococcosis. (a) Noncontrast and (b) contrast-enhanced computed tomography scan of the brain, axial view show rim enhancing lesions in the basal ganglia (arrows). (c) Magnetic resonance imaging T2, axial view demonstrates increased signal intensity in the lesions. (Gelatinous pseudocysts) (arrows). (d) Magnetic resonance imaging T1 postcontrast shows rim enhancement (arrowheads), (e) B1000 data set and corresponding ADC map (f) show restricted diffusion (arrows). (g) MR spectroscopy shows inverted NAA/Cho ratio with lipids and lactate consistent with infection (h) GSM stain shows fungal organism (arrow).
VASCULAR CEREBRAL FUNGAL INFECTIONS
Fungal infections can result in meningeal vasculitis with vessel thrombosis and localized brain infarctions and are often due to infections with Aspergillus species and Zygomycetes. The induced inflammatory response in the vessel wall results in in situ thrombosis as a possible source of emboli [Figure 9]. Disruption of the elastic laminae of the vessel wall may lead to its focal dilatation and formation of mycotic aneurysms.[2] Mycotic aneurysms are usually located within the anterior circulation, infecting long segments of proximal portions of the large cerebral vessels [Figure 10].[2]
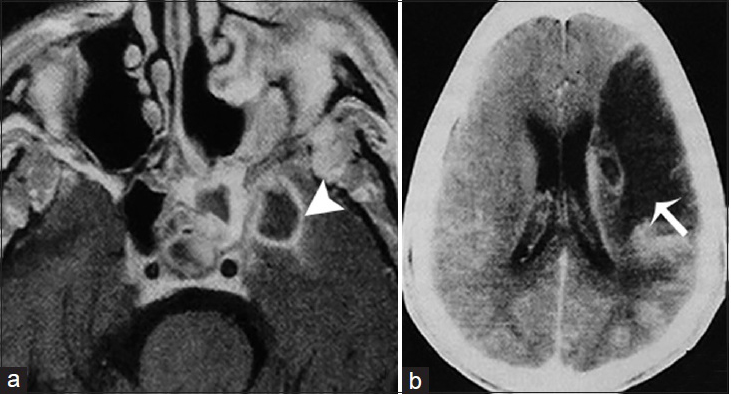
- A 51-year-old man, who presented with right-sided paresis and had a history of uncontrolled diabetes mellitus, is diagnosed with chronic invasive fungal Mucormycosis. (a) Magnetic resonance imaging T1 postcontrast imaging of the brain in axial view demonstrates an infectious process in the left sphenoidal sinus and an intracranial intraparenchymal ring enhancing extension in the medial temporal lobe indicative of an abscess (arrowhead). (b) Contrast-enhanced computer tomography image of the same patient shows and acute infarct in the vascular territory of the left middle cerebral artery secondary to intracranial vasculitis (arrow).
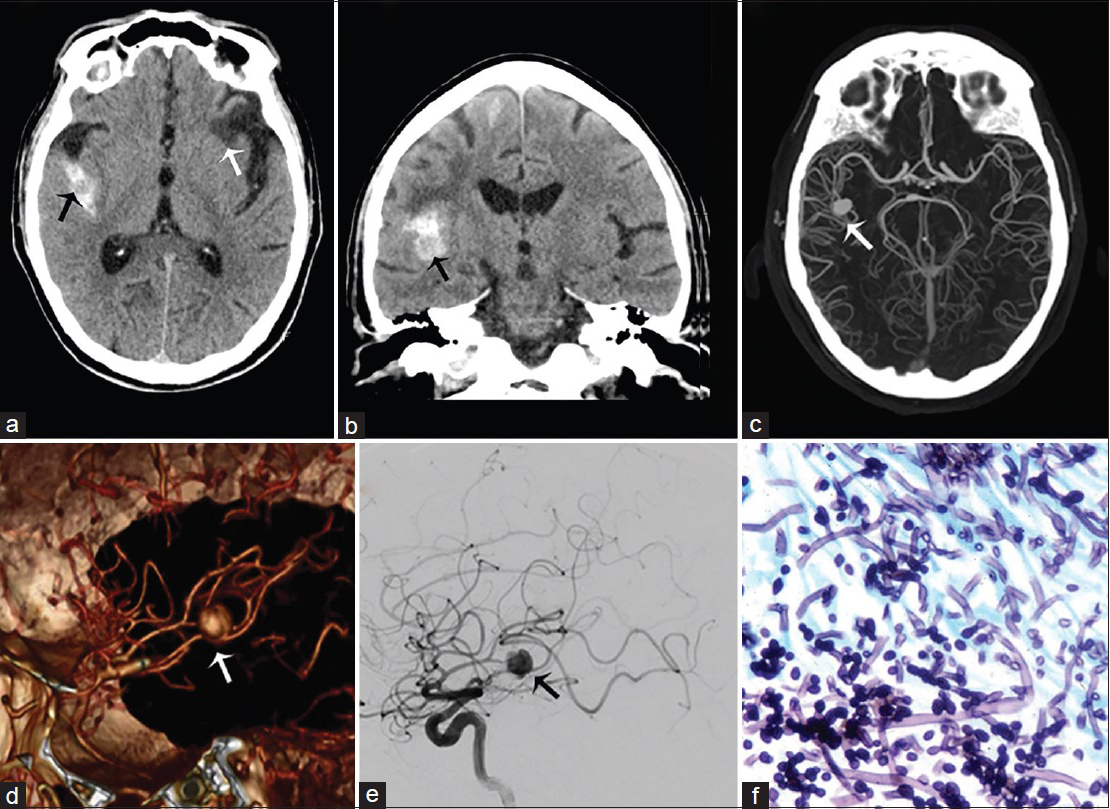
- A 55-year-old man with history of intravenous drug abuse, headache, and sepsis due to a Candida sp. infection, diagnosed with mycotic aneurysm of the M2–M3 right middle cerebral artery. Computed tomography imaging of the brain without contrast in axial (a) and coronal (b) view demonstrate subarachnoid hemorrhage in the right lateral fissure surrounding right middle cerebral artery aneurysm (black arrow) and areas of encephalomalacia of prior ischemic events (white arrow). (c) Maximum intensity projection computed tomography angiogram, (d) three-dimensional reconstruction and (e) lateral selective angiogram show a saccular aneurysm arising from the M2–M3 right middle cerebral artery (arrows). (f) H and E stain with hyphae of Candida albicans.
CONCLUSION
Fungal CNS infections present diagnostic challenges. Although neuroradiological manifestations of fungal CNS infections are often nonspecific, recognition of typical imaging patterns on CT and MRI are imperative to help refine the differential diagnosis and initiate early treatment.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/24/184244
REFERENCES
- Fungal infections of the central nervous system. Neuroimaging Clin N Am. 2012;22:609-32.
- [Google Scholar]
- Imaging features of central nervous system fungal infections. Neurol India. 2007;55:241-50.
- [Google Scholar]
- Diffusion imaging in brain infections. Neuroimaging Clin N Am. 2011;21:89-113. viii
- [Google Scholar]
- Imaging of cerebritis, encephalitis, and brain abscess. Neuroimaging Clin N Am. 2012;22:585-607.
- [Google Scholar]
- Comparative evaluation of fungal, tubercular, and pyogenic brain abscesses with conventional and diffusion MR imaging and proton MR spectroscopy. AJNR Am J Neuroradiol. 2007;28:1332-8.
- [Google Scholar]
- Differentiation of pyogenic and fungal brain abscesses with susceptibility-weighted MR sequences. Neuroradiology. 2014;56:937-45.
- [Google Scholar]






