Translate this page into:
Semantic Dementia Diagnosed by F-18 FDG PET/MRI: Co-registered Images
Address for correspondence: Dr. Prashant Jolepalem, William Beaumont Hospital 3601 W. 13 Mile Road Royal Oak-48073, Michigan. E-mail: pjolepalem@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
We report a case of a 61-year-old male who presented with a sudden change in mental status. From a psychiatric standpoint, his symptoms were consistent with a bipolar disorder. A neurology consult raised suspicion for vascular dementia, given the sudden onset of symptoms; however, the magnetic resonance angiography (MRA) was unremarkable. The magnetic resonance imaging (MRI) had findings that were suggestive of both vascular and frontotemporal lobe dementia based on parenchymal atrophy and a lacunar infarct near the thalamus. However, by co-registering the magnetic resonance images with a subsequent fluorine-18 Fluorodeoxyglucose positron emission tomography (F-18 FDG PET), and combining the functional data with the anatomic appearance, the diagnosis was narrowed to semantic dementia, which is one of the lesser known subtypes of frontotemporal lobe dementia (FTD).
Keywords
Fluorodeoxyglucose brain positron emission tomography frontotemporal dementia
positron emission tomography/magnetic resonance imaging
semantic dementia

INTRODUCTION
Diagnosing a patient with a specific dementia on clinical examination can be very challenging. Functional metabolic imaging of the brain using fluorine-18 fluorodeoxyglucose (FDG) positron emission tomography (PET) is one of the most useful tools in distinguishing the patient's symptoms from the various types of dementia by elucidating global and regional deficits in cerebral glucose metabolism.[1] Magnetic resonance imaging (MRI) often plays a role as well, as it is more widely available, and is used to evaluate structural abnormalities within the brain parenchyma.[2]
We present a case where hybrid imaging of PET/MRI was used to diagnose a patient with semantic dementia (SD), which is one of the lesser known subtypes of frontotemporal lobe dementia (FTD). SD is characterized by a loss of language and verbal skills with late stage behavioral changes. It is associated with predominantly temporal lobe atrophy (left greater than right) and is often referred to as temporal variant FTD.[3]
CASE REPORT
A 61-year-old, right-handed male presented with a sudden change in mental status preceded by a significant decline in functionality over the past 2 years. He was previously a highly functional person and worked as an executive for a Fortune 500 company. His change in mental status was characterized by several behavioral changes; including cruelty toward family members and pets, promiscuity, adultery, drug abuse, wild spending habits, and lack of empathy. His family had also noticed a change in his speech patterns in terms of phonation and pronunciation of words.
His initial diagnosis was bipolar disorder; however, he did not respond to mood stabilizing drugs. After losing his job, he was admitted to the psychiatric ward with suicidal tendencies. MRI and magnetic resonance angiography (MRA) were ordered to evaluate for dementia. The MRI [Figure 1] demonstrated diffuse parenchymal atrophy, which was slightly more extensive in the temporal lobes. There was a lacunar infarct in the right thalamus that was suggestive of vascular dementia. However, the MRA portion was unremarkable and did not support this diagnosis.
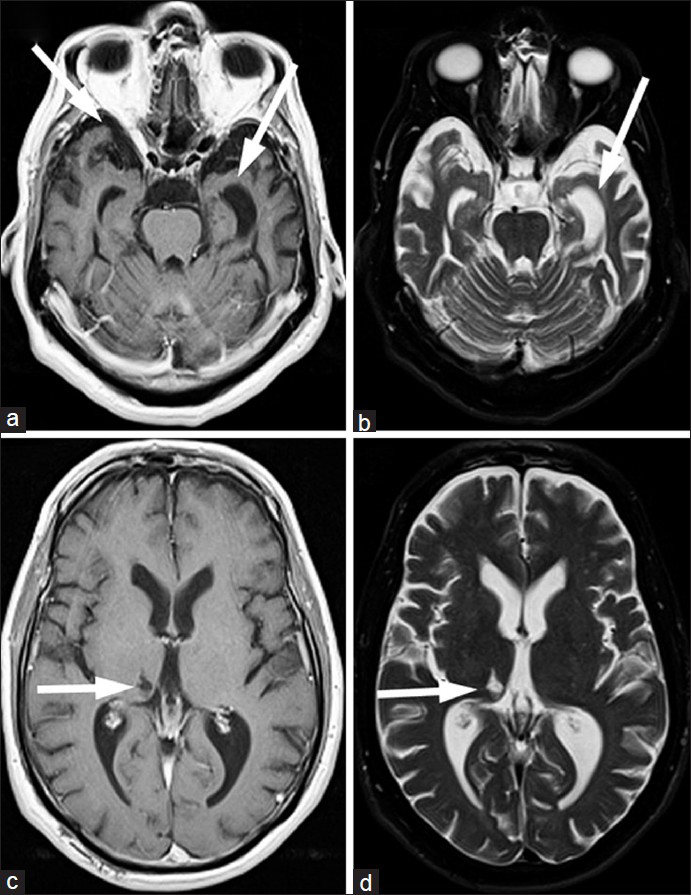
- 61-year-old male with change in mental status and decline in function diagnosed with semantic dementia. (a and b) The magnetic resonance imaging (MRI) transaxial views of the T1-weighted and (c and d) T2-weighted images with proton density and fluid suppression sequences demonstrate diffuse parenchymal atrophy, slightly more substantial in the temporal lobes (arrows on a and c). These non-specific findings do raise the possibility of frontotemporal dementia (FTD). A thalamic infarct (arrows on b and d), along with subtle white matter abnormalities are also seen, which are suggestive of vascular dementia, though inconsistent with the negative magnetic resonance angiography findings.
A FDG PET scan was performed the same day for functional metabolic evaluation. The images were co-registered with the MRI using the Institute of Electrical and Electronics Engineers (IEEE) mutual information algorithm.[4] The resultant PET/MR images [Figure 2] demonstrated a pattern of decreased FDG uptake that was predominantly confined to the temporal lobes and was particularly severe in the amygdalae and hippocampi. The decrease in activity was greater on the left side and extended superoposteriorly toward the parietal lobe and Wernicke's area. The other atrophic areas and white matter changes seen on the MRI had intact glucose metabolism. The lacunar infarct also had decreased FDG uptake, but that was considered to be an incidental finding. The decreased glucose metabolism affecting only the temporal lobes (worse on the left side) with the concordant areas of atrophy on the MRI were highly specific for semantic dementia and allowed for a definitive diagnosis to be made.[356] Unfortunately, the only management available for this condition is supportive care.
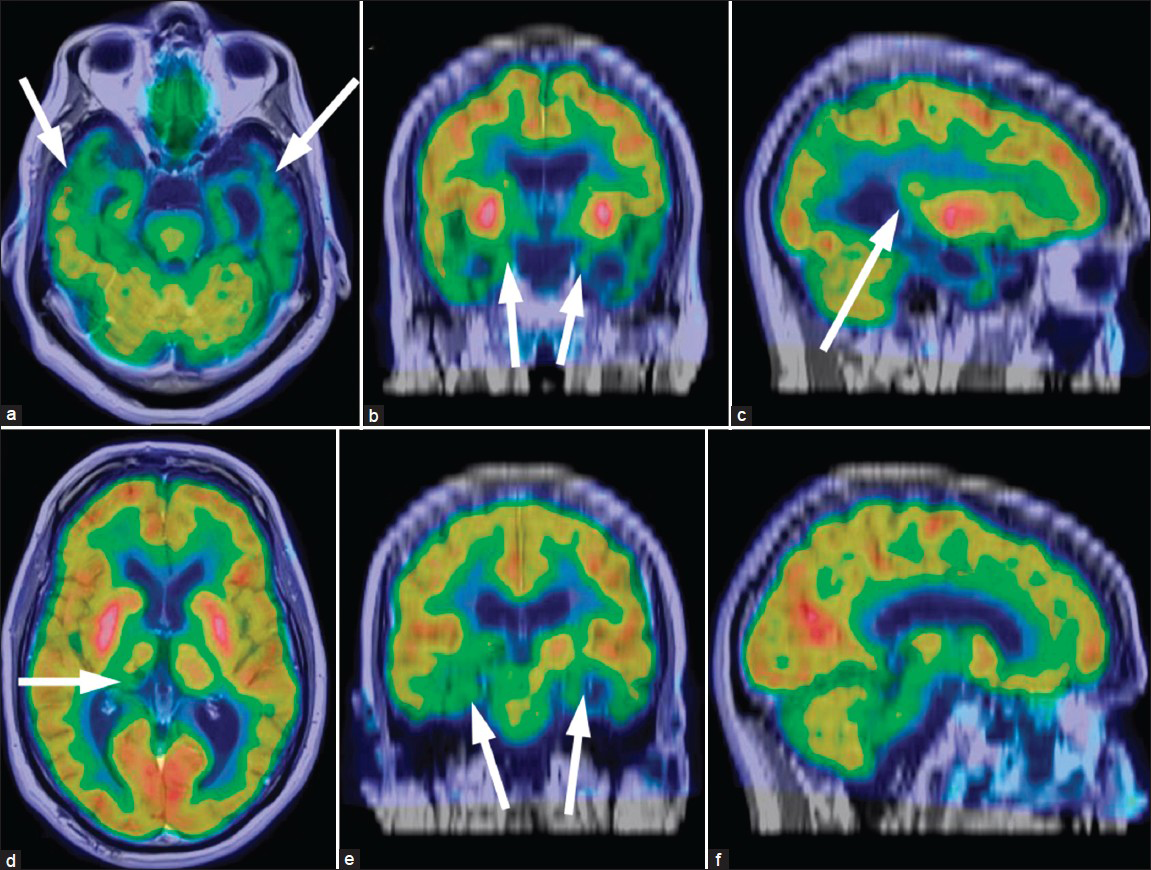
- 61-year-old male with change in mental status and decline in function diagn¬osed with semantic dementia. The fluorine-18 fluorodeoxyglucose (F-18 FDG) positron emission tomography (PET) of the brain was obtained and coregistered with the T1-weighted sequences of the MRI. Two separate triangulated cross-sections of the reconstructed images in (a and d) axial views, (b and e) coronal views, and (c and f) sagittal dimensions show significantly reduced uptake corresponding to atrophy in the temporal lobes (arrows on a), involving the amygdalae (arrows on b), hippocampi (arrows on e), and Wernicke's area (arrow on c). The function is well preserved in the remaining areas of global atrophic change and white matter changes. There is also reduced activity in the location of the suspected right thalamic infract (arrow d). These findings are pathognomonic for semantic dementia.
DISCUSSION
Although each type of dementia has its own constellation of symptoms, there is significant overlap and most patients present with features of several different psychiatric and neurologic disorders.[78] The majority of patients present with some type of memory loss with a variety of non-specific behavioral changes. Making a clinical diagnosis based on symptoms can be extremely challenging. Anatomic imaging is often not useful, other than to exclude physical causes such as stroke or hydrocephalus. There may be subtle clues to a diagnosis, but nothing definitive.[36]
Functional imaging provided by FDG PET evaluation of brain glucose metabolism is a valuable modality for dementia. Alzheimer's dementia (AD) is characterized by decreased glucose metabolism in the temporal and parietal lobes with sparing of the frontal lobes. FTD, as the name suggests, demonstrates decreased activity in the frontal and temporal lobes with sparing of the parietal lobes. Dementia with Lewy bodies (DLB) has pattern of decreased activity similar to AD, but also demonstrates decreased activity in the occipital lobe.[1] However, many early stages of dementia may have atypical or variable appearances on FDG PET.[9] Additionally, some variants or subtypes may not be recognized by the interpreting physician. For example, semantic dementia has involvement confined to the temporal lobe, but the deficits are often not as striking as in the other types of dementia on FDG PET.[9]
On MRI, the aforementioned types of dementia may show global atrophy, with more extensive changes in the involved lobes (such as more frontal lobe atrophy in FTD). However, these structural changes often occur late in the disease progression and can be non-specific. Additionally, there is no typical structural abnormality in DLB. Often at times, the FDG PET will show metabolic deficits before atrophic changes become apparent on MRI.
In this case, we were able to combine metabolic deficits with the anatomic information provided by MRI. This allowed us to corroborate the decreased activity confined to the temporal lobe with specific structures or areas within the brain that were involved. The amygdale, hippocampi, and Wernicke's area all showed focal decreases in activity compared with the overall reduction of radiotracer activity in the temporal lobes. Correlation with the co-registered MRI made identification of these structures possible. This level of detailed findings would not be possible on the PET images alone. The FDG PET, on the other hand, showed that the decreased activity was greater on the left side, which is classic for semantic dementia, whereas the atrophy on the MRI seemed relatively symmetric.
These combined imaging findings were pathognomonic for semantic dementia.[15] Since the affected areas of the brain are involved in empathy (amygdala), meaningful word usage (Wernicke's area), memory (hippocampus), and conceptual knowledge (amygdala and hippocampus interaction); the patient's symptoms could be easily explained.[810]
CONCLUSION
This case demonstrates an example of the value of combined PET-MRI in clinical practice. Co-registration of images provides essential information that is needed to make a diagnosis in challenging cases. Thus, clinical symptoms can be related to lesions in specific parts of the brain.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2013/3/1/35/117459
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Cerebral metabolic patterns at early stages of frontotemporal dementia and semantic dementia. A PET study. Neurobiol Aging. 2004;25:1051-6.
- [Google Scholar]
- Hippocampal volumes in Alzheimer's disease, Parkinson's disease with and without dementia, and in vascular dementia: An MRI study. Neurology. 1996;46:678-81.
- [Google Scholar]
- Patterns of temporal lobe atrophy in semantic dementia and Alzheimer's disease. Ann Neurol. 2001;49:433-42.
- [Google Scholar]
- A comparison between the surface-based and mutual-information-based methods for co-registering SPECT and MR images. 29th Annual International Conference of the IEEE Lyon: EMBS; 2007. p. :868-71.
- [Google Scholar]
- Automated MRI-based classification of primary progressive aphasia variants. Neuroimage. 2009;47:1558-67.
- [Google Scholar]
- Patterns of atrophy in pathologically confirmed FTLD with and without motor neuron degeneration. Neurology. 2006;66:102-4.
- [Google Scholar]
- Distinct behavioural profiles in frontotemporal dementia and semantic dementia. J Neurol Neurosurg Psychiatry. 2001;70:323-32.
- [Google Scholar]
- Patterns of brain atrophy in frontotemporal dementia and semantic dementia. Neurology. 2002;58:198-208.
- [Google Scholar]
- Classification of primary progressive aphasia and its variants. Neurology. 2011;76:1006-14.
- [Google Scholar]






