Translate this page into:
Implications of Ambient Glucose Variation on the Target-to-Background Ratio of Hepatic Tumors By 18FDG-PET Imaging
Address for correspondence: Dr. Prashant Jolepalem, Department of Diagnostic Radiology and Molecular Imaging, Nuclear Medicine Section, William Beaumont Hospital, 3601 W 13, Mile Road, Royal Oak, MI 48073, USA. E-mail: pjolepalem@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objectives:
To investigate the effects of ambient glucose on quantitative analysis of hepatic tumors on 2-deoxy-2-(18F)-fluoro-D-glucose (18FDG) positron emission tomography (PET) and to establish a method for glucose correction.
Patients and Methods:
Eighty-six patients with hepatic lesions identified on 18FDG PET/computed tomography (CT) were analyzed. The serum glucose level (Glc) was recorded prior to imaging, and the maximum standardized uptake value (SUV) in the hepatic tumors and the average SUV in normal liver were determined. The inverse relationship of SUV to glucose can be defined as d (SUV)/d (Glc) = g*SUV/(Glc), where g is the glucose sensitivity. Simulations using glucose level from 70 to 250 mg/dl were performed to evaluate the effects of Glc on the maximum SUV of malignant hepatic lesions and normal liver.
Results:
By logarithmic transformation and linear regression, g for metastasis was significantly higher than that for normal liver (−0.636 ± 0.144 vs. −0.0536 ± 0.0583; P = 0.00092). Simulation studies showed that the SUV in malignant lesions will decrease rapidly when Glc level is >120 mg/dl, while background liver remains relatively constant up to 250 mg/dl.
Conclusion:
The tumor FDG uptake is much more sensitive to ambient glucose level variation than the background liver. Therefore, correction by the glucose sensitivity factor will result in more accurate SUV measurements and make semi-quantitative analysis of 18FDG PET scans more reliable.
Keywords
Background liver
18-fluorodeoxyglucose positron emission tomography
glucose sensitivity
hepatic metastases
standardized uptake value
tumor to background ratio
INTRODUCTION

2-deoxy-2-(18F)-fluoro-D-glucose (18FDG) positron emission tomography (PET) is now a well-established imaging modality, with greater emphasis being placed on reporting the standardized uptake value (SUV), as this quantitation is easily understood by referring physicians as opposed to the variable esoteric descriptions of visual interpretation. SUVs can also provide a simple and reproducible method for evaluating disease progression and treatment response.[1] As a result, there have been numerous investigational studies on semi-quantitative SUV analysis, particularly in the area of stratifying benign from malignant activity. These studies have typically relied upon establishing wide divergent separation of the mean SUVmax between two population groups (e.g. disease and disease-free).
We have previously described one such method using the tumor-to-liver background ratio that dramatically improved the accuracy of 18FDG PET/CT in characterizing hepatic lesions over standard visual interpretation or correlative imaging alone.[2] However, one of the unanswered questions remaining is how variations in serum glucose (Glc) at the time of radiotracer injection would impact the accuracy of this method, because it was well established right from the advent of 18FDG PET that ambient glucose levels can adversely affect tumor uptake and decrease the sensitivity.[3456] Given the already normally high background 18FDG liver activity, this potential pitfall is even more apparent when evaluating hepatic tumors.
While elevated Glc at the time of image acquisition is a known problem, there have been only a limited number of studies quantifying its effects, particularly when evaluating hepatic lesions.[7] As more studies are performed investigating semi-quantitative analyses of 18FDG PET to evaluate unknown lesions and track treatment response, it will become more vital to understand the impact of Glc on SUV measurements.[89] The purpose of this study was to investigate how variations in ambient glucose levels would affect the divergent SUV between malignant hepatic tumors and normal liver tissue, and to derive a method for SUVmax correction in the setting of elevated glucose levels.
PATIENTS AND METHODS
Patients
After receiving permission from our Human Investigations Committee, 86 patients with biopsy-proven malignant hepatic lesions identified on 18FDG PET were retrospectively analyzed. There were 38 male patients and 48 female patients. The age range was 42–95 years. Patients with many different types of primary malignancies were included, with the breakdown summarized in Figure 1. There was no limitation placed on the size of the lesion as long as it was described on an imaging study. If the lesion was identified on an imaging study other than PET, then these patients were only included if the PET scan was performed within 2 weeks to avoid any significant interval change. Patients were excluded if they were undergoing chemotherapy, had any attempted surgical resection of the lesion in question or any directed therapy such as trans-arterial chemo-/radio-embolization or radiofrequency ablation. Patients with a body mass index (BMI) of over 35 kg/m2 were excluded to preserve consistent image quality.

- Chart showing the total numbers of lesions by tumor types that were analyzed in the study.
Image acquisition
The protocol used an intravenous injection of 10-20 mCi of 18FDG, followed by an uptake time of approximately 90 min. Serum glucose level (Glc), body weight, dose assay time, injection time, and imaging time were all recorded.
All patients were imaged on a dedicated 16-slice PET/CT scanner (GE Discovery DST; GE Medical Systems, Milwaukee, WI, USA). The whole-body PET images were obtained for 3 min per bed position, usually with six positions to cover from the base of the skull to the middle of the thighs, using a 3-slice overlap. The images were acquired using the three-dimensional high-sensitivity mode with an axial field of view in a 256 × 256 matrix. Then iterative image reconstruction was performed on a 128 × 128 matrix using ordered subset expectation maximization (OSEM) algorithm for 30 subsets and two iterations, with a 7.0-mm post-reconstruction filter.
Concomitant CT data were used for attenuation correction of all PET images using 140 kVp, 120–200 mA, 1.75 pitch, and a 3-mm slice thickness. All patients received oral contrast. SUV was calculated by the following formula:
SUV = ROI activity concentration in tissue (μCi/g)/[dose (μCi)/total body weight in grams].
Image analysis
On each patient's PET scan, a region of interest (ROI) was placed over the most FDG-avid hepatic lesion to measure the SUVmax, which quantifies the activity in the area with the highest uptake. Then, five 2-cm ROIs were placed on background or normal-appearing liver tissue to measure the average SUV of each region, as the average rather than the maximum SUV was felt to be more representative of the background liver activity for control purposes. The mean of those five ROIs was used to represent the overall background liver SUV (SUVbkg) for that study. This procedure was repeated for all 86 patients.
Data analysis
The inverse relationship of SUV to Glc has been previously defined by the differential equation (eq. 1):[10]
d (SUV)/d(Glc) = g*(SUV/Glc) (1)
This equation can be graphically represented following logarithmic transformation. Linear regression of the resultant curve is defined by the following equation:[11]
ln (SUV) = G* ln (Glc) + C (2)
where G is the group mean sensitivity factor for hepatic tumors or background liver tissue and C is a constant factor unique to each tissue type.[12] Using the respective tissue-specific C obtained for hepatic tumors and background liver, the individual glucose sensitivity (g) can be calculated by the equation:[11]
g = {ln (SUV) − C}/ln (Glc) (3)
Finally, Equation (eq. 1) can be solved and rewritten to use g as a dimensionless exponent by the following equation:[12]
SUV1/SUV2= (Glc1/Glc2)g (4)
The above formula was used to run simulation studies to solve for SUV at different Glc levels using average g for a specific tumor type. Therefore, Equation (eq. 4) can be expressed as:
SUVGlcX = SUV*(GlcX/Glc)g (5)
SUV and Glc were the measured values for the study and SUVGlcX yielded the resulting SUV at the simulated glucose level (GlcX). Simulations were performed using GlcX ranging from 70 to 250 mg/dl to observe how differences in Glc at image acquisition would affect SUV measurements. Student's t-tests were used for testing the means, with statistical significance set at P < 0.05.
RESULTS
The mean administered dose of 18FDG was 16.37 ± 1.81 mCi. The mean uptake time between injection and imaging was 91.5 ± 13.8 min. The mean patient BMI was 25.40 ± 3.56 kg/m2. The mean Glc for all the patients at the time of imaging was 106 ± 26 mg/dl (range 70-212 mg/dl). The mean SUV for malignant lesions was 8.45 ± 6.55. The mean SUV for background liver was 1.99 ± 0.58.
Using the linear regression of (ln SUV)/(ln Glc), the C value was 2.21 and 0.68 for hepatic tumors and background liver, respectively. The mean glucose sensitivity (g) for all metastatic lesions was −0.636 ± 0.144 compared with −0.0536 ± 0.0583 for background liver (P = 0.00092). The closer g is to −1, the more sensitive it is to ambient Glc.[11] The rate of change of SUV over Glc (Equation 1) was about two orders higher for metastatic lesions at −0.0561 SUV/(mg/dl) of glucose than for background liver at −0.0003 SUV/(mg/dl) (P < 0.00001).
The differences in the mean SUV of malignant lesions and background liver across various simulated Glc are plotted in Figure 2. The simulation studies using Equation (5) showed that the SUV for metastatic lesions was more dramatically affected than for background liver by Glc levels beyond the normal range, showing a sharp decline as Glc reached 120 mg/dl. SUVs measured in patients with elevated Glc would have been significantly higher if they had been within the target normal Glc range at the time of injection. The linear regression analysis after logarithmic transformation of Equation (1) is graphically represented in Figure 3, with the slopes of the regression lines equaling the rate of SUV change over Glc level derived from Equation (1) for each tissue type.

- Linear plot of the mean SUV of each group against the various simulated glucose levels. The malignant lesions show a sharp acceleration in the rate of decline of SUV as Glc crosses 120 mg/dl. Background liver reveals very little change as Glc increases.
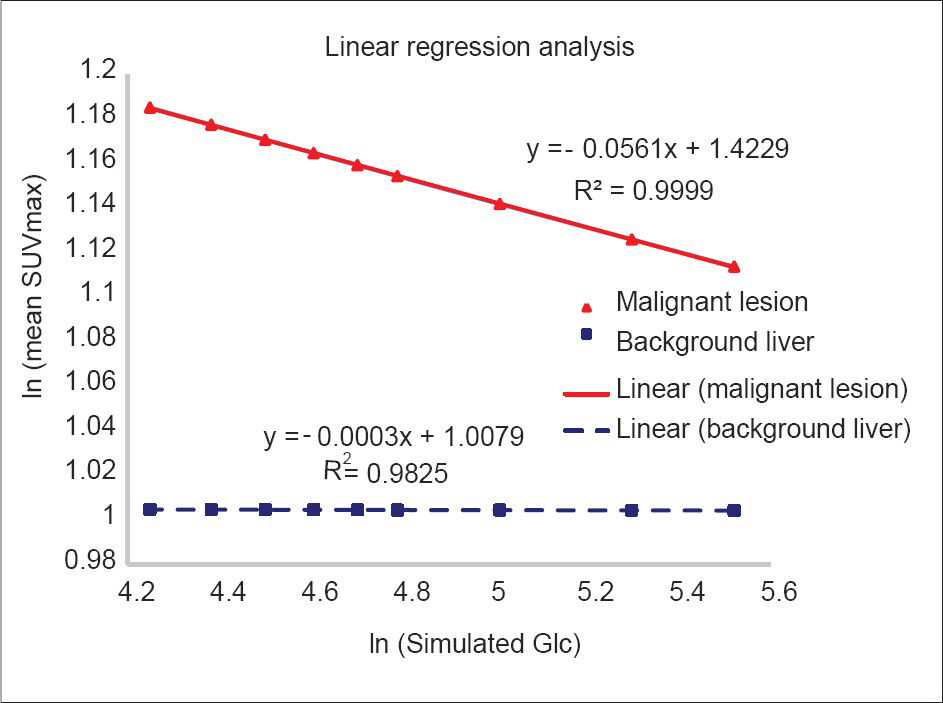
- Linear regressions following logarithmic transformation of the simulated data. The slopes of graphs match the mathematically derived rates of change from Equation (1).
Limitations
There was no Gaussian distribution of Glc levels for the cohort data, with a skew more toward normal levels. The potential differences in SUV between camera types, patient weight, and uptake time were not evaluated, although these factors were all equalized for the simulation data. The potential difference in glucose utilization by different tumor phenotypes was also not evaluated.
DISCUSSION
The quality of 18FDG PET imaging can be affected by a number of patient variables at the time of acquisition. One of the most important factors is the Glc since the radiotracer is a glucose analog.[13] In fact, the Society of Nuclear Medicine and Molecular Imaging strongly recommends reporting the Glc in the technical details of interpretations as a best practice measure.[14] It has been proven that the rate of 18FDG uptake will vary depending on tissue type.[15] Additionally, there have been prior studies demonstrating ambient Glc variation does affect uptake differently in several primary tumor types, including head and neck, lung, lymphoma, and pancreatic cancers.[10161718] However, the effects were not well quantified in such a way that could be easily incorporated into clinical practice.
In practice, glucose sensitivity is one of several variable factors that influence SUV, perhaps the most controllable factor aside from uptake time. Yet, the serum glucose level (Glc) has been somewhat neglected, apart from the tentative effort taken to control it within a physiologic range deemed “acceptable”. This is despite its potential for correction using Equation (4) to work backward and solve for the SUV under ideal Glc levels. This study quantified the impact ambient Glc had on SUV measurements, and demonstrated that the effective variability (g) was far more significant (by a factor of 100) in malignant hepatic lesions compared to normal liver, despite the liver being primarily involved in glucose metabolism. This difference in g is perhaps the best way to underscore the impact of Glc on the sensitivity of SUV. The data presented would appear favorable for universal applicability due to the wide variety of tumor types studied and the demonstrated volatility of liver metastases to variable ambient Glc. It also offers a simple method of correction for SUV at different Glc values, using the dimensionless exponent g.
Previously proposed semi-quantitative analyses of suspicious hepatic lesions, that relied on comparing tumor uptake to background tissue, would be adversely affected by elevated Glc, as there would be decreased separation between tumor SUV and normal liver SUV.[1920] Conversely, the study did prove that background liver uptake is an effective “control tissue” to perform such ratio (tumor to background) -driven semi-quantitative analyses due to its relative stability across a wide range of Glc. The rates of change derived from Equation (1) indicate that background liver is quite insensitive to variation in ambient Glc, while malignant lesions will be subject to far more volatility. Figure 2 shows that the SUV of a malignant hepatic lesion will significantly decrease, with an acceleration occurring at abnormal Glc >120 mg/dl, whereas background liver activity remains virtually constant.
The possibility of glucose-corrected SUV measurements could have very important clinical applications, especially in patients undergoing follow-up FDG PET scans during and after treatment. During treatment, patients are given various medications that may impact ambient glucose levels, such as steroids, to minimize treatment side effects. Patients can also have diseases, such as diabetes or occult glucose intolerance, which become much more difficult to control during the treatment process. Additionally, endogenous insulin sensitivity can be altered by body weight fluctuations from cancer and its therapies during and after the treatment course. These complicating factors can make glucose regulation very difficult. Yet, it may be still necessary to somehow compare the patient's clinical situation at a time of high glucose to prior scans when glucose control had been more feasible.
In order for valid temporal comparison of 18FDG PET scans, one could reason that the SUV should be obtained under similar conditions.[21] Using the quantitative corrections for tumors, it may be possible to know whether a decrease in SUV of a patient presently being treated is due to treatment response or just due to a temporal differences in Glc. Additionally, differences in the normal liver uptake between scans on the same patient could alert the interpreter that changes in SUV could be reflective of biologic rather than pathologic changes. Application of this type of glucose correction could save scans repeated for this technical factor and improve accuracy when interpreting serial PET scans to monitor tumor progression or treatment response.
The glucose sensitivity of different phenotypes may be an interesting area for future investigation. Unfortunately, we did not have enough number of cases of each tumor type to fully evaluate this factor. It will also be important to determine how patient weight, radiotracer dose variation, and uptake time between injection and image acquisition would affect SUVmax of hepatic lesions. Differences in diabetic patients would seem to be a reasonable consideration in this project; however, our institution gives detailed and individualized instructions for diabetic patients to ensure they are not in a high insulin state at the time of injection. Patients with non-fasting or hyperinsulin status at the time of injection were easily identified, and excluded on the basis of not having reasonable imaging quality. Therefore, there was no reason to stratify diabetic patients as a separate sub-group.
CONCLUSION
Malignant liver lesions have significantly greater sensitivity than background liver tissue to variations in serum glucose level at the time of radiotracer injection. As the glucose level rises, the measured SUV of the malignant lesions may decrease rapidly, while the background liver tissue SUV remains relatively constant. Consideration of glucose sensitivity not only provides the framework for correcting SUV from potential ambient glucose variation to provide more accurate temporal comparison, but also justifies the use of the simple tumor to normal liver ratio for evaluating suspicious hepatic lesions by 18FDG PET imaging.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/39/137832
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Progress and promise of FDG-PET imaging for cancer patient management and oncologic drug development. Clin Cancer Res. 2005;11:2785-808.
- [Google Scholar]
- Improvement of hepatic lesion characterization by 18F-FDG PET/CT with the use of the lesion to background liver activity ratio. Clin Nucl Med. 2013;38:869-73.
- [Google Scholar]
- Effects of blood glucose level on FDG uptake by liver: A FDG-PET/CT study. Nucl Med Biol. 2011;38:347-51.
- [Google Scholar]
- Clinical role of FDG PET in evaluation of cancer patients. Radiographics. 2003;23:315-40. quiz 533
- [Google Scholar]
- Normal blood glucose level and (18) F-FDG PET/CT. Rev Esp Med Nucl Imagen Mol. 2012;31:210-2.
- [Google Scholar]
- FDG PET: Elevated plasma glucose reduces both uptake and detection rate of pancreatic malignancies. J Nucl Med. 1998;39:1030-3.
- [Google Scholar]
- The influence of blood glucose levels on [18F] fluorodeoxyglucose (FDG) uptake in cancer: A PET study in liver metastases from colorectal carcinomas. Tumori. 1997;83:748-52.
- [Google Scholar]
- Quantitation of glucose uptake in tumors by dynamic FDG-PET has less glucose bias and lower variability when adjusted for partial saturation of glucose transport. EJNMMI Res. 2012;2:6.
- [Google Scholar]
- Effect of corrections for blood glucose and body size on [18F] FDG PET standardised uptake values in lung cancer. Eur J Nucl Med. 2001;28:919-22.
- [Google Scholar]
- Glucose-normalized standardized uptake value from (18) F-FDG PET in classifying lymphomas. J Nucl Med. 2005;46:1659-63.
- [Google Scholar]
- Investigating the existence of quantum metabolic values in non-Hodgkin's lymphoma by 2-deoxy-2-[F-18]fluoro-D-glucose positron emission tomography. Mol Imaging Biol. 2007;9:43-9.
- [Google Scholar]
- 2-deoxy-2-[F-18]fluoro-D-glucose-positron emission tomography sensitivity to serum glucose: A survey and diagnostic applications. Mol Imaging Biol. 2005;7:361-8.
- [Google Scholar]
- Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. J Nucl Med. 2006;47:885-95.
- [Google Scholar]
- Dynamic changes of FDG uptake and clearance in normal tissues. Mol Imaging Biol. 2013;15:345-52.
- [Google Scholar]
- The influence of plasma glucose levels on fluorine-18-fluorodeoxyglucose uptake in bronchial carcinomas. J Nucl Med. 1993;34:355-9.
- [Google Scholar]
- In vivo effects of insulin on tumor and skeletal muscle glucose metabolism in patients with lymphoma. Cancer. 1994;73:1490-8.
- [Google Scholar]
- Influence of the blood glucose concentration on FDG uptake in cancer: A PET study. J Nucl Med. 1993;34:1-6.
- [Google Scholar]
- Optimization of FDG-PET/CT imaging protocol for evaluation of patients with primary and metastatic liver disease. Int Semin Surg Oncol. 2007;4:17.
- [Google Scholar]
- Detection of hepatic metastases using dual-time-point FDG PET/CT scans in patients with colorectal cancer. Mol Imaging Biol. 2011;13:565-72.
- [Google Scholar]
- Effects of noise, image resolution, and ROI definition on the accuracy of standard uptake values: A simulation study. J Nucl Med. 2004;45:1519-27.
- [Google Scholar]






