Translate this page into:
Diffuse Hepatic Hemangiomatosis in the Adult without Extra-hepatic Involvement: An Extremely Rare Occurrence
Address for correspondence: Dr. Miguel Ramalho, Department of Radiology, Hospital Garcia de Orta, Avenida Torrado da Silva, Pragal - 2801-951, Almada, Portugal. E-mail: Miguel-ramalho@netcabo.pt
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
We present a case of a pathologically proven multinodular diffuse hepatic hemangiomatosis (DHH) with no extra-hepatic involvement in a 68-year-old male. Cavernous hemangioma is the most common hepatic tumor. However, DHH, which is characterized by extensive replacement of liver parenchyma with hemangiomatous lesions, has been rarely reported in adults. The etiology and clinical course are not completely understood because of its rareness, although the diagnosis might be suggested by the magnetic resonance imaging findings.
Keywords
Adult
hemangioma
hepatic hemangiomatosis
magnetic resonance imaging
ultrasound
INTRODUCTION

- Dr. Alexandre Batista
Liver hemangiomas are the most common benign liver tumors. Diffuse hepatic hemangiomatosis (DHH) is a rare disorder, usually occurring in the neonate, characterized by extensive replacement of liver parenchyma with hemangiomatous lesions.[1] It is frequently associated with a palpable abdominal mass and high-output heart failure, with high mortality rate in neonates. DHH in the adult, especially without extra-hepatic involvement, is extremely rare.[2] Hemangiomatosis differs from multiple or giant hemangiomas in that the boundary of the lesion is ill-defined, while in the latter, the liver parenchyma surrounding the lesion is compressed by a fibrous capsule forming the wall of the hemangioma.[3] The etiology and clinical course are not completely understood because of its rareness. The diagnosis may be based on the magnetic resonance imaging (MRI) findings. The final diagnosis is based on histological confirmation.[4]
CASE REPORT
An asymptomatic 68-year-old man was referred to our hospital due to the presence of numerous nodular hepatic lesions accidentally detected on ultrasound. There was a history of heavy drinking (alcohol intake around 70-90 g/d) and no prior history of medication or steroid intake. The other personal and family history was unremarkable. Physical examination upon admission revealed diffuse hepatomegaly, with no associated cardiac murmur or leg edema. Laboratory investigations showed a slight elevation of γ-glutamyl transpeptidase (76 UI/L; (normal value: 0-51 UI/L).
Abdominal ultrasound (US) showed multiple ill-defined sub-centimeter hyperechoic hepatic nodules, predominately in the right lobe, with no posterior acoustic shadowing or significant through transmission and absence of high vascularity on color Doppler [Figure 1].
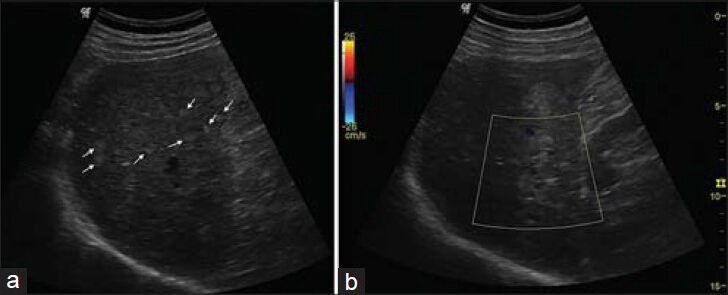
- 68-year-old male referred to our hospital due to multifocal hepatic lesions, subsequently diagnosed as diffuse hepatic hemangiomatosis. a) Ultrasound examination demonstrates multiple ill-defined yperechoic nodules dispersed throughout the hepatic parenchyma, mostly sub-centimeter and with right hemi-liver predominance. Some nodules are larger and better defined (arrows). b) Color Doppler shows no evident vascularity in the lesion.
Abdominal MRI was performed for further evaluation. Countless predominantly sub-centimeter nodular lesions with low T1-weighted signal intensity (SI) and moderately high T2-weighted SI were depicted. Liver lesions showed moderately restricted diffusion, and post-contrast fat-suppressed T1-weighted images showed predominantly hypovascular features with retention of contrast in later phases. A discontinuous centripetal filling appearance was noted on larger lesions [Figure 2]. There was no evidence of extra-hepatic masses or lymphadenopathy. A US-guided biopsy using an automated gun with an 18-gauge needle was performed collecting three representative fragments.
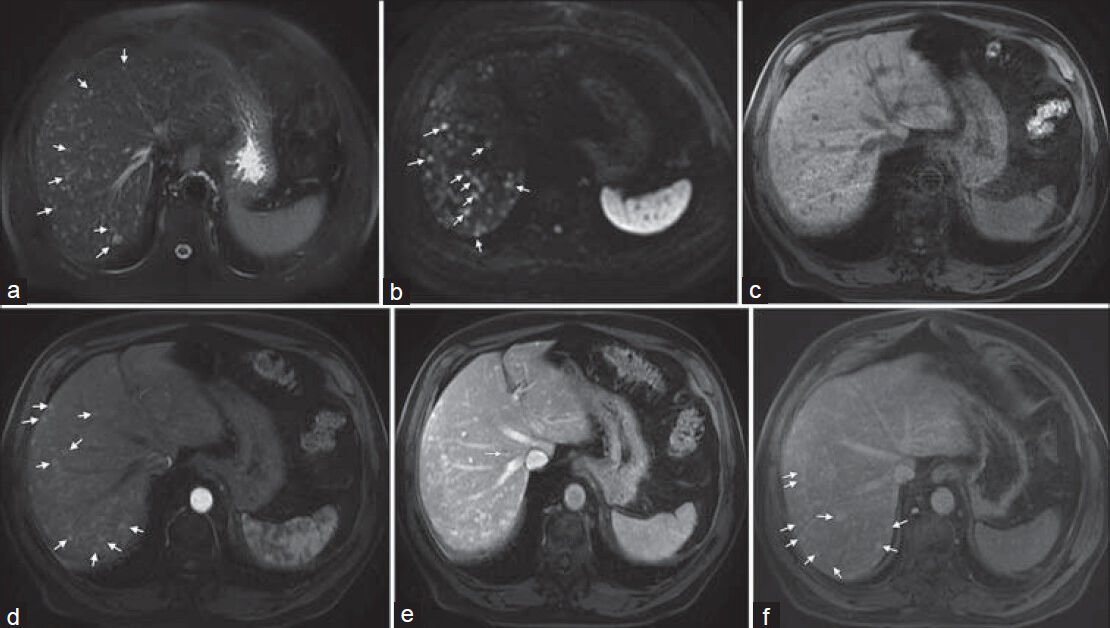
- 68-year-old male referred to our hospital due to multifocal hepatic lesions, subsequently diagnosed as diffuse hepatic hemangiomatosis. a) Axial fatsaturated T2-weighted image shows countless small-sized moderately bright lesions (arrows). b) Diffusion weighted image shows these lesions with high signal intensity (b = 600 sec/mm2) (arrows), which may be seen in liver hemangiomas secondary to T2 shine-through effect. Axial c) pre-contrast and (d-f) post-contrast fat-saturated T1-weighted images show arterial homogeneous enhancement (arrows, d), some exhibiting portal phase peripheral nodular enhancement (arrow, e) and late centripetal filling (arrows, f).
Pathology revealed endothelial-lined sinusoidal proliferation with erythrocyte content, consistent with hepatic hemangiomatosis [Figure 3]. No sarcomatous changes were seen. No evidence for extra-hepatic hemangiomatosis was revealed on further imaging studies. Definitive diagnosis of DHH was established based on a combination of imaging and pathological findings and clinical/imaging follow-up. Imaging follow-up included annual abdominal MRI investigation. Two years following the diagnosis, the patient's hepatic function and imaging findings remain stable, with no relevant associated symptoms.
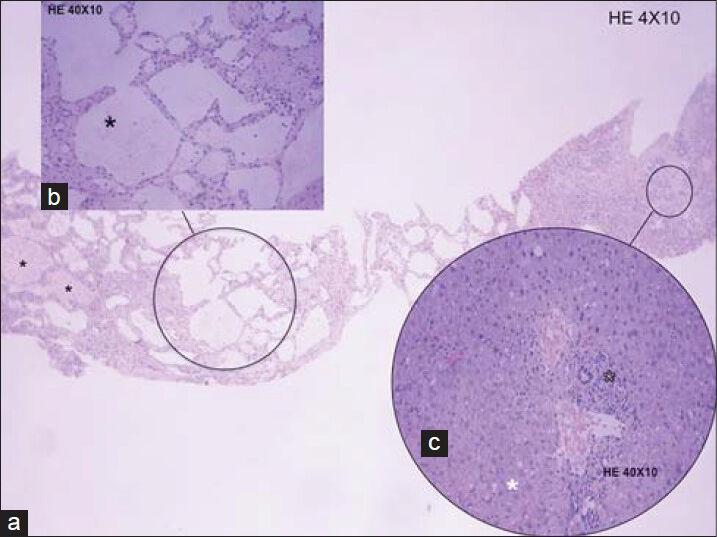
- 68-year-old male referred to our hospital due to multifocal hepatic lesions, subsequently diagnosed as diffuse hepatic hemangiomatosis. Ultrasound guided biopsy samples stained with hematoxylin and eosin a) 4 × 10, b) 40 × 10; c) 40 × 10). reveal numerous dilated nonanastomotic vascular spaces, which were lined by flat endothelial cells and supported by fibrous tissue endothelium. Some vascular spaces display erythrocyte content (black asterisks, a and b). Chronic inflammatory infiltrates with peri-portal distribution are also present (black outlined asterisk, c). Note the presence of associated macrovesicular steatosis (white asterisk, c).
DISCUSSION
Imaging plays a vital role in the detection and characterization of multifocal liver lesions in adults. There are numerous causes including benign and malignant neoplasms, infectious lesions, and inflammatory conditions. The US evaluation showed multiple sub-centimeter, ill-defined, hyperechoic nodules dispersed throughout the liver parenchyma, predominantly in the right hemi-liver, with very little or no flow seen on color Doppler. The differential diagnoses included metastatic disease, multifocal nodular fatty infiltration of the liver, bile duct hamartomas (BH), or other less-frequent entities such as DHH or angiosarcoma. MRI was performed to aid in the differential diagnosis. The imaging findings were suggestive of DHH.
Metastatic liver involvement usually occurs in a patient with a known extra-hepatic primary neoplasm. Small hypervascular liver metastases (e.g. from breast or neuroendocrine tumors) may mimic small early uniform enhancing hemangiomas; however, these lesions tend to fade or wash out, whereas hemangiomas are more likely to maintain the SI or fade toward background isointensity.[5] Also, the majority of liver metastases shows heterogeneous or ring enhancement on the arterial phase images, confirming their definite diagnosis.
Multifocal nodular fatty deposition is an unusual pattern of liver steatosis that consists of multiple round or oval fat foci randomly distributed throughout the liver. MRI is usually required to make the diagnosis. Fat deposits appear isointense or hyperintense to the liver on in-phase images, dropping SI on out-of-phase images. Fat accumulation does not show restriction or shine-through effect on diffusion-weighted images. Other clues indicative of multifocal fat deposition are the lack of mass effect, stability in size over time, and contrast enhancement similar to or less than that of the surrounding liver parenchyma.
BH, also called von Meyenburg complexes, originate from embryonic bile ducts that fail to involute. On ultrasound, BH have been described as innumerable tiny hypoechoic or hyperechoic foci measuring less than 10 mm and distributed uniformly throughout the liver, with some displaying a comet tail artifact.[6] Differences in echogenicity may be due to the size of the dilated bile duct component, which, at a certain size, would behave like other microcystic structures.[6] On MRI, bile duct hamartomas usually appear as multiple tiny cystic lesions, strongly hyperintense on T2-weighted images. After gadolinium administration, some authors described a thin rim of enhancement, which correlates with the compressed liver parenchyma surrounding the lesions.[7] Internal progression or retention of contrast is not seen in BH.
Angiosarcoma is the most frequent malignant mesenchymal hepatic tumor and is commonly multiple. It is very aggressive and sometimes may mimic hemangioma on dynamic studies, showing high SI on T2-weighted images, since both tumors contain abundant blood-filled vascular spaces; also, heterogeneous centripetal and progressive enhancement has been described.[8] Its definite diagnosis is based on histological findings. As no definite radiological findings can precisely distinguish DHH from angiosarcoma, the final diagnosis was histologically established.[4]
DHH is a rare, although benign, condition that is characterized by diffuse replacement of liver parenchyma by hemangiomatous lesions.[4] The natural history and prognosis of hepatic hemangiomatosis are poorly understood due to its rarity. Hemangiomatosis is different from multiple or giant hemangiomas in that the boundary of the lesion is ill-defined, while in the latter, the liver parenchyma surrounding the lesion is compressed by a fibrous capsule that forms the wall of the hemangioma.[3]
DHH can occur in all age groups, but is most frequent in the neonate where it can result in high-output cardiac failure and significant mortality. It is usually asymptomatic in adulthood, being extremely rare when not associated with giant cavernous hemangiomas and even rarer when no other organ is affected besides the liver.[2]
The imaging findings of DHH are comparable to those of other common hepatic hemangiomas.[49] There is an association with giant cavernous hemangiomas.[10] Two patterns of DHH are described, the diffuse (non-nodular) and the nodular pattern, the former being the predominant pattern, representing approximately two-thirds of the cases.
Sonographically, the liver affected by DHH appears as homogeneous hyperechoic areas with poorly defined margins (diffuse pattern) or a heterogeneous echo pattern with multiple discrete or coalescent small hyperechoic nodules (multinodular pattern). These nodular lesions are variable in size (usually <5-10 mm), appearing as countless oval or round hyperechoic nodular lesions. Very little or no flow is seen on color Doppler.[910]
On MRI, the areas affected by hemangiomatosis may also display a diffuse non-nodular pattern composed of tiny cystic spaces separated by numerous thin hypointense septa, creating a lattice-like appearance or a diffuse nodular pattern consisting of multiple small discrete and coalescent nodules on T2-weighted imaging.[10] The extremely low velocity of blood flow within hemangioma relates to the absence of flow void seen in MRI imaging. Dynamic contrast-enhanced MRI shows heterogeneous enhancement in these areas during the arterial phase that becomes more homogeneous during portal and delayed phase imaging. The multinodular type, as was our case, exhibits small discrete and coalescent nodules with early homogeneous enhancement or the typical peripheral discontinuous enhancement during the arterial phase, showing uniform late retention of contrast.[4910]
CONCLUSION
In conclusion, DHH with no associated cavernous hemangiomas and without extra-hepatic involvement is an extremely rare condition. Despite its rarity, it can be suggested in adults presenting with multiple, predominantly sub-centimeter hepatic lesions demonstrating MRI features of hemangiomas. MR imaging may be superior to US and computed tomography (CT) for the diagnosis and is the preferable cross-sectional imaging technique for long-term follow-up.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/43/139733
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Diffuse neonatal hemangiomatosis without cutaneous lesions in an adult: A case report. Angiology. 2002;53:235-7.
- [Google Scholar]
- Diffuse neonatal haemangiomatosis: New views on diagnostic criteria and prognosis. Acta Paediatr. 1999;88:93-7.
- [Google Scholar]
- Diffuse hepatic hemangiomatosis without extrahepatic involvement in an adult patient. Korean J Radiol. 2008;9:559-62.
- [Google Scholar]
- Hepatic hemangiomas: A multi-institutional study of appearance on T2-weighted and serial gadolinium-enhanced gradient-echo MR images. Radiology. 1994;192:401-6.
- [Google Scholar]
- Sonographic features of biliary hamartomas with histopathologic correlation. J Ultrasound Med. 2006;25:1631-3.
- [Google Scholar]
- Cystic focal liver lesions in the adult: Differential CT and MR imaging features. Radiographics. 2001;21:895-910.
- [Google Scholar]
- Angiosarcoma of the liver mimicking cavernous hemangioma on dynamic CT. J Comput Assist Tomogr. 1989;13:910-2.
- [Google Scholar]
- Association of hepatic hemangiomatosis with giant cavernous hemangioma in the adult population: Prevalence, imaging appearance, and relevance. AJR Am J Roentgenol. 2011;196:809-15.
- [Google Scholar]






