Translate this page into:
Imaging Spectrum of Hemangioma and Vascular Malformations of the Head and Neck in Children and Adolescents
Address for correspondence: Dr. Venkatraman Bhat, 309, Greenwoods Apartment, Royal Gardenia, Bommasandra, Bangalore - 560 099, Karnataka, India. E-mail: bvenkatraman@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Vascular lesions of the head and neck region in children constitute an interesting group of lesions that benefit immensely from imaging techniques. Imaging is essential for identification, characterization, and delineation of the extent of lesion and subsequent follow-up. Infantile hemangiomas, which are vascular tumors with a specific evolution pattern, constitute a large majority of these lesions. On the other hand, there are vascular malformations, which are anomalies of the vascular system, consisting of a range of vascular tissues associated with various flow patterns. When diagnosis is clinically evident, imaging should utilize non-radiation techniques and address the issues necessary for management. Timing and interpretation of imaging methods employed in assessing childhood vascular lesion should also take into consideration the natural history so that imaging is performed to address a specific question. This review highlights the typical appearance of a hemangioma and a group of vascular malformations of the head and neck. For descriptive purpose, an attempt has been made to group lesions into specific subsites, with each one having specific clinical significance. Cases included illustrate the spectrum of the disease ranging from classical form in young children to slightly differing manifestations of the disease in adolescents and adults. The illustrations also provide a novel way of presenting image data using volume-rendering techniques of 3D data. Multi-modality team interaction and management strategies of these complex lesions are also emphasized.
Keywords
Hemangioma
head and neck
imaging
vascular malformation
venous malformation
INTDODUCTION

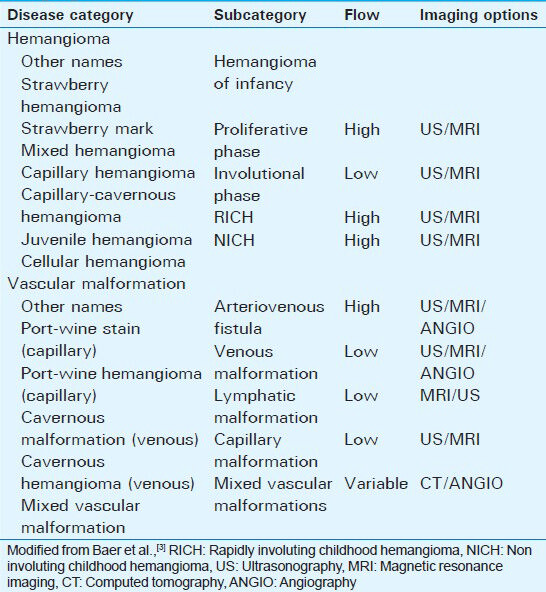
In 1982, Mulliken and Glowacki proposed a biological classification of vascular birthmarks on the basis of their clinical manifestations, histopathologic features, and natural history.[1] Following further refinement by multiple contributors, the modern classification scheme for vascular anomalies was accepted by the International Society for the Study of Vascular Anomalies (ISSVA) in 1992.[123456] Understanding this classification is necessary for proper interpretation of imaging features of the lesions. While infantile hemangiomas represent vascular tumors evolving a few weeks after birth, vascular malformations (VMs) are present at birth and grow proportionally with the growth of the child. VMs have normal endothelial turnover and do not spontaneously regress.[1] VMs are categorized into arterial, venous, and capillary types and are subcategorized by their relative flow velocity.[1] There is large variation in the terminology used in literature describing various vascular lesions. Imaging findings do not provide discrete differentiating features of one type of lesion over another. Understanding the current nomenclature in relation to previous literature is necessary for consistent description [Table 1].
The life cycle of a hemangioma differs from that of most tumors in that it demonstrates a phase of rapid proliferation followed by slow spontaneous involution, and in most cases, complete resolution. The mechanisms that control the involution of hemangiomas are poorly understood. Genetic techniques have been useful in linking some rare hereditary vascular disorders to the defective regulation of angiogenesis and vasculogenesis.[7] During the proliferative phase, hemangiomas are composed of densely packed endothelial cells that form small capillaries. In the phase of involution, there is a decrease in cellularity and an increase in the fibrous and fatty components with reduced vascularity. Imaging appearances parallel the life cycle very closely.
A careful imaging plan is necessary for the evaluation of these lesions. The role of imaging is much more vital in the diagnosis of lesions covered by skin. In view of the self-limiting nature of some of the entities, central theme of imaging methods should be focused on employing non-radiation methods. Ultrasonography and magnetic resonance imaging (MRI) techniques should be the initial choice and in follow-up studies. Once identified, the assessment of the lesion should differentiate slow- and fast-moving circulation. Doppler sonography differentiates arteriovenous malformations (AVMs) from other lesions such as venous-malformations and capillary hemangiomas. Size and volume of lesion is important in hemangioma, in order to establish growth pattern and response to therapy, if employed. Vascular pattern, including feeding arteries, draining vein, and matrix, is of importance to the interventional radiologist, if specific therapy is envisaged. Surgical management aimed at cosmetic improvement or debulking requires additional information regarding the tissues involved and underlying bony structures. Regarding the demonstration of the extent of lesion, volume of the affected area, and anatomical information of the surrounding vital structures, MRI techniques provide the best option.[2] Although MR vascular imaging techniques provide information regarding the feeder vessels, they lack detailed information required for vascular intervention. Computed tomography (CT) angiography provides comprehensive information on long-standing lesions, lesions in adolescent and adult patients, and lesions involving osseous structures. In a study focused on the facial region, the role of ultrasound as a cost-effective and efficient tool in the management of hemangioma of the maxillary region is emphasized.[89] In particular, vessel density as depicted on Doppler sonography has been used in differentiating VM from a variety of other masses.[10] Donnelly et al., found sonographic depiction of abundant low-flow vascular channels to be a predictor for the potential success of percutaneous sclerosis.[1011] Sonography also provides per-operative guidance for lesion resection by localizing leading margin and for needle placement during percutaneous sclerosis.[10] Effective management is possible by a multidisciplinary team of pediatricians, hematologists, surgeons, and radiologists for diagnostic work-up and therapeutic options.
This pictorial essay illustrates the vascular tumors and malformations involving the head and neck, mostly in children and adolescents.
HEMANGIOMA-VM SPECTRUM IN THE HEAD AND NECK REGION
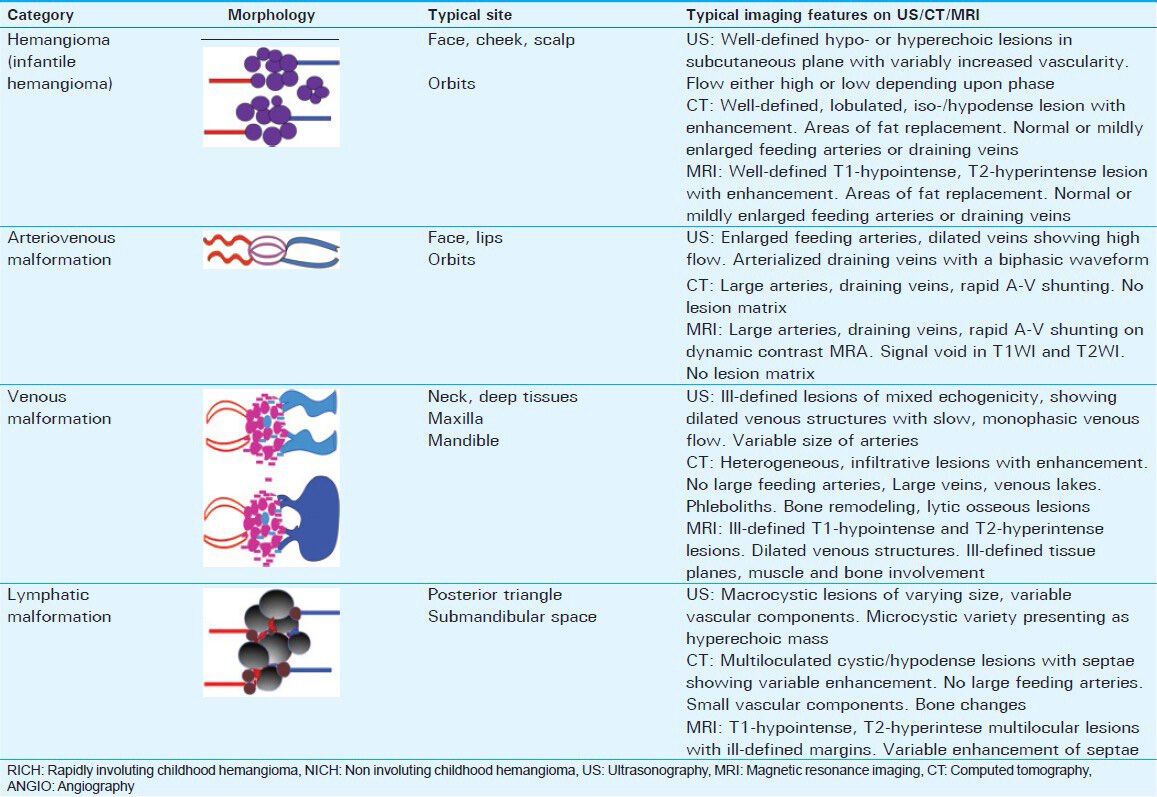
Infantile hemangioma is the most common vascular tumor of the newborn, with an incidence of 12-23% among preterm infants with low birth weight and a female to male ratio of 3:1. The head and neck region is the most frequently affected area (60%).[3] Although most lesions (80%) are solitary, approximately 20% of affected infants present with five or more lesions.[38] At birth, the lesions are either absent or small. When visible, they present either as an erythematous macule, a blanching macule, or an area of localized telangiectasia.[58] Rapid proliferation occurs shortly after birth and lasts several months. During the growth period, there is high expression of angiogenic factors such as vascular endothelial growth factor and basic fibroblast growth factor. The typical hemangioma will start to involute approximately 10 months after birth and half the lesions are completely resolved by the age of 5 years.[1011] From an imaging point of view, congenital hemangiomas have the same imaging features as hemangiomas of infancy except that congenital hemangioma can have intravascular thrombi, larger venous components, and sometimes vascular aneurysms.[23] Precise location of the hemangioma in relation to the anatomical space has a great bearing on symptoms and prognosis. An orbital apical lesion tends to cause visual defects by compressing the optic nerve. Lesions in the external auditory canal can lead to conductive deafness. Subglottic lesions present with stridor and respiratory difficulty. Early identification of the lesion is essential in all these patients for prompt management. The following presentation will illustrate various site-specific examples of vascular lesions under the basic classification of VMs [Table 2].
Hemangioma
Major categories include hemangioma of infancy and congenital hemangioma. Site and extent of the lesion vary greatly as illustrated by their presence at several locations of the head and neck region. Congenital hemangiomas are categorized into rapidly involuting congenital hemangioma (RICH) and non-involuting congenital hemangioma (NICH). Involuting lesions generally regress within 14 months.[3] Contrary to the popular belief, infantile hemangiomas are not associated with Kasabach-Merritt phenomenon. Typically, MRI and ultrasonography are used in the evaluation. On MRI examination, lesions are well-defined, lobulated, homogeneously T1 hypo-isointense, and show gross T2 hyperintensity. The lesion becomes heterogenous with small focal areas of fat replacement during the involutional phase. Variable vascularity may be demonstrated. Extent of the lesion is well demonstrated by contrast-enhanced examination.
Scalp
The scalp is one of the typical sites for hemangioma in infants. Lesion size could greatly vary from a tiny focus to a large mass [Figure 1]. Clinical presentation ranges from asymptomatic incidental detection to consumption coagulopathy. High-resolution sonography with power and color Doppler study is the best modality for evaluation of small superficial, generally flat or minimally elevated lesions on the scalp. Superficial scalp lesions are easily detected by high-resolution sonography in the proliferative phase. Lesions vary greatly in echogenicity, and show sizable lesion matrix and high vascularity. On Doppler examination, there is no arterialized wave pattern seen in draining veins.

- 3-month-old male child incidentally found to have a soft compressible lesion over the vertex, diagnosed as scalp hemangioma. a) T1-weighted axial and b) T2-weighted sagittal MR images of head demonstrate a focal lesion of intermediate signal intensity, without showing intracranial communication (open arrows).
Skull
Occasionally the bony calvarium is the site of a hemangioma presenting as an expansile bone lesion involving predominantly the outer table with coarse trabecular pattern [Figure 2]. The margins usually are not sclerotic or beveled. Prominent vascular grooves can be seen. Typical patterns like sunburst appearance and honeycomb appearance have been described on radiography.[12] MRI examination is the method of choice for the demonstration of small lesions. Additionally, it allows for the assessment of intracranial extension and shows associated neural malformations.

- 3-month-old female with nontender, left parietal scalp swelling diagnosed with skull hemangioma. On MR examination of head, a) T1 axial image shows hypointense and T2 images b) axial and c) sagittal views show hyperintense lesion in left posterior parietal region (arrows). Lesion predominantly involves outer cortex and d) contrast image shows enhancement (arrow) and focal deficiency of inner table (arrowhead).
Orbits
Hemangiomas constitute a common cause of orbital mass. Histologically, most of them are cavernous hemangiomas. The rest constitute capillary hemangiomas, hemangioendotheliomas, and AVMs. In terms of orbital compartments, all orbital spaces can be involved [Figures 3 and 4]. In an analysis of a large group of orbital lesions, cavernous hemangiomas contributed to 9% of the total number of cases. The most frequent location was the outer lower quadrant[13] [Figure 5]. The orbital bony wall could also be the site of a primary hemangioma. Imaging modalities like CT and MRI are used for the precise delineation of the lesion in relation to the orbital cone, to assess compression of the optic nerve, to check for tumor extension into the intracranial compartment, and for the demonstration of specific vascular components of the lesion. On CT imaging, the lesion is homogenous, isodense to minimally hypointense to muscle, and shows uniform contrast enhancement. MRI examination reveals T1 iso-hypointense lesion with a homogenous pattern in the proliferative phase. Heterogenous pattern is noted in the involuting phase due to fat replacement. Lesions show moderate-marked T2 hyperintensity. Most of the lesions enhance intensely on intravenous gadolinium. Prominent draining veins and occasionally mildly enlarged feeding arteries are demonstrated on MR angiography.

- 2-year-old male with right orbital swelling, suspected to be an orbital hemangioma. a) Axial CECT at the level of the mid orbit demonstrates a diffusely enhancing hemangioma (arrow) of the right upper eyelid. b) 5-year-old male with left proptosis diagnosed with orbit hemangioma, contrast CT examination shows extraconal, uniformly enhancing lesion (arrow) along the medial aspect of the left orbit.

- 2-month-old female with mild right orbital proptosis diagnosed with orbital hemangioma. a) Non-contrast axial CT image demonstrates well-defined intraconal lesion (black arrow). b) Contrast-enhanced T1W MR images show marked, uniform enhancement suggesting an intraconal hemangioma (black arrow).

- 18-year-old male with right periorbital swelling diagnosed with orbital hemangioma. MR T2-weighted images a) axial b) coronal demonstrate a large hyperintense, lobulated soft tissue lesion extending to orbit in the extraconal space, lateral to the orbital cone (arrows). c) Contrast-enhanced T1 image shows extensive near-homogeneous enhancement. Extent of the lesion is better demonstrated following intravenous contrast (arrow).
Face-focal lesions
Hemangioma of the face leads to facial deformity and disfigurement. Infantile hemangiomas are the most frequent lesions encountered in the face [Figures 6 and 7]. These lesions are found in the skin and subcutaneous tissues of the facial region, with typical specific subsites being lips [Figure 8], cheek [Figure 9], the nasal cavity [Figure 10], airways [Figure 11], etc., Larger lesions tend to affect all tissue planes. The mandible and maxilla are frequently affected by large vascular lesions of the facial region [Figure 12]. Salivary glands, the parotid gland in particular, could be the site of a VM [Figure 13]. Sonography plays a major role in the characterization of facial lesions [Figure 14]. However, additional imaging modalities like CT are often required to demonstrate the complete extent. Involvement of the bony structures and relation of vascular structure to bone is best studied by CT. Careful comparison of plain and contrast-enhanced CT (CECT) allows visualization of intra-osseous and extra-osseous components of the malformation.

- 2-month-old neonate with a disfiguring left facial lesion involving the ear lobe, diagnosed as hemangioma. a) T1W axial image shows hypointense lesion (arrow) and b) T2W coronal MR images show well-defined, lobulated hyperintense lesion involving ear, parotid, and adjacent cheek (arrow).

- 5-year-old male with swelling in the left cheek adjacent to the nose diagnosed as hemangioma of the face. MRI a) coronal T1W and b) postcontrast T1W fat-suppressed images show an enhancing lesion in medial cheek extending to the ala of nose (arrows).

- 7-month-old female with hemangioma in the upper lip. a) Clinical photograph demonstrates localized, compressible soft tissue swelling involving the right lateral aspect of the upper lip. Contrast-enhanced MRI b) axial and c) sagittal T1 images show intensely enhancing localized lesion involving the upper lip. Dark areas at the anterior aspect of the lesion (arrows) represent signal void due to high flow in blood vessels.

- 8-month-old female with hemangioma of left cheek. MRI a) T1W axial image shows hypointense lesion (open arrow), b) T2W axial image demonstrates well-defined lobulated hyperintense lesion involving left side of cheek (open arrow), c) contrast-enhanced MIP images of MR venogram demonstrate intense enhancement of the lesion (arrow).

- 10-year-old male presenting with a nasal obstruction; endoscopic and biopsy diagnosis was cavernous hemangioma. a) T1-weighted MR image shows the enlargement of the inferior turbinate (arrow) with T1 hypointensity, almost involving the whole length. Contrast-enhanced T1 images in b) axial and c) sagittal planes show enhancement of the lesion (star) with some non-enhancing component anteriorly.

- 14-year-old female with muffled voice diagnosed with hemangioma. Axial a) plain and b) CECT images of neck demonstrate uniformly enhancing hemangioma of the left pyriform sinus (white arrows). The lesion is projecting into and compromising the airway. There are prominent branches of the left external carotid artery (black arrow).

- 10-year-old male with right facial swelling diagnosed as hemangioma. a) Panoramic view of mandible shows an osteolytic lesion in the mandible (arrow). b) Non-contrast coronal CT image shows expansile lytic lesion in the right mid body of mandible with preserved cortex (open black arrow). c) T2-weighted MR image shows signal void within the body of mandible (arrow), indicating a high-flow lesion. d) Post-contrast axial CT image demonstrates marked enhancement of the cavity, indicating vascular nature of the lesion (solid arrow). Image also shows additional enhancing soft tissue component (arrowhead). Finding indicative of mixed vascular malformation with intra-osseous component.

- 8-month-old female with compressible non-tender swelling of the right parotid gland diagnosed with hemangioma. a) Axial non-contrast and b) contrast-enhanced images demonstrate a uniformly hypodense enlargement of the right parotid gland showing homogeneous intense enhancement (star). There is no significant enlargement of the carotid branches. Finding characteristic of a hemangioma.

- 13-year-old female with lower lip swelling suggestive of hemangioma. a) Clinical picture demonstrating diffuse non-tender and compressible swelling of lower lip. b) Sonographic, gray-scale image shows hyperechoic lesion with a large blood vessel (arrow). c) Power Doppler and d) color Doppler images of the region demonstrate enlarged vessels (open arrows) and high density of blood vessels within the lesion.
Extensive lesions of the face
Extensive hemangiomas of the neck and face may be associated with multisystem anomalies as in PHACES syndrome [Figure 15]. Components of PHACES syndrome include posterior fossa malformations, hemangioma of the cervico-facial region, arterial anomalies, cardiac anomalies, eye anomalies, and sternal or abdominal clefting or ectopia cordis. This syndrome has a marked female predilection (female:male ratio of 9:1) and is thought to represent a developmental defect that occurs between 8 and 10 weeks of gestation.

- Female neonate with PHACES syndrome. a) Clinical image demonstrates an extensive vascular malformation involving the right side of face, cheek, and lip. b) Coronal cranial T2W MR image demonstrates right cerebellar, vermian hypoplasia (black arrow) with enlargement of cisterna magna. c) Enhanced T1W MR coronal image demonstrates uniformly enhancing lesion involving a subtemporal, and infratemporal regions with a subcutaneous extension (star). d) Contrast-enhanced axial CT image shows an enhancing lesion in the right internal auditory canal (open arrow), representing a small hemangiomatous component intracranially.
Recent observations suggest that infants with these associated arterial anomalies are at risk for cerebrovascular occlusive disease. In some patients, hemangiomatous lesions can also be seen in posterior fossa, particularly in the region of internal auditory canal, as illustrated in our cases [Figure 16a and b]. Intracranial VMs tend to be ipsilateral to the predominant facial lesions. MRI evaluation plays a critical role in defining the total extent of the lesion and associated intracranial malformations. Large facial hemangioma may be associated with visceral hemangiomas which increase the morbidity significantly.[11]

- Newborn female with infantile hemangioma of the face. a) Photograph demonstrates the extensive hemangiomatous lesion involving the left side of face, orbit, forehead, and the cheek. b), c) CECT images in axial plane demonstrate a grossly enhancing orbital lesion with pre-septal, post-septal components (open arrow). Widened extracerebral CSF spaces in both temporal regions (star). d) Axial CT image at the level of internal auditory canal demonstrates enlargement of the left internal auditory canal (arrow). Following the IV contrast, there is intense enhancement of soft tissues in the left internal auditory canal (c). e) T2-weighted coronal MR image demonstrates grossly hyperintense vascular lesions involving the left orbit, surrounding the globe (open arrow). f) T1 fat-suppressed coronal contrast-enhanced MR shows lesions involving facial and neck soft tissues, reaching up to the occipital region (open arrow). g) MR image at the level of internal auditory canal shows focal intensely enhancing lesion (circle). h) Clinical follow-up picture of the patient after medical management for 18 months (patient's age 1 year 9 months) shows marked regression of the lesion.
Neck
Subglottic hemangiomas represent an important manifestation of infantile hemangioma. Tracheal lesions are associated with cutaneous lesions in more than 50% of cases [Figure 17a and b]. Hence, a careful scrutiny for skin lesions is recommended in any infant with stridor or hoarseness of voice. MR imaging plays a major role in detecting the airway lesions after thorough scrutiny by microlaryngobronchoscopy. Due to the small size of the lesion, careful MR imaging technique is required for identification of this lesion. Lesions are bright on T2 sequence and, hence, can be demonstrated with ease despite their small size. Other locations in the neck include classical subcutaneous lesions and occasional lesions in multiple tissue planes [Figure 18a and b]. Deep-seated lesions in the nasopharynx and neck spaces pose a particular challenge for imaging diagnosis as well as for detection.

- 3-month-old female child with stridor and cutaneous lesions diagnosed as hemagioma of the neck. a) Coronal T1-weighted, contrastenhanced image shows enhancing lesion in the right infraglottic region of the trachea and on right side of the face (black open arrow). b) Contrast-enhanced axial image demonstrates a multifocal enhancing lesion on the right side of the subtemporal region and both sides of the suboccipital and occipital regions (arrows). c) Reconstructed CT sagittal image shows subglottic polypoidal lesion (arrow). d) Axial T2W MR image at the same level shows a marked T2 hyperintensity of the lesion (arrow), characteristic of hemangioma.

- Adolescent male with neck swelling and difficulty in swallowing diagnosed with neck hemangioma. Contrast-enhanced reconstructed CT images in coronal plane. a) Early phase shows few dot-like enhancing areas representing feeding arteries (arrow); large part of the lesion is not enhancing. b) Mid arterial phase shows a patchy enhancement of the lesion, some of them representing dilated veins (open arrow). c) Venous phase demonstrates a large component of the lesion to be vascular with some venous component along the oropharynx (black arrowhead). 3D-rendered volumetric sagittal d) arterial, e) venous, and f) coronal venous images demonstrate arterial and venous components of the lesion. Arterial components are noted surrounding the internal carotid artery (black open arrow). Venous components along naso-oropharynx (black arrows).
Vascular malformation
VMs are identified by the predominant vessel type and grouped into arterial, venous, capillary, and mixed malformations. Clinical and imaging expressions of the subsets of complex vascular lesions belonging to VM category are better appreciated in adolescent age groups due to clear expression of the primary and secondary imaging features. Most such lesions are venous malformations or mixed VMs. Venous malformations are the most common lesions of head and neck region after hemangiomas. Ultrasound plays a major role in differentiating the lesion into high- and low-flow lesions. Time-of-flight (TOF) MR angiography is of limited value in demonstrating specific details of feeding vessels. Although contrast-enhanced angiography is superior to TOF angiography in demonstrating arterial structure, it does not give specific details needed for interventional procedures [Figure 19].
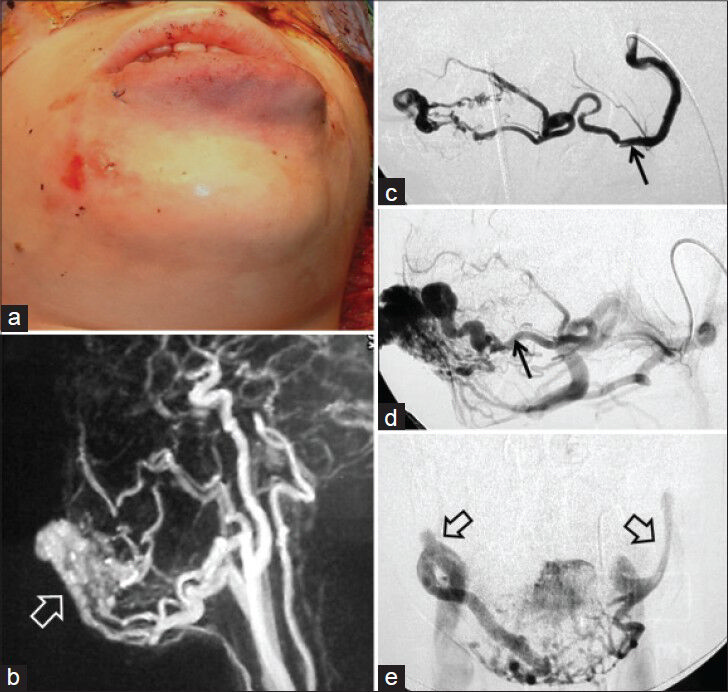
- 13-year-old female with hemangioma of the lower lip. a) Clinical photograph demonstrates diffuse swelling of the lower lip. b) Contrast-enhanced MR angiography demonstrates vascular supply to the lesion as well as lesion matrix (open white arrow). c and d) Serial images of angiographic study following selective lingual artery injection demonstrate early (arrow) and late arterial phases (arrow) of an arteriovenous malformation. e) Frontal images in venous phase show the draining veins bilaterally (open arrows). MR angiogram, although useful, lacks specific details.
Arteriovenous malformation
This lesion shows high-flow pattern consisting of enlarged feeding arteries draining into veins without a capillary network. Hence, there is minimal or insignificant parenchymal component. Presentations vary widely in phase and growth pattern. Hormonal changes, infection, trauma, and surgical injury can all result in rapid enlargement of the lesion. On grayscale sonography, it is seen as a poorly defined heterogenous structure interspersed with fat. Doppler ultrasound reveals a network of multiple arteries with increased diastolic flow and arterialized draining veins with a biphasic waveform.[2] MR and CT angiography can be valuable in visualizing the characteristic appearance of AVM. Venous enhancement is early and intense, and venous lakes are generally not present. Like proliferating hemangiomas, high-flow lesions produce flow voids on spin echo sequences and high signal intensity on gradient echoimaging.[2] Sclerotherapy is contraindicated for high-flow lesions.
Venous malformation
Slow-flow lesions like venous malformations appear on imaging as lobulated or multilobulated isodense/isointense lesions. On cross-sectional imaging, lesions crossing the tissue planes and showing multiple phleboliths are best seen on CT [Figure 20a–d]. Enhancement is variable and phasic following intravenous contrast.[14] Multiphasic dynamic imaging with contrast enhancement is necessary for demonstrating the temporal changes within various components of the lesions. A characteristic of a venous malformation is the tendency for the lesion to spread along different tissue planes, a behavior that is not unlike that of a malignant lesion. At times, imaging methods may not clearly differentiate a venous malformation from other highly vascularized tumors. Lesions in adolescents often show an extensive involvement of structures including the bone, muscles, and subcutaneous planes. Many present with bone overgrowth, gross deformities, and abnormal bone remodeling [Figure 21a and b]. CT is a valuable modality in the diagnosis of venous malformations because it readily identifies small calcified phleboliths associated with the lesion. CT can also demonstrate bone involvement and remodeling secondary to the lesion, a finding characteristic of VMs but uncommon to hemangiomas. Pathognomonic appearance of a phlebolith in VM has been demonstrated in 15-25%. of lesions. 3D reconstructions of CT images show a typical pattern of multiple “shining pearls” in venous malformation (Bhat et al., under publication, IJA Thieme) [Figure 22]. These findings are increasingly seen on CT images. Multimodality discussion and planning is essential for the management of extensive venous malformation in adolescents. A combination of treatment options is to be envisaged while dealing with different components of these lesions. Numerous surgical techniques call for a demonstration of the tissue matrix of the VM along with the delineation of a major feeding artery and draining veins.

- 11-year-old female with mixed vascular malformation on the right side of the face with osseous involvement. a) 3D surface-rendered image shows gross soft tissue swelling. b) Image highlights prominent superficial vessels of the lesion. 3D-rendered contrast-enhanced arterial phase images in c) coronal and d) sagittal planes highlight prominent vessels constituting the lesion (star). 3D reconstruction in venous phase in e) coronal and f) sagittal planes show extensively enhancing lesions without clear boundary, involving the infratemporal fossa, tongue, and adjacent overlying facial soft tissues. g) Coronal contrast-enhanced image shows enhancing lesions without clear demarcation, involving the subcutaneous tissues, muscles, and bone (open arrow). h) 3D bone reconstruction shows grossly enlarged mandible with osteolytic lesions. Note also few “shining pearls” (arrow) indicating phleboliths.

- 14-year-old female with facial deformity, diagnosed as mixed vascular malformation involving the lower jaw, tongue, and mandible. a) CT data 3D rendering of the bony structures demonstrates abnormal shape of the mandible, involving the alveolar process and associated deformity of the teeth (arrow). b) Additional window settings show intrinsic coarse trabecular structure of the mandible. Also note large foramen on the right side, indicating large vascular pathways traversing (open arrow). c) Contrast-enhanced axial CT images demonstrate enlarged lingual arteries (black arrows) leading to a large vascular malformation of the tongue and left hemi-mandible. d) 3D-rendered sagittal image with vascular preset provides realistic impression of the feeding arteries (open arrow), lesion matrix, and relation of lesion to the osseous structures.

- Adult with a swelling in the neck diagnosed as venous malformation. a) Non-contrast CT oblique–sagittal image shows multiple phleboliths in a lobulated lesion behind the sternomastoid. b) 3D-rendered image shows “shining pearl sign,” a CT hallmark of venous malformation.
Lymphatic malformations
Lymphatic malformations are lesions that involve lymphatic structures. Head and neck is a common site, constituting 74% of the lymphatic malformation lesions.[15] Posterior triangle and submandibular spaces are favorite sites in the neck. Lymphatic malformations do not spontaneously regress. Skeletal distortion, including mandibular overgrowth and osteolysis, constitutes part of the lymphatic malformation.[16] As with venous malformation, lymphatic malformation with low-flow lesion shows a micro- or macro-cystic character on imaging. MRI and ultrasonography are the imaging methods of choice. On contrast-enhanced MRI, lymphatic malformation does not enhance except in the region of septation, where the lymphatic component may be in the form of a mixed malformation.
Capillary malformation
Sturge-Weber syndrome (encephalotrigeminal angiomatosis) is a rare disorder in which capillary malformations are seen characteristically along the distribution of the ophthalmic division of the trigeminal nerve (V1). The syndrome involves leptomeningeal angiomatosis ipsilateral to the facial lesion. Patients present with seizures, transient neurologic deficits, glaucoma, and mental retardation[317] [Figure 23]. MR imaging offers a single comprehensive solution for assessment.

- 5-month-old female child demonstrates port-wine stain on the right side of the face and buphthalmos of the right eye, diagnosed as Sturge-Weber syndrome. a) Clinical photograph shows facial capillary malformation. b) Plain CT shows cerebral atrophy and right hemispherical subcortical calcification (black arrow). c and d) Contrast-enhanced T1FS axial MR images show leptomeningeal enhancement of both hemispheres (arrows) and enhancing enlarged glomus of choroid plexus (angled arrow) (c).
In a retrospective review of patients with extracranial AVMs of the head and neck, Kuhote et al., studied the natural history and effectiveness of treatment. AVMs were more frequent at the midface (69%) region. The rest were distributed over the upper third of the face (14%) and the lower third (17%). Specific regional sites were the cheek (31%), ear (16%), nose (11%), and forehead (10%). A large majority (59%) of the lesions were apparent at birth. Among the rest, 10% of patients reported an onset in childhood, another 10% in adolescence, and 21% in adulthood. Exacerbation of the lesions was experienced during puberty and pregnancy. Bony involvement largely involving the maxilla and mandible was observed. Schobinger's clinical staging had prognostic value: Stage I (quiescence) lesions were stable for long periods and Stage II (expansion) lesions were usually followed by pain, bleeding, and ulceration, leading to destruction (Stage III). Symptomatic patients inevitably progressed until the malformation was resected. For surgical management, resection margins (which are critical for a good outcome) were best determined intraoperatively and by Doppler assessment. Subtotal excision or proximal ligation frequently resulted in rapid progression of the AVM. The overall cure rate was 60%. In relation to Schobinger's staging, the cure rate was 75% for Stage I, 67% for Stage II, and 48% for Stage III malformations. Combined embolization–resection did not significantly alter the cure rate in small malformations.[18]
CONCLUSION
Head and neck region is a frequent site of hemangioma and VMs. Self-limiting lesions with a typical clinical appearance do not need imaging studies. When surgical or interventional therapy is contemplated, imaging plays a major role. Imaging methods have a capacity to provide physiological and anatomical information. Lesions needing conservative management are best evaluated with ultrasonography or MRI, which will provide most of the required information. VMs, in particular venous and mixed malformations in the facial regions with extensive involvement of cutaneous, subcutaneous, muscular, and osseous structures, need a complex imaging strategy requiring multiple modalities. Multiple detector CT (MDCT) imaging has a crucial role in the surgical management of extensive lesions and delineating osseous abnormalities.
ACKNOWLEDGMENTS
The authors would like to profoundly acknowledge the valuable contribution of all clinical colleagues who contributed immensely to the quality of this work. Additionally, the authors would also like to thank Philips Inc. for their Workstation Intellispace Portal, through which most of the image processing has been done for 3D evaluation.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/31/135179
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Haemangiomas and vascular malformations in infants and children: A classification based on endothelial characteristics. Plast Reconstr Surg. 1982;69:412-22.
- [Google Scholar]
- Vascular anomalies: What a radiologist needs to know. Pediatr Radiol. 2010;40:895-905.
- [Google Scholar]
- Hemangiomas and vascular malformations of the head and neck: A simplified approach. Neuroimaging Clin N Am. 2011;21:641-58.
- [Google Scholar]
- Venous malformations: Classification, development, diagnosis, and interventional radiologic management. Radiol Clin North Am. 2008;46:545-97.
- [Google Scholar]
- Childhood hemangiomas and vascular malformations: Angiographic differentiation. AJR Am J Roentgenol. 1983;141:483-8.
- [Google Scholar]
- Head and neck vascular anomalies in children. Int J Pediatr Otorhinolaryngol. 2009;73(Suppl 1):S71-6.
- [Google Scholar]
- Characterization of maxillofacial soft tissue vascular anomalies by ultrasound and color Doppler imaging: An adjuvant to computed tomography and magnetic resonance imaging. J Oral Maxillofac Surg. 2003;61:19-31.
- [Google Scholar]
- Vascular malformations and hemangiomas: A practical approach in a multidisciplinary clinic. AJR Am J Roentgenol. 2000;174:597-608.
- [Google Scholar]
- Combined sonographic and fluoroscopic guidance: A Modified technique for percutaneous sclerosis of low-flow vascular malformations. AJR Am J Roentgenol. 1999;173:655-7.
- [Google Scholar]
- Hemangiomas and vascular malformations of infancy and childhood. Pediatr Clin North Am. 1993;40:1177-200.
- [Google Scholar]
- Hemangiomas in the calvaria: Imaging findings. AJR Am J Roentgenol. 1995;164:683-7.
- [Google Scholar]
- An analysis of 2,480 space-occupying lesions of the orbit from 1976 to 2011. Ophthal Plast Reconstr Surg. 2013;29:79-86.
- [Google Scholar]
- Management of head and neck lymphatic malformations in children. J Otolaryngol. 1997;26:253-8.
- [Google Scholar]
- Magnetic resonance imaging of musculoskeletal soft tissue masses. Radiol Clin North Am. 2001;39:701-20.
- [Google Scholar]
- Management of lymphatic malformations. Curr Opin Otolaryngol Head Neck Surg. 2004;12:500-4.
- [Google Scholar]
- Arteriovenous malformations of the head and neck: Natural history and management. Plast Reconstr Surg. 1998;102:643-54.
- [Google Scholar]






