Translate this page into:
Imaging of Dual Ophthalmic Arteries: Identification of the Central Retinal Artery
Address for correspondence: Prof. Louise Louw, Department of Otorhinolaryngology, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa. E-mail: gnanll@ufs.ac.za
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Identification of the origin of the central retinal artery (CRA) is imperative in tailoring angiographic studies to resolve a given clinical problem. A case with dual ophthalmic arteries (OAs), characterized by different origins and distinct branching patterns, is documented for training purposes. Pre-clinical diagnosis of a 9-year-old child who presented with a sharp wire in the left-side eyeball was primarily corneal laceration. For imaging, a selected six-vessel angiographic study with the transfemoral approach was performed. Embolization was not required and the wire could be successfully removed. Right-side OA anatomy was normal, while left-side dual OAs with external carotid artery (ECA) and internal carotid artery (ICA) origins were seen. The case presented with a left-side meningo-ophthalmic artery (M-OA) anomaly via the ECA, marked by a middle meningeal artery (MMA) (origin: Maxillary artery; course: Through foramen spinosum) with normal branches (i.e. anterior and posterior branches), and an OA variant (course: Through superior orbital fissure) with a distinct orbital branching pattern. A smaller OA (origin: ICA; course: Through optic foramen) with a distinct ocular branching pattern presented with the central retinal artery (CRA). The presence of the dual OAs and the M-OA anomaly can be explained by disturbed evolutionary changes of the primitive OA and stapedial artery during development. The surgical interventionist must be aware of dual OAs and M-OA anomalies with branching pattern variations on retinal supply, because of dangerous extracranial–intracranial anastomotic connections. It is of clinical significance that the origin of the CRA from the ICA or ECA must be determined to avoid complications to the vision.
Keywords
Dual ophthalmic arteries
imaging
meningo-ophthalmic artery anomaly
source of central retinal artery
source of retinal artery
INTRODUCTION

Despite comprehensive studies on the anatomy and embryology of the ophthalmic artery (OA), variability of the OA had remained a teaching challenge in the past.[1] Today, imaging has paved the way for improved identification of OA variants, embryological explanations, and clinical interpretations. Upon studying the literature on dual OA origins [i.e. from the external carotid artery (ECA), internal carotid artery (ICA), or ECA and ICA], four studies over a period of three decades were encountered.[1234] Regarding OAs with ECA origin, three studies over a period of four decades reported on meningo-ophthalmic artery (M-OA) anomalies.[567] M-OA anomalies are marked by the anomalous origin of the OA from the stem of the middle meningeal artery (MMA), with or without MMA branch variations. Those of relevance to this paper are dual OAs with ECA and ICA origins, and identification of the source of the central retinal artery (CRA). Questions pertaining to these dual OA origins include why they exist and to what extent they are of clinical significance. These questions are addressed herein. Although the renowned work of Padget[8] on specimens more than 60 years ago is a tremendous contribution to human embryology, it does not explain all variations encountered in clinical practice. The concept of Lasjaunias and co-workers[9] on the development of the OA has been well accepted in the neuroradiological field for more than 40 years. Therefore, this concept was followed for the embryological explanations of the case under discussion. It is of clinical significance that the surgical interventionist must be aware of dangerous extracranial–intracranial anastomotic connections to avoid neurological complications, i.e. embolic stroke and cranial nerve palsies. Documentation on various OA origins and on variations in branching patterns for orbital supply is knowledge that is essential to the endovascular and microsurgical surgeon.
CASE REPORT
A 9-year-old girl was admitted to our academic hospital with trauma to the left-side eyeball. The pre-clinical diagnosis was corneal laceration with a sharp wire. Clinically, the patient presented with the following: A bruised eye and traumatic conjunctivitis, and intact vision, but third cranial nerve (CNIII) palsy with ptosis and a dilated pupil. Workup computed tomography (CT) scans and magnetic resonance imaging (MRI) revealed probable related trauma with unreliable history, namely left periorbital soft tissue swelling with retrobulbar stranding in keeping with a small amount of hemorrhage and inflammatory change [Figure 1], and also, incidental left cerebellar focal hemorrhage [Figure 2]. Pre-operative angiography with the transfemoral approach was performed. Access to the right common femoral artery was gained with a 22 G Jelco and a micro-puncture guide wire was advanced into the right common femoral artery. A 4 F pediatric sheath was advanced over the micro-puncture guide wire into the right common femoral artery. A Headhunter catheter was used and 50 ml of Jopamiron 300 was administered intra-arterially without any side effects. A six-vessel study, i.e. ECA, ICA, and vertebral artery (VA) on both sides, was selected to exclude an underlying cause for the hemorrhage observed. Injections appeared normal and no pseudo-aneurysms, arterio-venous fistulae, extravasation of contrast, or abnormal blush were demonstrated. No embolization was required and the sharp wire was successfully removed. Although cranial palsies can be a complication of embolization, the CNIII palsy was not procedure related. The patient was treated for eye infection and pain before being discharged from hospital with a remaining CNIII palsy.
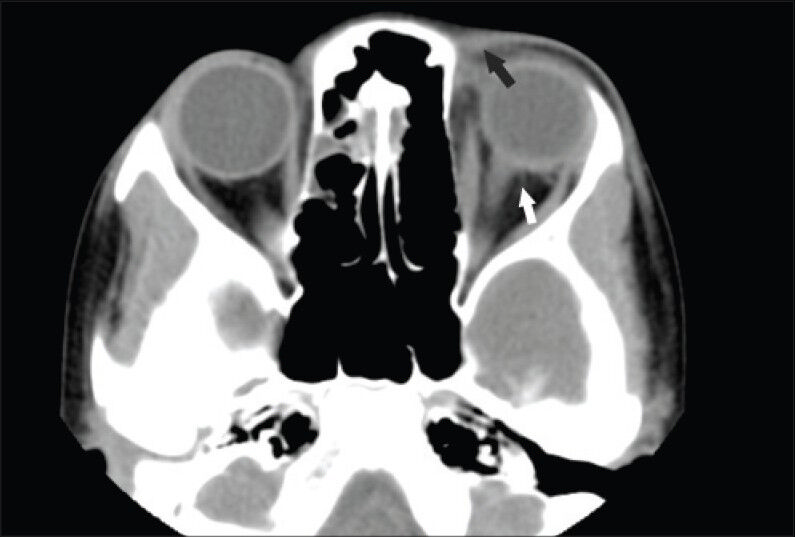
- Nine-year old girl presented with left-side orbital trauma and third cranial nerve palsy with ptosis later diagnosed with left-side dual ophthalmic arteries with external carotid artery (ECA) and internal carotid artery (ICA) origins. Computed tomography scan shows periorbital soft tissue swelling (black arrow) with retrobulbar stranding in keeping with a small amount of hemorrhage (white arrow).
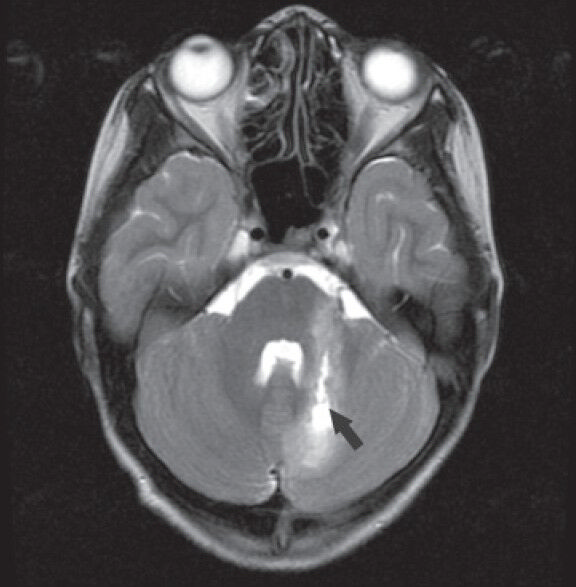
- Nine-year old girl presented with left-side orbital trauma and third cranial nerve palsy with ptosis later diagnosed with left-side dual ophthalmic arteries with external carotid artery (ECA) and internal carotid artery (ICA) origins. Magnetic resonance image shows left cerebellar focal hemorrhage (black arrow).
Conventional angiograms revealed normal right-side vascular anatomy, but on the left-side dual OAs were observed. An OA with normal origin from the ICA that coursed through the optic foramen presented with the CRA [Figure 3]. An OA variant with M-OA origin via the maxillary artery (MA) from the ECA coursed through the superior orbital fissure [Figure 4]. A lateral view angiogram illustrated both OAs, caused by overflow of the injected ECA [Figure 5]. The M-OA consisted of an MMA (course: Through the foramen spinosum) with normal branches (i.e. anterior and posterior branches) and an OA variant (course: Through superior orbital fissure) with a distinct branching pattern, namely supraorbital, lacrimal, and ethmoidal branches [Figures 4 and 5]. A smaller OA (origin: ICA; course: Through optic foramen) with a distinct branching pattern, namely terminal branches (i.e. supratrochlear and nasal branches) and ocular branches (i.e. ciliary and muscular branches), as well as the CRA (as witnessed by choroidal blush) were observed [Figure 3]. The left-side vertebral artery followed normal anatomy. Right-side angiograms showed normal anatomy of all the vessels.

- Nine-year old girl presented with left-side orbital trauma and third cranial nerve palsy with ptosis later diagnosed with left-side dual ophthalmic arteries with external carotid artery (ECA) and internal carotid artery (ICA) origins. Left lateral view angiogram of head shows central retinal artery (broken arrow) as a branch of one ophthalmic artery (black arrow) with normal internal carotid artery origin (white arrow).

- Nine-year old girl presented with left-side orbital trauma and third cranial nerve palsy with ptosis later diagnosed with left-side dual ophthalmic arteries with external carotid artery (ECA) and internal carotid artery (ICA) origins. Left antero-posterior view angiogram of head shows meningoophthalmic artery anomaly (black arrow) and ophthalmic artery variant (white arrow) with external carotid system origin.

- Nine-year old girl presented with left-side orbital trauma and third cranial nerve palsy with ptosis later diagnosed with left-side dual ophthalmic arteries with external carotid artery (ECA) and internal carotid artery (ICA) origins. Left lateral view angiogram of head shows ophthalmic artery variant (black arrow) and meningo-ophthalmic artery anomaly (white arrow). The ophthalmic artery with internal carotid artery origin (broken arrow) is visible because of injected external carotid artery overflow.
DISCUSSION
The OA may originate from the MMA as the main source, with a small contribution from the ICA, or from the MMA, without an orbital supply from the ICA. A previous study had reported an incidence of 2.4% in the USA where the OA had two trunks, namely a larger one arising from the MMA and a smaller one arising from the ICA.[2] A summary of dual OA origins reported in the literature[1234] is given [Table 1]. Dual OA origins and M-OA anomalies reported in the English literature appear to be relatively rare. The present case revealed dual OAs with a pattern variation on orbital supply not previously reported in the literature.

According to the concept of Lasjaunias and co-workers[9] the primitive ventral ophthalmic artery (PVOA) (origin: Anterior cerebral artery; course: Through the optic foramen) is the ocular artery and the primitive dorsal ophthalmic artery (PDOA) (origin: ICA at the siphon; course: Through the superior orbital fissure) is the orbital artery. At a point in time, the proximal parts of the PVOA and the PDOA regress, resulting in the formation of the adult OA. Discrepancies regarding the embryological origin of the CRA, i.e. whether the PVOA or PDOA is the source, exist. It seems that whatever the source, the OA that gives off the CRA passes through the optic foramen. Of further particular importance is that the stapedial system and its modification during fetal life were considered to be the pivot of numerous arterial anomalies, including M-OA anomalies with partial or sole orbital supply.[9] The distinct pattern of orbital supply witnessed in the current case was in contrast with the concept of Lasjaunias and co-workers.[9] According to their concept, the supraorbital artery branch of the stapedial artery (SA)] also passes through the superior orbital fissure and gives off branches to the orbit (i.e. ethmoidal, nasal, and lacrimal branches). However, in this case, nasal branches originated from the OA with ICA origin and passed via the optic foramen.
For this particular case, the embryological explanations are as follows. The PDOA (course: Through the superior orbital fissure) gave rise to a larger OA (variant), and the PVOA (course: Through the optic foramen) gave rise to a smaller OA (normal) from which the CRA originated. At a point in time during further embryological evolutionary changes, it is postulated that the proximal SA regressed; anastomotic connections between the MMA (ECA origin) and the primitive OA (ICA origin) as well as the orbital artery (stapedial origin) opened; the proximal trunk of the primitive OA disintegrated; and a smaller OA (ICA origin) remained as an anastomotic loop of the primitive OA. Hereby, an OA variant (M-OA origin) manifested and an OA with ICA origin remained [Figure 6]. It is noteworthy that the distal OA (including the CRA) may be supplied by the MMA as a remnant of the SA, and that the M-OA and MMA may be the sole or partial supply of the orbit, depending on the presence or absence of an OA with ICA origin.[10]
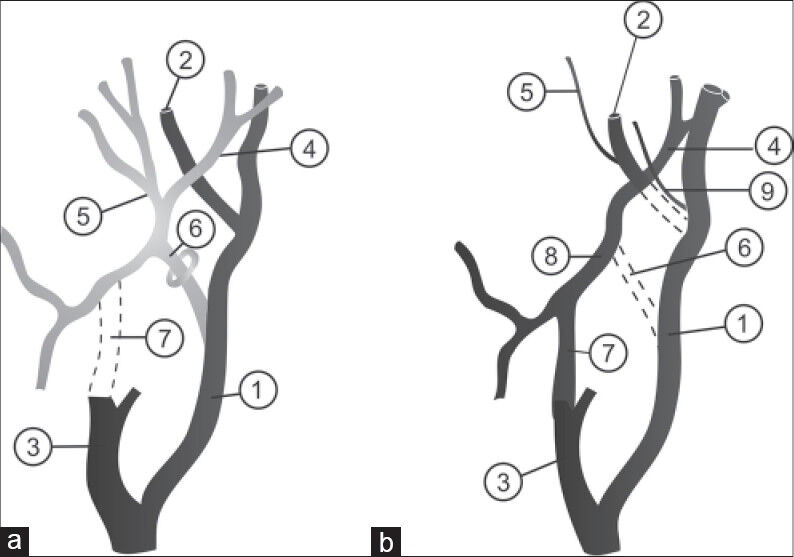
- Schematic presentations. a) Diagram shows normal development of the ophthalmic artery and b) diagram indicates probable development of the dual ophthalmic arteries and meningo-ophthalmic artery anomaly. (1: Internal carotid artery; 2: Ophthalmic artery; 3: External carotid artery; 4: Middle meningeal artery; 5: Orbital branch; 6: Stapedial artery; 7: Maxillary artery; 8: Meningoopthalmic artery; 9: Additional ophthalmic artery).
It is of clinical significance that proximal embolization of the MA and MMA can cause blindness, if an M-OA anomaly is the source of the CRA in cases where patients need embolization for surgical interventions. If OA embolization is required, research has revealed that a safety point distal to the second intraorbital segment exists and that visual damage can be restricted by embolization more distal to this point. The arterial ring that develops around the optic nerve, and eventually partially regresses, determines this safety point. However, for a given patient, the point of safety will be determined by the individual anatomy of the region.
CONCLUSION
Sound knowledge of orbital anatomy is indispensable for accurate diagnosis of orbit-related trauma. Pre-operative angiography required for decision-making on removal of the sharp wire in this patient excluded embolization procedures. Nevertheless, the M-OA anomaly that had been documented remains a valuable contribution to the literature and serves as a useful guideline for trainees.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/40/137833
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- An anomalous ophthalmic artery arising from the middle meningeal artery. Anat Anz. 1980;147:473-6.
- [Google Scholar]
- Internal carotid origin of double ophthalmic arteries. Neuroradiology. 1990;32:508-10.
- [Google Scholar]
- Dural caroticocavernous fistula with both ophthalmic arteries arising from middle meningeal arteries. Neuroradiology. 1996;38:806-8.
- [Google Scholar]
- Anomalous origin of the ophthalmic artery in a patient with amaurosis fugax. Arch Neurol. 1981;38:315-7.
- [Google Scholar]
- Impact of anomalous origin of the ophthalmic artery from the middle meningeal artery on selection of surgical approach to skull base meningioma. Surg Neurol. 2007;68:568-72.
- [Google Scholar]
- The development of the cranial arteries in the human embryo. Contrib Embryol. 1948;32:205-62.
- [Google Scholar]
- Maxillary artery blood supply to the orbit: Normal and pathological aspects. Neuroradiology. 1975;9:87-97.
- [Google Scholar]
- Selective thrombolysis performed through meningo-ophthalmic artery in central retinal artery occlusion. J Clin Neurosci. 2012;19:462-4.
- [Google Scholar]






