Translate this page into:
Castleman's Disease Presenting as Localized Abdominal Mass and Paraneoplastic Pemphigus
Address for correspondence: Dr. Santosh Kumar, Department of Urology, Postgraduate Institute of Medical Education and Research, Chandigarh, India. E-mail: santoshsp1967jaimatadi@yahoo.co.in
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Castleman's disease is a rare, benign lymphoproliferative disorder of unknown origin. Paraneoplastic pemphigus is a common association which presents as oral mucosal ulcerations. Abdominal and retroperitoneal Castleman's disease present either as a localized disease or as a systemic disease. We hereby present a 15-year-old male patient with oral mucosal lesions with localized vague right lower abdominal mass who was diagnosed to have Castleman's disease with paraneoplastic pemphigus which was surgically excised.
Keywords
Abdominal mass
Castleman's disease
paraneoplastic
pemphigus

INTRODUCTION
Castleman's disease is a rare autoimmune disorder mimicking lymphoma with varied clinical presentation and also known as angiofollicular lymph node hyperplasia, it involves hyperactivation of the immune system with multiple organ system dysfunction. It may be localized to a single lymph node (unicentric) or occur systemically (multicentric). Pemphigus as paraneoplastic syndrome is a common association and presents as painful oral ulceration with polymorphic skin eruptions.
CASE REPORT
A 15-year-old male patient presented to our institute with complaints of blisters or oral lesions of 3 months duration along with vague abdominal pain. The oral ulcers progressed to lesions with encrustation and bled on touch. Patient complained of dull aching pain in the right lower abdomen without any bowel complaints. Patient was admitted in the Dermatology Department for further diagnosis and treatment. Detailed history revealed significant weight loss in the last 3 months, probably due to decreased oral intake secondary to oral ulcers. There was no significant family history, allergic reaction, or any history of drug abuse. General examination revealed oral lesion [Figure 1], and vague abdomen lump in the right lower abdomen. Multiple subcentimetric lymph nodes were palpable in the neck and bilateral inguinal region which were nontender and mobile. Routine hematology and biochemistry test were normal. Histological examination of biopsy tissue from oral lesion revealed hyperplastic squamous epithelium with mixed dense inflammation composed of neutrophils, lymphoplasmacytic cells, and capillaries suggestive of pemphigus vulgaris. Patient was started on steroid and responded well. Ultrasound abdomen reported 6 cm × 5 cm solid mass arising from the pelvis. Contrast-enhanced magnetic resonance (MR) imaging revealed well-defined, heterogenous soft tissue mass lesion 6.2 cm × 5.4 cm seen in the extraperitoneal region of right hemipelvis which was slightly hyperintense on T1W1 and hyperintense on T2W1 with multiple fine internal septations [Figure 2]. The mass also showed homogenous, intense postcontrast enhancement. Further, positron emission tomography-computed tomography (CT) showed faintly fluorodeoxyglucose avid heterogenous mass arising from the right hemipelvis [Figure 3]. Biopsy tissue from the pelvic mass revealed atretic lymphoid follicles with proliferation of spindle-shaped cells and moderate amount of cytoplasm. The patient was taken up for surgery after optimization. Midline infraumbilical incision showed the mass to be extraperitoneally close to the right common iliac vessels. Mass was circumferentially mobilized and removed preserving major iliac vessels [Figure 4]. Patient was discharged on day 4 with uneventful postoperative course. Final histopathology confirmed Castleman's disease [Figure 5]. Patient is presently on tablet prednisolone 20 mg and oral lesions are healing [Figure 6].
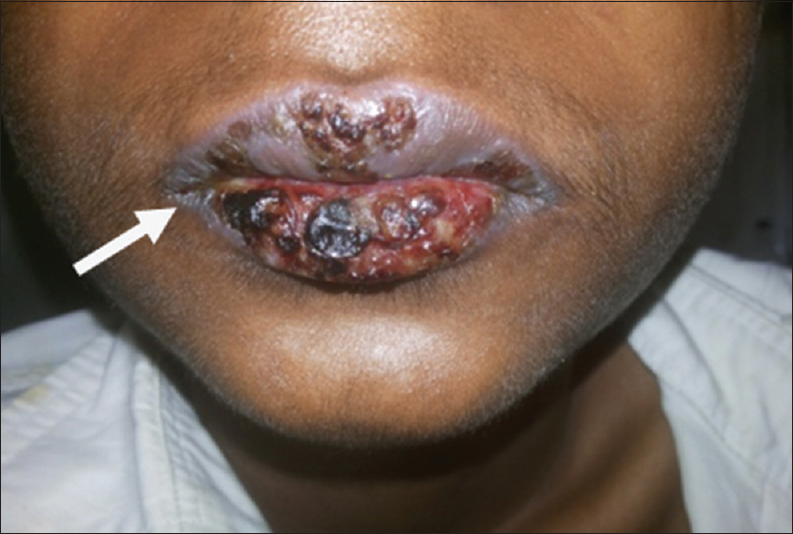
- 15-year-old male presented with oral mucosal lesions with encrustation and blisters and abdominal pain diagnosed as due to Castleman's disease. Photograph shows the oral lesions.
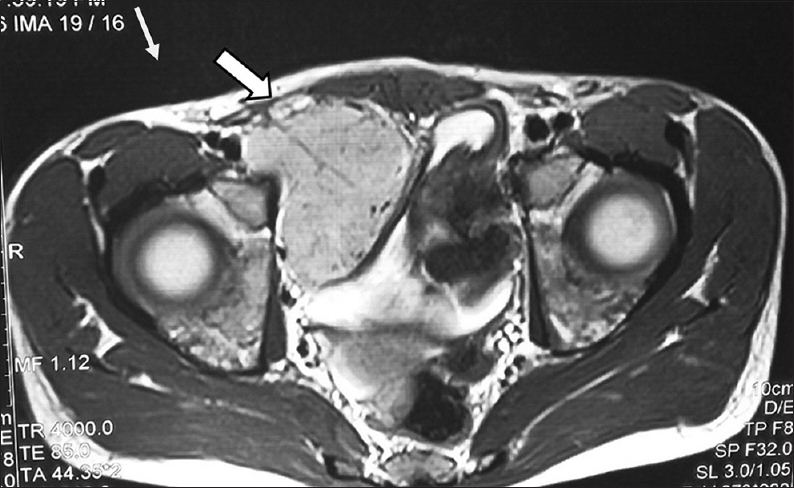
- 15-year-old male child presented with oral ulcers and abdominal pain diagnosed as due to Castleman's disease. Magnetic resonance imaging of the abdomen, axial image shows homogeneous mass 6 cm × 5 cm (arrow) arising from right hemipelvis.
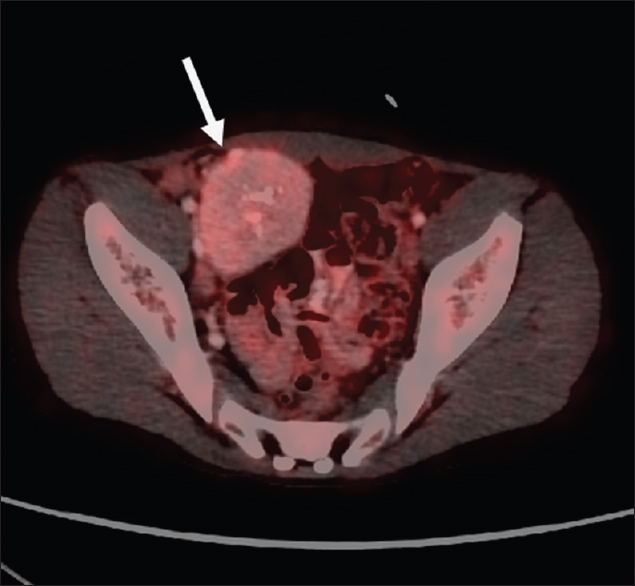
- A 15-year-old male child presented with oral ulcers and abdominal pain diagnosed as Castleman's disease with fluorodeoxyglucose positron emission tomography axial image showing faintly fluorodeoxyglucose avid soft tissue mass in the right hemipelvis (arrow).
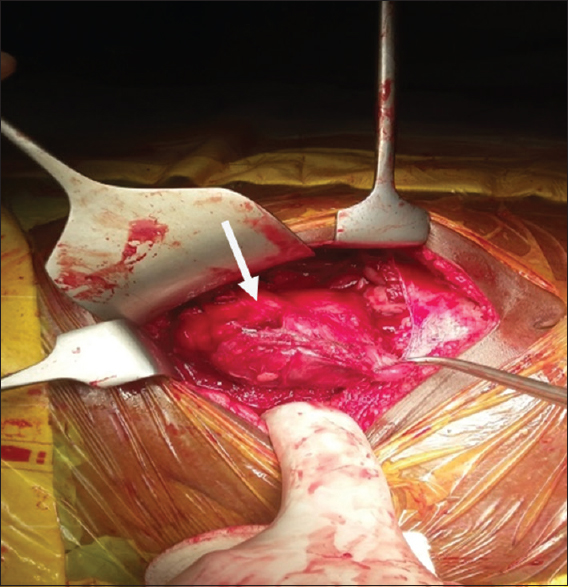
- 15-year-old male child presented with oral ulcers and abdominal pain diagnosed as due to Castleman's disease., Intraoperative image shows the mass during dissection.
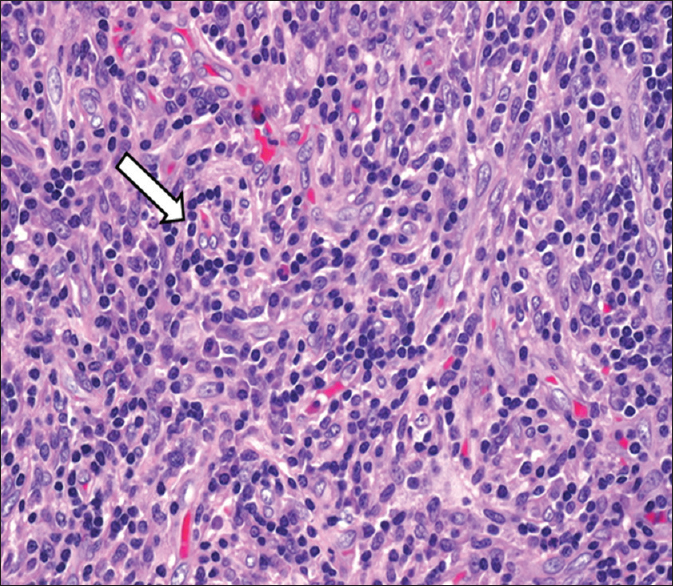
- 15-year-old male child presented with oral ulcers and abdominal pain diagnosed as due to Castleman's disease. Hematoxylin and eosin stained biopsy tissue (×10 magnification) of the oral mucosal lesion shows dense lymphocytic infiltrates (arrow) with capillaries around them. Direct immunofluorescence revealed intraepidermal intercellular staining with immunoglobulin G (not uploaded).
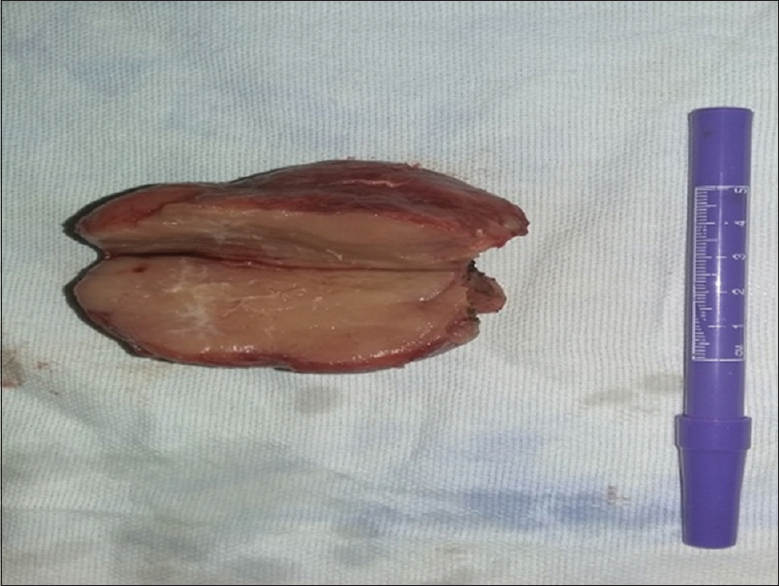
- 15-year-old male child presented with oral ulcers and abdominal pain diagnosed as due to Castleman's disease. Postsurgical resection of abdominal mass cut section of the mass lesion shows healing of the oral lesions.
DISCUSSION
Castleman's disease is a rare lymphoid disorder. It commonly involves the mediastinum and presentation as a pelvic mass is rare. Retroperitoneum and base of the bladder are other sites involved.[12] It may present as localized or systemic disease and is often characterized histologically into four types; hyaline vascular variant, plasma cell variant, human herpesvirus 8 (HHV-8) associated variant, and multicentric variant.[3] The enlarged lymph nodal mass may be an incidental finding. In our index case, patient presented with vague abdomen pain and was found to have pelvic mass located extraperitoneally. Myasthenia gravis and paraneoplastic pemphigus are a well-known association of Castleman's disease. Other common associations are non-Hodgkin lymphoma, polyneuropathy, organomegaly, and skin changes syndrome. Anhalt first described paraneoplastic pemphigus as an autoimmune disease associated with lymphoproliferative disorder.[456] The clinical manifestation is varied and involve skin and mucosa. The lesion on mucosa are blisters, erosions or ulcers, or erosive lichen planus like lesions.[7] Our patient presented with ulcerated blisters with encrustations in the oral mucosa which bled on touch. Radiologically, on CT, the hyaline vascular variant appear as solitary enlarged lymph nodal mass mostly in the thorax as mediastinal mass which on MR classically shows heterogenous hyperintensity on T2 and T1. Plasma cell variant typically shows fewer enhancement on contrast administration as compared to hyaline vascular variant and often it is multicentric with more predilection for pelvic and abdominal involvement. Imaging findings of HHV-8 variant are often indistinguishable from the plasma cell variant, and it also shows mild postcontrast enhancement, concomitant immunosuppressant is often the distinguishing point.[8] Our case also showed T2 and T1 hyperintense localized pelvic mass with postcontrast enhancement. Localized lymph nodal mass is treated by surgical excision. Even after complete resection of Castleman's disease, the autoimmunity remains active for up to 2 years, and disease may progress.[7] Nikolskaia et al. studied 28 patients presenting with stomatitis and lichenoid skin eruptions diagnosed to have Castleman's disease. On follow-up, 22 patients died over a period of 2 years following respiratory failure. Interleukin 6 (IL-6) has been responsible for progressive autoimmune damage and treatment with anti-IL-6 may show some improvement.[2]
CONCLUSION
Castleman's disease may present as localized abdominal mass in association with paraneoplastic pemphigus. A nonhealing ulcer should raise a strong suspicion and biopsy is usually confirmatory. Radiologic investigation is essential to rule out systemic disease. Surgical excision is a treatment for localized disease. Chemotherapeutic and immunomodulatory agents are used in adjuvant setting.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
The authors thank the patient for allowing them to publish the case report and use the images taken during his stay in hospital.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/6/177552
REFERENCES
- Extra thoracic Castleman's disease associated with myasthenia gravis. Indian J Thorac Cardiovasc Surg. 2010;26:275-6.
- [Google Scholar]
- Paraneoplastic pemphigus in association with Castleman's disease. Br J Dermatol. 2003;149:1143-51.
- [Google Scholar]
- Paraneoplastic pemphigus. An autoimmune mucocutaneous disease associated with neoplasia. N Engl J Med. 1990;323:1729-35.
- [Google Scholar]
- Lichenoid paraneoplastic pemphigus in the absence of detectable antibodies. J Am Acad Dermatol. 2007;56:153-9.
- [Google Scholar]
- Outcome and late complications of radiotherapy in patients with unicentric Castleman disease. Acta Oncol. 2006;45:1126-31.
- [Google Scholar]






