Translate this page into:
An Uncommon Case of Neurofibromatosis Type 2: A Tribute to the Intracranial Calcifications
Address for correspondence: Dr. Anıl Özgür, Department of Radiology, Mersin University Faculty of Medicine, Ihsaniye Mahallesi Zeytinlibahçe Caddesi, 4935 Sok. 33070 Mersin, Turkey. E-mail: anilozgur@yahoo.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Neurofibromatosis Type 2 (NF2) is a genetic disorder associated with schwannomas, meningiomas, and ependymomas. Intracranial calcifications, either tumoral or non-tumoral, are relatively lesser known features of NF2. Here, we present a case of NF2, in which the diagnosis was suspected due to the presence of choroid plexus and subependymal calcifications, although no obvious schwannoma or meningioma was detected initially on standard computed tomography or magnetic resonance imaging. This case highlights the importance of further evaluation with appropriate imaging techniques.
Keywords
Computed tomography
intracranial calcification
neurofibromatosis type 2
INTRODUCTION

Neurofibromatosis Type 2 (NF2) is an autosomal dominant disease characterized by schwannomas, meningiomas, and ependymomas.[12] Bilateral vestibular schwannomas, occurring in the majority of adult patients with NF2, are diagnostic of NF2. They usually present as an enhancing mass extending into the cerebellopontine cistern from the internal acoustic canal. However, in some cases, these tumors may be absent or too small to be detected with standard computed tomography (CT) and/or magnetic resonance imaging (MRI). In such cases, presence of the non-tumoral calcifications with or without tumoral calcifications may suggest the possible diagnosis of NF2.
In this paper, we present a case of NF2, who was diagnosed on the basis of intracranial calcifications.
CASE REPORT
A 34-year-old man with a complaint of mild hearing loss in the left ear was referred to our department for temporal bone CT. His family history was unremarkable. Temporal bone CT [Figure 1a–d], showed subependymal calcification at the foramen of Monro [Figure 1a], choroid plexus calcifications within the lateral ventricles [Figure 1b], a small calcified lesion adjacent to the right cavernous sinus [Figure 1c], and a partially calcified midline mass adjacent to the fourth ventricle [Figure 1d]. These findings were found highly suspicious for NF2, although no prominent mass was seen within the cerebellopontine cisterns or internal acoustic canals. Thus, further imaging with MRI was recommended. Cerebral MRI with intravenous contrast administration showed no major findings except an enhancing mass at the posterior aspect of the fourth ventricle Figure 2. Inner ear MRI with the aid of fast imaging employing steady state acquisition (FIESTA) [Figure 3a] and contrast enhanced T1-weighted sequences with thin slices [Figure 3b] revealed bilateral small intracanalicular lesions consistent with schwannomas. In addition, the small calcified lesion adjacent to the right cavernous sinus showed contrast enhancement indicating a meningioma. Spinal MRI [Figure 4a and b], which was performed to evaluate possible additional tumors, showed a mass causing focal cord expansion at C5-C6 level [Figure 4a] and multiple enhancing small intramedullary lesions [Figure 4b] consistent with ependymomas in the cervical cord. All these findings supported the final diagnosis of NF2. Posterior fossa mass was operated and diagnosed as WHO Grade 2 ependymoma histopathologically [Figure 5].
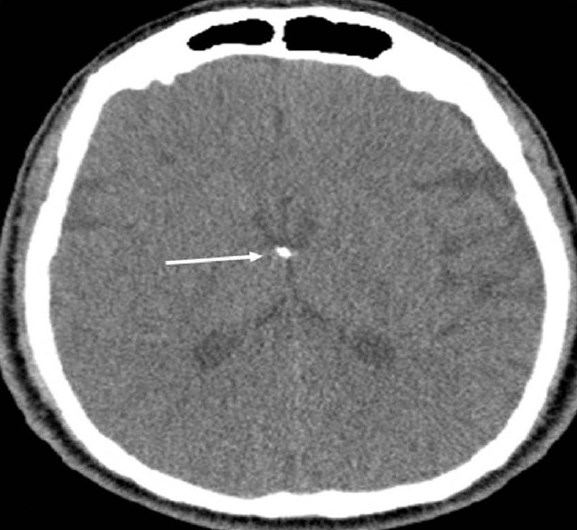
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Axial computed tomography (parenchymal window) of the temporal bone shows subependymal calcification (arrow) at the foramen of Monro
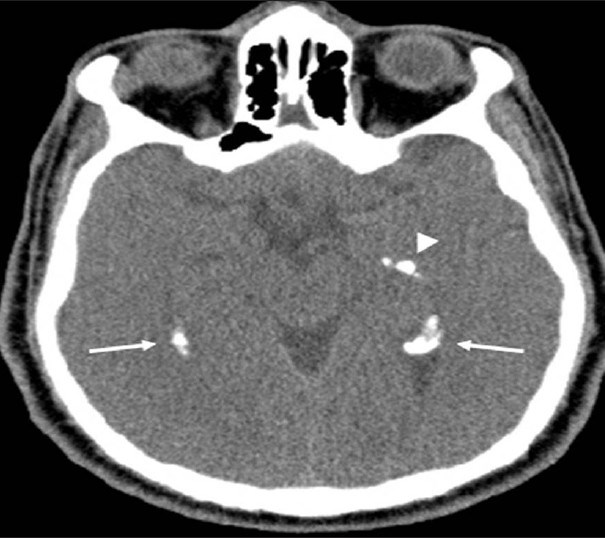
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Axial computed tomography (parenchymal window) of the temporal bone shows extensive choroid plexus calcifications within both atria (arrows) extending into the left temporal horn (arrowhead)
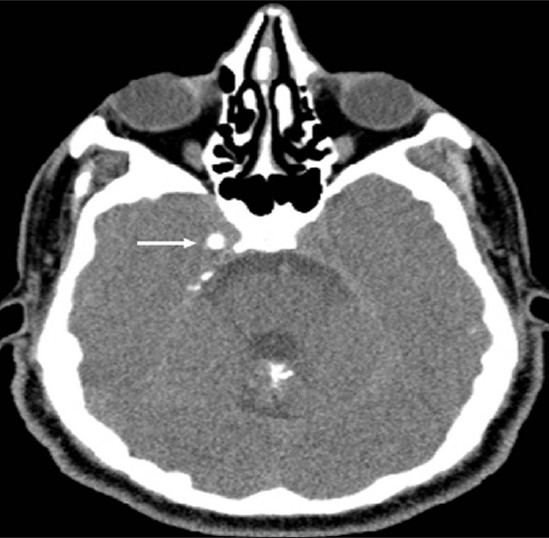
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Axial computed tomography (parenchymal window) of the temporal bone reveals a small calcified lesion (arrow) adjacent to the right cavernous sinus
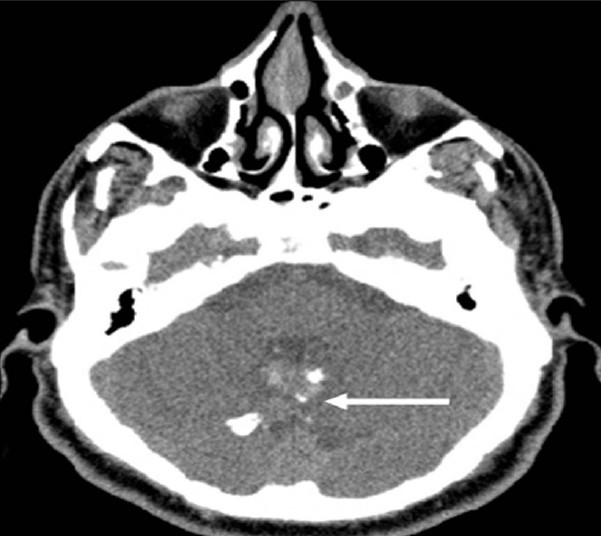
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Axial computed tomography (parenchymal window) of the temporal bone shows a midline mass (arrow) with coarse calcifications at the posterior aspect of the fourth ventricle. Note the normal appearance of bilateral pontocerebellar cisterns
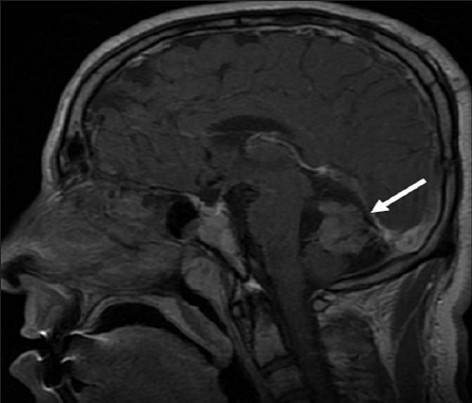
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Cerebral magnetic resonance. Sagittal postcontrast T1-weighted image shows an enhancing midline mass (arrow) within the posterior fossa.
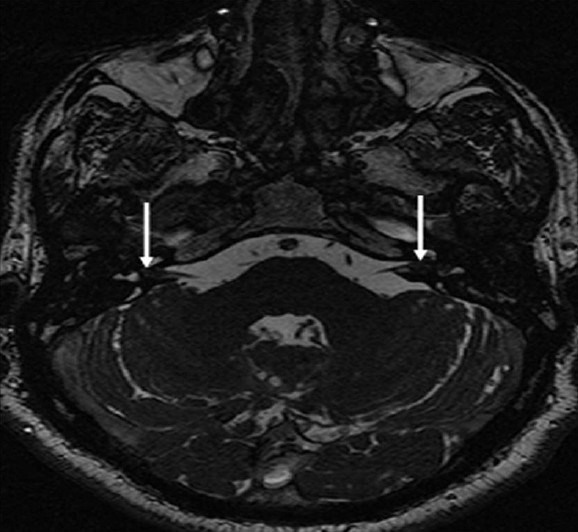
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Inner ear magnetic resonance imaging. Axial fast imaging employing steady state acquisition image demonstrates small intracanalicular lesions (arrows) obliterating the fundus of the both internal acoustic canals
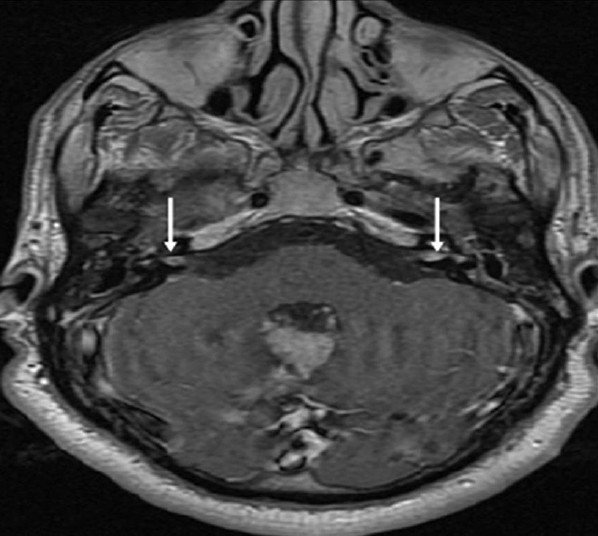
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Inner ear magnetic resonance axial post-contrast T1-weighted image shows enhancement within the small tumors (arrows) of the both internal acoustic canals
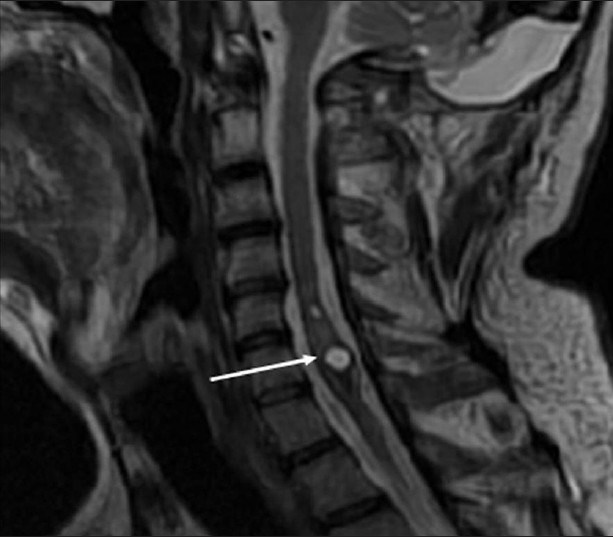
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Cervical spinal magnetic resonance sagittal T2-weighted image shows a mass (arrow) causing cord expansion at C5-C6 level with cystic component and hemosiderin at caudal margin, which are typical findings of a cord ependymoma
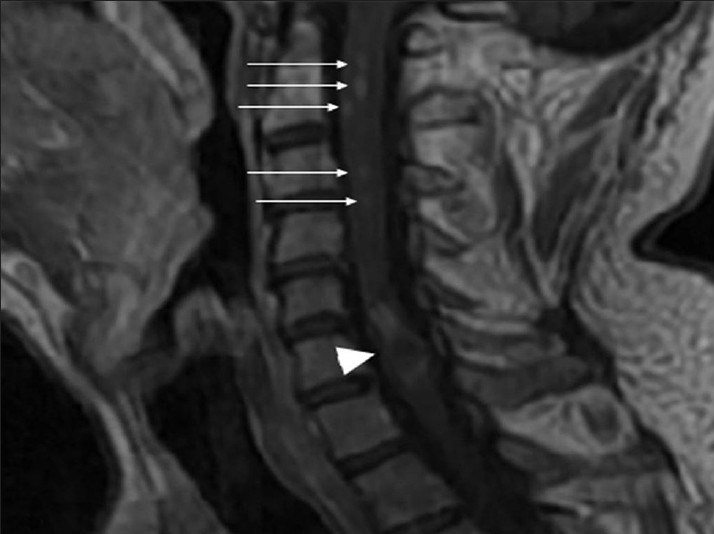
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Cervical spinal magnetic resonance sagittal postcontrast T1-weighted image shows enhancement within the mass at C5-C6 level (arrowhead) and additional small intramedullary tumors with the appearance of “string of pearls” (arrows)
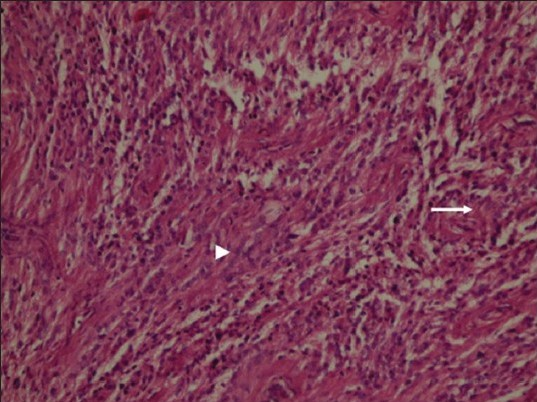
- Neurofibromatosis Type 2. A 34-year-old man with mild hearing loss in the left ear. Photomicrograph of hematoxylin and eosin stained tissue at ×10 shows neoplastic ependymal cells with nuclear uniformity (arrowhead) and fibrillary cytoplasmic processes (arrow) remain attached to blood vessels, ×10)
DISCUSSION
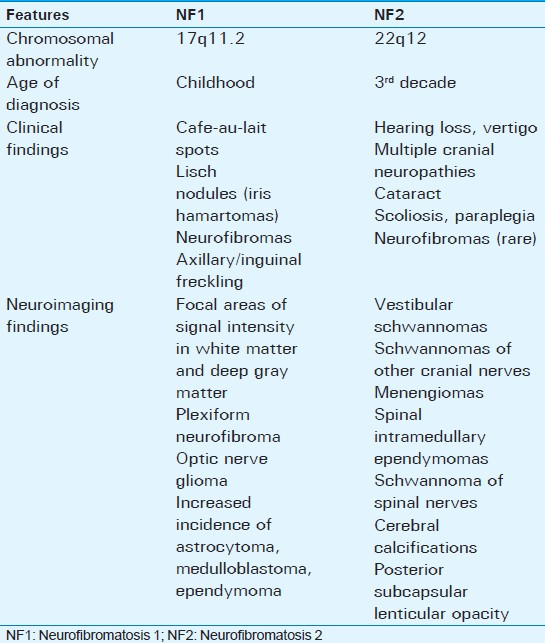
NF2 is a hereditary syndrome characterized by the nervous system tumors including schwannoma, meningioma, and ependymoma. Although sharing the same name, NF1 and NF2 are distinct disorders arising from separate chromosomes with different clinical and imaging findings [Table 1]. A mutation causing the inactivation of a tumor suppressor gene on the long arm of chromosome 17 is responsible for NF1. However, NF2 is caused by a defect in the gene located on chromosome 22, which normally encodes a tumor suppressor protein called “Merlin.” Neurofibromas, which are hallmarks of NF1, are seen in a minority of patients with NF2. Therefore, the term “multiple inherited schwannomas, menengiomas, ependymomas,” which enables a better definition of the disease is now being widely used.[12]
Diagnosis of NF2 depends mainly on the presence of vestibular schwannoma and/or family history.[12] Bilateral vestibular schwannomas or unilateral vestibular schwannom (age < 30 years) plus a first-degree relative with NF2 are the criteria for a definite diagnosis of NF2. In the absence of vestibular schwannoma, a first-degree relative with NF2 and any two of the lesions: Meningioma, glioma, schwannoma or juvenile posterior subcapsular lenticular opacity is necessary for the definite diagnosis. In the absence of family history, presumptive diagnosis of NF2 is possible if unilateral vestibular schwannoma (age < 30 years) with characteristic disease features are found. Despite all these criteria, diagnosis may be problematic in patients who do not have bilateral vestibular schwannomas at the initial examination.[2]
Radiological evaluation plays a major role in the diagnosis of NF2. Diagnosis is relatively easy when typical appearance of bilateral vestibular schwannomas bulging into cerebellopontine cisterns from internal acoustic canals is seen. However, some patients, especially children and young adults, can present with schwannomas of other nerves, meningiomas, ependymomas, and ocular abnormalities. Therefore, presentation of a young patient with single/multiple schwannomas or multiple extra-axial brain or spine tumors should prompt screening for NF2.[1] In such cases, initial radiological examination may be a standard CT or MRI in which small intracanalicular tumors may be easily overlooked. For this reason, axial and coronal thin slice contrast enhanced T1-weighted sequences should be performed to detect small schwannommas of vestibular or other cranial nerves. Furthermore, steady state free precession sequences, such as FIESTA or constructive interference steady state, which allow detailed evaluation of cranial nerves and precise detection of small tumors should be included in the imaging protocols.
Arachnoid cyst, epidermoid cyst, meningioma, metastasis, sarcoidosis, menengitis, and aneursym should also be considered in the differential diagnosis of the cerebellopontine angle (CPA) lesions. Arachnoid cyst follows cerebrospinal fluid (CSF) signal on all magnetic resonance sequences and shows no contrast enhancement. Epidermoid cyst may mimic cystic schwannoma; however, characteristic features such as diffusion restriction and the absence of contrast enhancement suggest the diagnosis of epidermoid cyst. Meningioma is typically a broad dural-based enhancing lesion with dural tail. Hyperostosis in the adjacent bone may be a helpful finding in the diagnosis of a meningioma. Metastasis and sarcoidosis may both cause dural-based lesions and/or leptomeningeal involvement in the CPA. Differential diagnosis may be relatively challenging if bilateral lesions are present. Metastasis is a more likely diagnosis in an older adult with known malignancy and multifocal involvement. Multifocal lesions associated with infundibular stalk involvement may suggest sarcoidosis. Bacterial, fungal or tuberculous menengitis may also cause leptomeningeal involvement in the CPA. Clinical information and CSF analysis may have a key role in the diagnosis of menengitis. Aneursym may present as a mass with heterogeneous signal intensity in the CPA. Pulsation artifact in the phase-encoding direction is an important clue for the diagnosis.
Intracranial calcifications are uncommon findings of NF2. They may be tumoral or non-tumoral in origin. Before the era of MRI, several reports have been published regarding non-tumoral calcifications on CT in patients with NF2. A report revealed three patients with subependymal calcifications.[3] In another study, benign calcifications were detected within choroid plexus, cerebellum, and cerebral parenchyma in 7 of 11 patients with NF2.[4] Unilateral or bilateral prominent vestibular schwannomas were found in all cases of these two reports. These reports indicate that the presence of non-tumoral calcifications is a notable feature of NF2. However, diagnostic significance of these calcifications seems to be underestimated with the increasing utilization of MRI throughout the world. Subependymal calcifications are almost always associated with tuberous sclerosis; however, similar calcifications may be seen in NF2. Calcifications of choroid plexus are rarely seen in children and are common after the age of 40 years.[5] Therefore, extensive choroid plexus calcifications in children and young adults may suggest the possibility of NF2. Cortical calcifications alone may have less diagnostic importance, because many other pathologies including congenital, infectious, metabolic, vascular, tumoral, and sequel lesions can cause similar calcifications. However, it is important to differentiate cortical calcifications and partially calcified tumors. Postmortem studies have shown glial microhamartomas in the cerebral cortex and basal ganglia of patients with NF2.[6] Non-tumoral calcifications may be related to these hamartomatous lesions, which do not seem to be preneoplastic.
Tumoral calcifications can also be seen in patients with NF2. Schwannomas rarely calcify.[7] However, calcifications can occur in nearly one-fourth of meningiomas.[7] Ependymomas are neoplasms, which are commonly associated with calcifications. In patients with NF2, intracranial ependymomas are not common except for the ones located in the brainstem. Furthermore, like in our case, a calcified ependymoma within the fourth ventricle in a NF2 patient is a very unusual finding. To our knowledge, such ependymoma has not been reported in the English literature. Unlike intracranial ependymomas, the presence of multiple ependymomas in the spinal cord is a well-known feature of NF2. A recent study regarding NF2 showed that ependymomas had a predilection for cervical cord or cervicomedullary junction, while brain involvement was not reported in that study.[8]
In our case, diagnosis of NF2 was suspected due to the presence of choroid plexus and subependymal calcifications on CT, although standard CT and MRI did not reveal any prominent mass within cerebellopontine cisterns or internal acoustic canals. With the aid of dedicated further investigations; bilateral small intracanalicular lesions, presumed calcified meningioma, histopathologically proven fourth ventricle ependymoma, and the intramedullary lesions consistent with ependymoma in the cervical spinal cord were detected all of which finally confirmed diagnosis of NF2.
CONCLUSION
Non-tumoral calcifications with or without tumoral calcifications may suggest the possibility of NF2, especially in those without obvious lesions within the cerebellopontine cistern. Choroid plexus and subependymal calcifications may be the only apparent signs of NF2 when CT is the initial radiological examination. Therefore, further evaluation with proper imaging techniques for NF2 should be done in the presence of these calcifications regardless of whether or not NF2-related tumors are detected initially.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2013/3/1/21/112802
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Neurofibromatosis type 2 imaging, 2011. Available from: http://www.emedicine.medscape.com/article/342667-overview
- [Google Scholar]
- Intracranial calcified deposits in neurofibromatosis. J Neurol Neurosurg Psychiatry. 1986;49:1317-20.
- [Google Scholar]
- Intracranial calcified deposits in neurofibromatosis type 2. A CT study of 11 cases. Neuroradiology. 1990;32:33-7.
- [Google Scholar]
- Distribution and immunoreactivity of cerebral micro-hamartomas in bilateral acoustic neurofibromatosis (neurofibromatosis 2) Acta Neuropathol. 1989;79:137-43.
- [Google Scholar]
- Spinal ependymomas in neurofibromatosis Type 2: A retrospective analysis of 55 patients. J Neurosurg Spine. 2011;14:543-7.
- [Google Scholar]






