Translate this page into:
A case report of mesenteric involvement in neurofibromatosis type 1

*Corresponding author: Nguyen Van Sang, Department of Radiology, Thai Nguyen University of Medicine and Pharmacy, Thai Nguyen, Vietnam. dr.nguyensang@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sang NV, Ninh TP, Thanh DT, Thinh NC. A case report of mesenteric involvement in neurofibromatosis type 1. J Clin Imaging Sci 2022;12:43.
Abstract
Mutations in the Neurofibromatosis Type 1 (NF-1) gene, which is located on chromosome 17q11.2, are the cause of NF-1, an autosomal dominant hereditary condition in which tumors of the nerve system develop. Neurological, skeletal, and cutaneous abnormalities are symptoms of the condition. Of all gastrointestinal tract lesions, mesentery lesions are the least common. There are many gastrointestinal problems that can accompany mesenteric neurofibromas, or they may not. We describe a case of a 5-year-old kid with mesenteric neurofibromatosis, which results in bowel obstruction and abdominal discomfort. On a CT scan, the mesenteric vasculature was completely encircled by homogenous soft tissue lesions without any vessel wall invasion. Diffuse mesentery lesions were discovered during surgery, although they could not be fully removed.
Keywords
Mesentery
Neurofibromatosis type I
Neurofibroma
Imagings
INTRODUCTION
The most prevalent multiorgan dominant genetic condition, Neurofibromatosis Type I (NF-1), is characterized by the development of nervous system-related tumors and is brought on by mutations in the NF-1 gene, which is located on chromosome 17q11.2.[1-3] Nerve, bone, and skin abnormalities are among the disease’s symptoms.[1,4] From the first accounts of NF-1 patients in the 15th century, to the most notable medical studies in the 18th century, to the discovery of molecular genetic studies in the late 20th century, doctors discovered that the disease had a variety of manifestations with the unusual and peculiar physical appearances.[1]
Gastrointestinal (GI) involvement in neurofibromatosis occurs in 10-25% of cases, but GI manifestations of NF-1 are often nonspecific, leading to misdiagnosis in clinical practice.[5] Mesentery involvement in neurofibromatosis is rare in adults and children and has been described in clinical case reports.[4] Most of the lesions are benign, just a few cases can progress to malignancy, so early detection is necessary to increase the effectiveness of treatment. The clinical presentation of mesenteric neurofibromatosis may be asymptomatic or present with various GI symptoms, including abdominal pain, weight loss, diarrhea, ulceration, bowel obstruction, intussusception, or volvulus. Due to nonspecific symptoms, it is easily confusing with a digestive disorder.[6]
In this article, we report a case of mesenteric neurofibromatosis in a 5-year-old boy which caused abdomen pain and bowel obstruction. The patient underwent an abdominal CT scanner and the diagnosis of neurofibromatosis was made after mesenteric biopsy for pathology due to the inability of lesion resection. Our case adds to the literature regarding this rare disease.
CASE REPORT
A 5-year-old male patient was admitted to the National Children’s Hospital with the complaint of abdominal pain and distention. Approximately 3 years ago, he presented with unexplained abdominal distention and intermittent abdominal pain accompanied by a sign of bowel loops floating in pain without vomiting. He had alternating episodes of constipation and diarrhea. He has been diagnosed with many different diseases such as prolonged digestive disorder, colitis, or chronic intestinal pseudo-obstruction in several hospitals. Last year, he underwent gastroscopy and colonoscopy with images of total colitis, congestive gastritis with H. Pylori positive, and pyloric regurgitation. The patient then was taken internal medicine with inconsiderable improvement in symptoms. This time, the patient was admitted to the hospital because of more severe abdominal pain and distention. A clinical examination showed his height to be 116 cm (+1SD), and weight 17 kg (-1SD). There was a café au lait spot found on the neck, about 2 × 1.8 cm in size. The abdomen was much distended with the sign of bowel loops floating, no response of the abdominal wall, and no peritoneal reaction. On anorectal examination, the rectal balloon was empty.
His abdominal X-ray image showed a lot of air-fluid levels, >2.5 cm in width [Figure 1]. On the abdominal ultrasound, the bowel was dilated and distended with air. On the enhanced CT images, there were diffuse lesions of small bowel mesentery and transverse mesocolon, encasing the mesenteric vessels without invasion or causing vessel stenosis/obstruction [Figure 2]. In addition, there were similar lesions surrounding the rectum. The lesions decreased in density on noncontrast CT images (~25HU), and slightly enhanced on portal venous phase CT images (~40HU). The small bowel loops were dilated with the largest transverse diameter of 45 mm. Some loops had wall thickening about 11 mm. There was free abdominal fluid accumulating predominantly in the pelvis cavity.
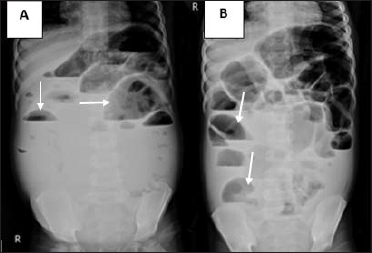
- A 5-year-old male patient with NF-1 presented with abdominal pain and distention. (A) Abdominal X-rays on the first day admitted to the hospital showed a lot of air-fluid levels (white arrow) suggesting a small bowel obstruction. (B) There was an elimination in the quantity of air-fluid levels (white arrow) after one week compared to the first day. No subdiaphragmatic air crescent was seen.
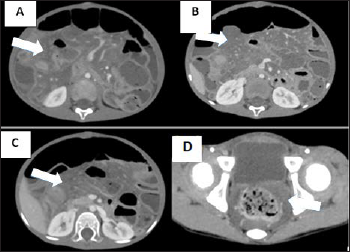
- A 5-year-old male patient with NF-1 presented with abdominal pain and distention. Portal venous phase CT images: (A, B, C) Diffuse lesions of small bowel mesentery (white arrow), slightly enhancing and encasing the mesenteric vessels without invasion or causing vessel stenosis/obstruction. (D) Similar lesions surrounding the rectum (white arrow).
The stool test showed pH = 7 and did not contain blood, mucus, pus, undigested meat fibers, and fungi. The blood test including complete blood count, coagulation tests, and blood chemistry tests (liver and kidney function tests, pancreatic enzymes) were normal.
The patient underwent an exploratory laparotomy. Intraoperative observation showed that the bowel loops were dilated about 4 cm in diameter, and the colon was soft in palpation without dilation. The entire small bowel mesentery and the right haft of the transverse mesocolon were thickened with yellow nodular cord-like structures along the vessels [Figure 3].
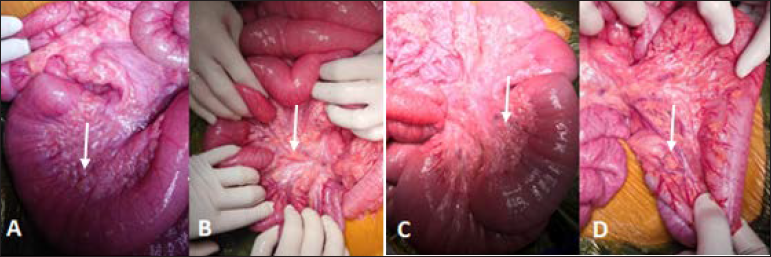
- A 5-year-old male patient with NF-1 presented with abdominal pain and distention. Intraoperative findings: (A) Dilated small bowel (white arrow); (B, C, D) The entire small bowel mesentery and the right half of transverse mesocolon had many yellow rigid nodules (white arrow), along the vessels.
The patient performed biopsies on the lesions of the mesentery and four bowel segments including dilated ileum, collapsed ileum, ascending colon, and transverse colon. The H&E staining specimens on the mesenteric lesions showed tumor tissue structure consisting of clusters of long slender spindle cells with wavy cytoplasm and slightly mucoid basal substances. There was blood vessel proliferation, interspersed with adipose tissue. No evidence of malignant transformation was seen. The immunohistochemical staining results showed S100 (++), SOX10 (-), CD117 (-) [Figure 4]. The final pathologic diagnosis was benign neurofibromatosis. On the specimens of dilated ileum, collapsed ileum, and ascending colon, no enteric ganglia were seen. On the specimens of the transverse colon, there were muscle tissue, enteric nerve plexus, and ganglion cells. Calretininx4 immunohistochemical staining result showed a loss of calretinin expression that indicated a decrease and loss of intestinal muscle function. The Synaptophysin stain result was positive. The pathologic diagnosis was enteric neuromuscular dysfunction.
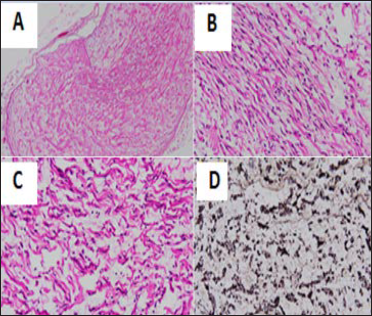
- A 5-year-old male patient with NF-1 presented with abdominal pain and distention. Pathologic images of the mesenteric lesions: H&E stain x40 (A), x200 (B, C) showed tumor tissue structure consisting of clusters of long slender spindle cells with wavy cytoplasm, slightly mucoid basal substances, blood vessels proliferation without malignant cells. (D) The immunohistochemical stain showed S100 (+).
When performing exploratory laparotomy, the surgeon found that the lesions were diffuse, encased in superior and inferior mesenteric vessels, and unable to completely resect. In addition, the pathologic result was benign. Therefore, the patient was treated with conservative therapy to relieve the symptoms and careful observation to detect malignant transformation early.
DISCUSSION
The NF-1 gene, which is found on chromosome 17q11.2, is the source of mutations that lead to NF-1, also known as Von Recklinghausen.[1,4] NF-1 has a prevalence rate of roughly 0.033 percent and affects people of all ethnicities and genders equally. Favorable family history is seen in only 50% of NF-1 cases. Due to the high incidence of mutation in the NF-1 gene, this illness could develop in the remaining 50% of individuals as a result of a new mutation.[7] There was no family history of NF-1 in our instance.
Neurofibromatosis Type I (NF-1) primarily affects the peripheral nervous system and the skin. Café-au-lait spots, neurofibromatosis nodules along the peripheral nerves, Lisch nodules of the iris, and axillary or inguinal freckles are its primary symptoms. One of the early signs of NF-1 is the development of café-au-lait spots, which can appear on the skin as flat, pigmented patches at birth and are frequently oval in form. In addition, to café au lait spots, freckles that are smaller than 5 mm in size and that appear in unexpected locations such as the armpits, groin, underarms, or neck are highly indicative of NF-1. This may enable a conclusive diagnosis of NF-1 in babies. Freckles in the skin creases often start to form between the ages of 3 and 5.[1]
About 10-25% of NF-1 individuals are said to experience abdominal involvement.[4-6] In the abdomen, lesions typically develop in the paraspinal region as well as the retroperitoneal space.[4] Mesenteric fibromatosis is a very uncommon NF-1 manifestation. The majority of instances of GI neurofibromatosis do not have overt symptoms.[5] The stomach and jejunum are the most typical locations for symptomatic tumors. The anatomical location of the lesions and the degree of mucosal involvement influence the manifestations. GI bleeding may result from mucosal involvement.[7] About 65% of patients with small mesenteric neurofibromatosis are asymptomatic. A palpable abdominal lump or discomfort and the distribution of a nerve are signs of abdominal neurofibromatosis.[8]
Neurofibromas show up as a distinct, homogenous, or heterogeneous hypoechoic mass on ultrasonography.[1,7] A barium small-bowel series reveals mucosal or submucosal polyps, mucosal ulceration, external mass effect, and bowel movement.[7,8]
Most neurofibromas are typically smooth, rounded, and homogeneously hypoattenuating on a CT scan, with precontrast attenuation values of around 20-30HU.[8,9] On the venous phase CT scans, the lesions exhibit a modest amplification, ranging from 30 to 50HU.[1,2,4,6] Due to the mucinous stroma of tumors that may be seen under a microscope, neurofibromas exhibit poor attenuation.[1,3] Lesions in the perirectal area frequently show up as perirectal fatty infiltration and can spread to other pelvic tissues like the vagina or uterus.[9] Invasion of the intestinal wall by mesentery neurofibromatosis can result in submucosal or mucosal masses or it may encroach on nearby bowel loops, causing an extrinsic mass impact on the serosal surface.[1] Spotty or widespread soft tissue thickening is a symptom of neurofibromas that infiltrate through the intestinal wall.
When evaluating a tumor in a patient with known NF-1, the MRI imaging findings of neurofibromas might be useful in clarifying instances and are distinctive.[1,7] When compared to muscle tissue, neurofibromas have a low signal intensity on the T1W sequence and high signal intensity on the T2W sequence.[1,7] These traits are brought on by regions of cystic degeneration and the high water content of the mucus matrix. On T2W, the lesion’s core area can be hypointense, indicating fibrosis.[1] The tumors exhibit a modest enhancement after gadolinium injection.[7] Tumors in individuals whose neurofibromatosis progresses to cancer are frequently heterogeneously hyperintense on T2-weighted imaging as a result of cystic degeneration or necrosis. Multiple biopsies should be done to rule out malignancy in cases with suspected malignant transformation.[1,4] Using a CT scan or MRI, the tumor has to be continuously monitored clinically and radiologically for any symptoms of a malignant change.[4,5,8]
The goal of therapy, where it is feasible, is full surgical resection.[4,5,10] Surgical methods need to be avoided, nevertheless, if the bulk cannot be entirely removed.[10] After several biopsies, a close clinical and radiological follow-up to check for symptoms of malignant transformation is the best course of action.[1,4,5] Fast tumor development or worse symptoms might be signs of malignancy.[5] In the care of symptomatic or progressing benign lesions, several adjuvant treatment options, such as ketotifen fumarate, cis-retinoic acid, interferon, and thalidomide, may be crucial.[5] Thus, early detection is essential before the tumor grows too big to be effectively treated for mesenteric neurofibromatosis.[4-6]
CONCLUSION
Mesenteric neurofibromatosis is a rare benign lesion that can transform into malignancy. It often does not show specific clinical symptoms, easily confused with other digestive disorders. Therefore, diagnostic imaging methods for early detection of GI lesions in patients with NF-1 or patients at risk are necessary. If detected at an early stage, the lesion can be completely removed by surgery. In cases of total resection impossible, the tumor should be periodically followed both clinically and radiologically to detect early signs of malignant transformation.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- From the archives of the AFIP: Abdominal neoplasms in patients with neurofibromatosis type 1: Radiologic-pathologic correlation. Radiographics. 2005;25:455-80.
- [CrossRef] [PubMed] [Google Scholar]
- Features of CT and EUS in mesenteric plexiform neurofibroma with Neurofibromatosis type I: A case report. Radiol Case Rep. 2021;16:2103-7.
- [CrossRef] [PubMed] [Google Scholar]
- Neurofibromatosis type 1: A diagnostic mimicker at CT. Radiographics. 2001;21:601-12.
- [CrossRef] [PubMed] [Google Scholar]
- Mesenteric involvement in neurofibromatosis type 1: CT and MRI findings in two cases. Abdom Imaging. 2003;28:822-6.
- [CrossRef] [PubMed] [Google Scholar]
- Intestinal mesenteric involvement with plexiform neurofibroma in neurofibromatosis type 1. Pediatr Int. 2006;48:337-9.
- [CrossRef] [PubMed] [Google Scholar]
- Mesenteric plexiform neurofibroma as a cause of weight loss and chronic diarrhea in a patient with YPEL3 variant. JPGN Reports. 2021;2:e098..
- [CrossRef] [Google Scholar]
- Radiological manifestations of abdominopelvic nerve tumours seen in neurofibromatosis type 1. Clin Radiol. 2014;69:431-5.
- [CrossRef] [PubMed] [Google Scholar]
- Imaging of uncommon retroperitoneal masses. Radiographics. 2011;31:949-76.
- [CrossRef] [PubMed] [Google Scholar]
- Imaging features of neurofibromatosis type 1 in the abdomen and pelvis. AJR Am J Roentgenol. 2021;216:241-51.
- [CrossRef] [PubMed] [Google Scholar]
- Surgical management of abdominal manifestations of type 1 neurofibromatosis: Experience of a single center. Am Surg. 2010;76:389-96.
- [PubMed] [Google Scholar]






