Translate this page into:
An Illustrative Review of Common Modern Craniotomies

*Corresponding author: Rebecca Tuan Le, Department of Radiology, Rochester General Hospital, Rochester, New York, United States. rebecca.le@rochesterregional.org
-
Received: ,
Accepted: ,
How to cite this article: Rao D, Le RT, Fiester P, Patel J, Rahmathulla G. An illustrative review of common modern craniotomies. J Clin Imaging Sci 2020;10:81.
Abstract
Craniotomy, or a surgical opening into the skull, has been observed as early as Paleolithic and Neolithic periods. Early craniotomies carried great morbidity and mortality and standardized during the 20th century, improving surgical outcomes. The simultaneous evolution of medical imaging and stereotactic navigation systems has allowed imaging to correlate findings with surgical approaches, further optimizing patient safety. We review the history of craniotomy and provide an imaging review of the most common craniotomy approaches.
Keywords
Craniotomy
Radiology
Neurosurgery
Medical history
Diagnosis
INTRODUCTION
The earliest evidence of non-accidental skull perforation, or trephination, dates to the late Paleolithic and early Neolithic period (8000–5000 BCE), and has been observed in France and in pre-Inca civilization, respectively.[1-4] The purpose of these early craniotomies is not fully understood. Some have suggested that trephination may have been used for therapeutic purposes related to trauma.[5] Others have suggested that openings in the skull were made as a part of ritual practice, as a result of ancient beliefs in the supernatural.[6,7]
From its mystical origins, craniotomy has evolved through the ages into a staple set of approaches for neurological diseases. The Egyptian physician Imhotep performed craniotomy, presumably related to head injuries that would have resulted from the numerous battles fought by the ancient Egyptians. The Edwin Smith Surgical Papyrus, which provided a scientific approach to craniotomy, dates between the 16th and 17th centuries BCE but is believed to have been originally written by Imhotep around 2900 BCE.[8] The ancient Greek civilization saw a further understanding of pathologies of the central nervous system. By the 5th century BCE, Hippocrates codified guidelines on the use of craniotomy for the treatment of intracranial pathology.[9] During the middle ages, the Arabic surgeon Abul-Qasim Al Zahrawi, known in Western literature as Abulcasis, wrote extensively on early depictions of neurosurgical diagnosis and treatments, including the treatment of head injuries, skull fractures, hydrocephalus, and subdural collections.[10] During the Renaissance, further progress was made in craniotomy techniques. Advances in firearms and grenades in the 16th and 17th centuries led to more cranial trauma. Military surgeons performed craniotomies to evacuate clots and pus.[10] During the second half of the 19th century, after the advent of antisepsis and general anesthesia, the use and technique of craniotomies evolved. By the 20th century, neurosurgery became an autonomous discipline and the modern era began.[10,11]
At first, neurosurgical approaches were performed with extended craniotomies. Large cranial openings were necessary for several reasons. First, the size of pathological lesions could not be determined. Second, intracranial lesions were usually diagnosed when they had reached large sizes. Third, because illumination in operating theaters was poor, the cranial opening had to be sufficiently large enough to allow sufficient light into the surgical field. In addition, with large surgical teams, many hands and instruments obscured the surgical field. Craniotomy approaches evolved overtime, making surgical intervention less dangerous, and the advent of stereotactic navigation has given rise to more focal and defined craniotomies over the site of the lesion.[12] This has led to lower complication rates, shorter hospital stays, and improved surgical outcomes.[13-15]
The development of neurosurgical techniques and approaches was greatly aided by the evolution of advanced diagnostic imaging. The ability to combine high-definition computed tomography images with magnetic resonance imaging (MRI) has improved pre-procedural planning. Image-guided navigation has evolved from frame-based stereotactic systems in the early 19th century to frameless systems currently used in virtually all craniotomy operations. The neurosurgeon selects the safest, most approachable surgical corridor for tumor, vascular, or functional surgery protecting the normal brain and vascular tissues while providing adequate exposure of a cranial lesion.
Optimal outcomes with cranial surgery in part depend on intraoperative patient positioning. There are two ways to position the head: unfixed and fixed. Unfixed is typically used when stabilization of the head is not essential. For example, in children younger than 3 years, pins are not recommended due to the risk of depressed skull fracture. There are different frames available for head fixation. The most commonly used frame is the Mayfield head holder, with the skull fixation pins placed away from the operative site.
The body can be positioned in a supine, prone, lateral, sitting, and semi-sitting position. Supine position is used most commonly, allowing for frontal, parietal, and temporal craniotomies. Pterional approaches, and some posterior fossa approaches for cerebellopontine angle masses, also make use of the supine position. Prone position is used for occipital and suboccipital craniotomy/craniectomy access to the posterior occipital region and posterior fossa. Lateral positioning is used for access to the posterior fossa including retrosigmoid, far lateral approaches, as well as posterior supratentorial craniotomies involving the parietal and occipital lobes. Sitting positioning, although historically used for posterior fossa access, is now rarely used for access to the posterior fossa and pineal region. The sitting position is associated with risks of venous air embolism and spinal cord infarction.[16] Semi-sitting position is also used for pineal region tumors, as well as parietal, occipital lesions, and supracerebellar infratentorial approach.
After the patient is positioned, the incision is planned. The incision is tailored to the underlying intracranial lesion while maximizing the potential for healing and cosmesis, and minimizing injury to large vessels and nerves, such as the superficial temporal artery and facial nerve. Incisions are placed behind the hairline when possible. Historically, incisions used to be planned on a vascular pedicle to optimize wound healing. At present, with the use of intraoperative neuronavigation, smaller linear, lazy S, and curvilinear incisions are utilized.[17-19] This enables craniotomies that limit exposure to normal parenchyma and maximize access to a lesion through the shortest corridor, thus limiting brain injury from retraction. These variations have evolved overtime along with technology. With enhanced imaging, the future may lie with endoscopic or minimally invasive tubular approaches for the treatment of intracranial pathology.[20-23] The following is a review of the most common types of craniotomies used by modern neurosurgeons.
TYPES OF CRANIOTOMY
Frontal
Frontal craniotomy [Figure 1] can be either unilateral or bilateral (bifrontal). The bifrontal craniotomy is used for large midline anterior and sometimes middle skull base lesions. Anterior cranial fossa meningiomas were first treated with bifrontal craniotomy in the late 19th century.[24,25] Lesions of the olfactory groove, planum sphenoidale, and tuberculum sella, as well as falcine and pituitary lesions, can be approached with a bifrontal craniotomy. This approach allows for exposure of anterior interhemispheric lesions, facilitating sufficient exposure of the lesion and its vascular supply while minimizing frontal lobe retraction and injury to adjacent neurovascular structures.[26] Patients are placed in the supine position. Multiple burr holes are placed, two posteriorly on either side of the sagittal sinus. Similarly, two anterior burr holes are placed adjacent to the superior sagittal sinus with additional holes placed over the pterion or lateral temporal region depending on surgeon preference, patient age, and exposure required. Additional burr holes can also be placed at the junction of the superior temporal line and orbital rim and posteriorly to the sphenoid wing. The inferior bony cut is made just superior to the frontal sinus. The frontolateral or unilateral subfrontal approach provides exposure to the anterior cranial fossa. The unilateral subfrontal approach has been used to access the sellar region and olfactory fossa.[27,28] Lesions associated with the optic chiasm, lamina terminalis, and anterior third ventricle can also be accessed.[29] Bifrontal craniotomy is associated with increased risk of cosmetic deformity and cerebrospinal fluid (CSF) leak, therefore, reconstruction of the calvarium is essential for a good outcome. In addition, bifrontal craniotomy may result in injury of the superior sagittal sinus. The bilateral frontal lobes may be injured secondary to retraction.[30]

- A 40-year-old man with planum sphenoidale meningioma on an axial T1 magnetic resonance imaging with gadolinium (a), requiring frontal craniotomy. Three-dimensional volume rendering (b) illustrates expected post-operative frontal craniotomy imaging. Key anatomy to consider is the superior sagittal sinus (c), which is exposed intraoperatively and must be avoided to prevent injury and thrombosis.
Temporal
Modern temporal craniotomy [Figure 2] was developed by Sir Victor Alexander Haden Horsley, who performed the first pituitary adenoma resection by lifting the temporal lobe.[31] The temporal approach is used for access to the middle cranial fossa, including extra-axial lesions, trigeminal nerve lesions, and intra-axial lesions including the mesial temporal lobe. Temporal craniotomy can be made through a linear or question mark incision. Burr holes are placed posterior to the insertion of the zygomatic arch and upper anterior portion of the zygomatic bone.
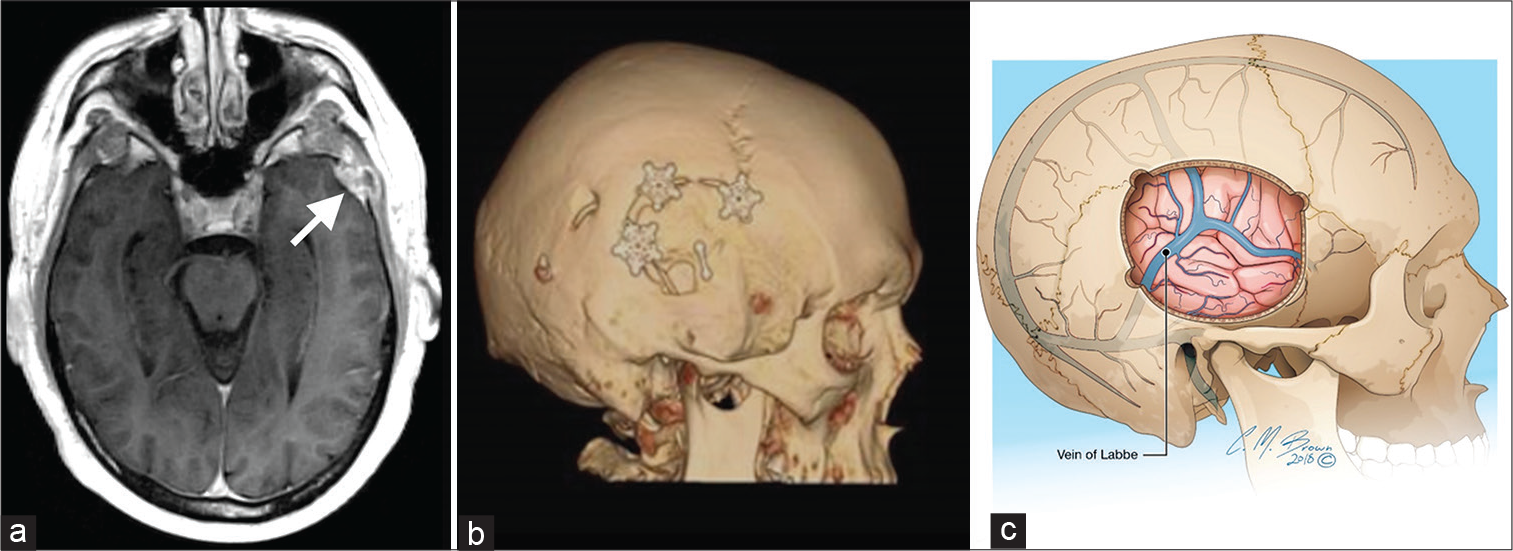
- A 63-year-old man with the left temporal sclerosing meningioma (a – arrow) on axial T1 MRI with gadolinium, requiring temporal craniotomy. Three-dimensional volume rendering (b) illustrates expected postoperative temporal craniotomy imaging. The vein of Labbe (c) traverses through the surgical field and must be avoided to minimize venous hemorrhage or infarction.
A subtemporal approach provides access to the middle fossa and petroclival areas and basal cisterns and superior clivus. The subtemporal approach can be combined with an anterior petrosectomy for lesions at the tip of the petrous bone and for those involving the middle clivus superior to the internal auditory canal.[32] The anterior superior brainstem can also be accessed. Small vestibular schwannomas and trigeminal lesions, and petroclival lesions, such as meningiomas or chondrosarcomas, can be accessed through the subtemporal approach. Basilar aneurysms can also be accessed. The subtemporal approach allows for repair of superior semicircular canal dehiscence. The superficial temporal artery and vein must be identified prior to incision. Identification of the vein of Labbe is required on preoperative imaging and must be preserved. Patients are typically placed in the supine position with the head rotated to expose the temporal and lateral scalp. During closure, adipose tissue can be placed as a barrier against CSF leakage.
Parietal
The parietal approach [Figure 3] provides exposure to the middle and posterior hemisphere. The motor and sensory cortex is visible at the anterior exposure, with the visual cortex posteriorly. Parietal craniotomy was designed to provide exposure to the mid to posterior cerebral hemisphere while sparing the motor and sensory cortices. The parietal approach can be used for both intra-axial and extra-axial lesions, including metastases, gliomas, vascular malformations, and meningiomas. There are two major approaches for lesions through the parietal craniotomy. First, the interhemispheric approach is used for parafalcine, medial parietal, and splenial lesions. Second, the transcortical route allows access to intra-axial lesions through the functionally “silent” superior parietal lobule. Two groups of veins are present along the medial and lateral surface of the parietal lobe. On the medial surface, veins can drain superiorly to or inferiorly from the superior sagittal sinus. On the lateral surface, superiorly and inferiorly draining veins are also present. The most important of the lateral veins is the vein of Trolard or the superior anastomotic vein with crosses from the Sylvian fissure to the superior sagittal sinus. Cortical mapping can be used for localizing sensorimotor cortex for intra-axial lesions. Additional imaging modalities that can be used with neuronavigation include fiber tracking and motor and sensory cortex localization with diffusion tensor imaging and fMRI, respectively. Patients are positioned in a three-quarter prone position with the head positioned so that the parietal scalp is toward the ceiling. Venous lakes can be numerous and have many tributaries into the superior sagittal sinus which can cause bleeding during drilling. Injury to the superior sagittal sinus and overlying cortical veins is a dreaded complication of parietal craniotomy, which can result in both bleeding and sinus/cortical vein thrombosis.
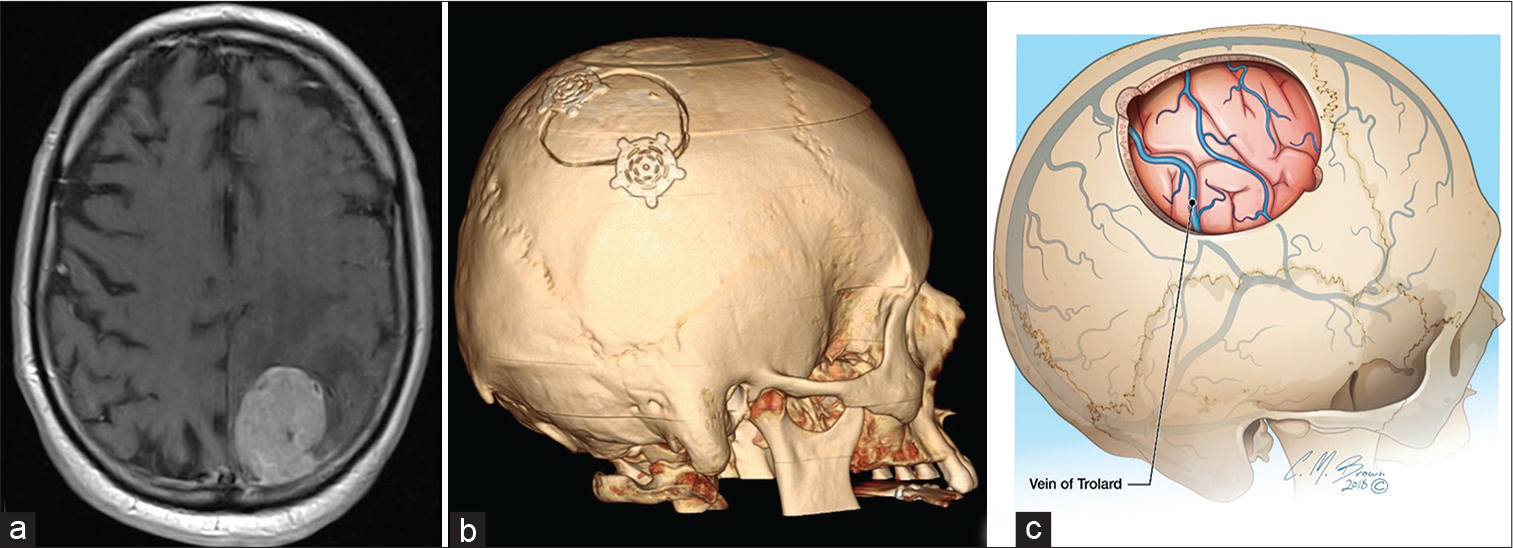
- A 78-year-old man with the left parietal melanoma metastasis (a) on axial T1 MRI with gadolinium. Three-dimensional volume rendering (b) demonstrates parietal craniotomy, the preferred approach for intra- or extra-axial lesions. The vein of Trolard (c) traverses through the surgical field of view and should be avoided to minimize venous hemorrhage or infarction.
Pterional (Frontotemporal)
The pterional, or frontotemporal, approach [Figure 4] was developed by Gazi Yasargil in the 1970s.[33,34] It is the most widely used approach for supratentorial lesions. This approach is the standard craniotomy for treating anterior Circle of Willis aneurysms and the cavernous sinus. Anatomical structures exposed through the pterional approach include the sellar and parasellar lesions, the optic nerves and chiasm, the Sylvian fissure, the frontal, temporal, and parietal operculum, the temporal lobe, and basal cisterns. There are many variants of the pterional craniotomy. All allow access to the frontal lobe, temporal lobe, and Sylvian fissure and allow for direct visualization of the microvasculature surrounding lesions in the suprasellar cistern.[35] This approach can be combined with a subfrontal approach to enter the anterior cranial fossa. Patients are placed in a supine position and the head turned away from the side of the approach. The amount of head turning depends on the location of the pathology. During scalp dissection, care must be made to avoid injury to the superficial temporal artery and temporalis fascia.[36] The frontal sinus, if large enough, may be violated and could require repair with a vascularized pericranial graft. The frontalis branches of the facial nerve should be identified and preserved to avoid frontalis palsy. Osteotomy involving the sphenoid wing may cause an unintentional fracture, which may extend into the optic canal and lead to blindness. Once the dura is opened, the frontal lobe can be gently retracted. The Sylvian fissure is opened around the opercular frontal gyrus which allows exposure of the lateral Sylvian fissure. Further dissection is used for more medial lesions, such as anterior communicating artery or basilar tip aneurysms.
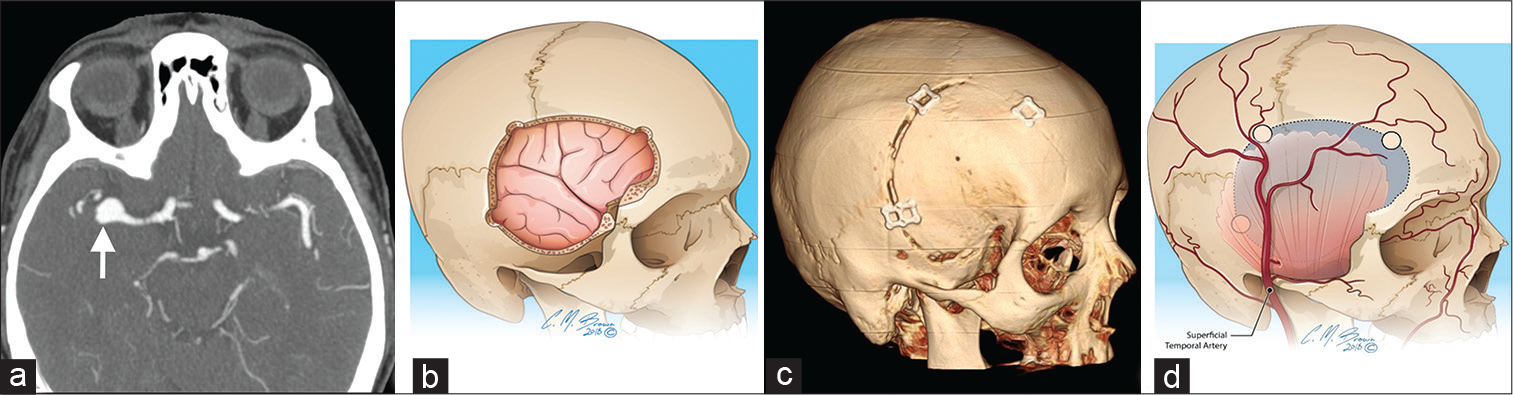
- A 69-year-old woman with the right middle cerebral artery aneurysm (a – arrow) on axial CT angiogram. Sagittal oblique illustration (b) and three-dimensional volume rendering (c) demonstrate pterional (frontotemporal) craniotomy, the preferred approach for Circle of Willis aneurysms or cavernous sinus lesions. The superficial temporal artery (d) traverses through the surgical field of view and should be identified and protected during incision.
Orbitozygomatic (OZ)
First described in 1977 by Pellerin and Hakuba, the OZ [Figure 5] craniotomy combined the fronto-orbital, zygomatic, and temporal craniotomies with removal of the posterolateral wall of the orbital bone and major sphenoid wing.[37,38] Modification of the old frontal craniotomy into a supraorbital variation was proposed by Jane et al. in 1982, which extends to the superior and lateral orbital rim and the zygoma, which allows for greater access to the subfrontal corridor and reduces the need for frontal lobe retraction when accessing the inferior anterior and middle cranial fossa. This “deeper” access is particularly important for high-riding aneurysms of the basilar tip and large posterior communicating artery aneurysms.[36] The OZ approach has been further modified into one or two piece osteotomies. The one-piece modification refers to removing the entire frontotemporal and supraorbital osteotomy as one bone flap. The two-piece approach is made by removing a traditional pterional bone flap followed by a supraorbital craniotomy. Both types of craniotomy have been used for lesions within the orbital apex, paraclinoid, and parasellar areas, the cavernous sinuses, the basal cisterns, and the upper clivus. The OZ approach can also be used for cranial base masses, which extend superiorly. Patient positioning and head position are similar to the pterional approach. Fracture of the orbital roof and rim can occur, leading to injury of the optic nerve within the optic canal. Fractures of the sphenoid and ethmoid sinuses can result in CSF leak.
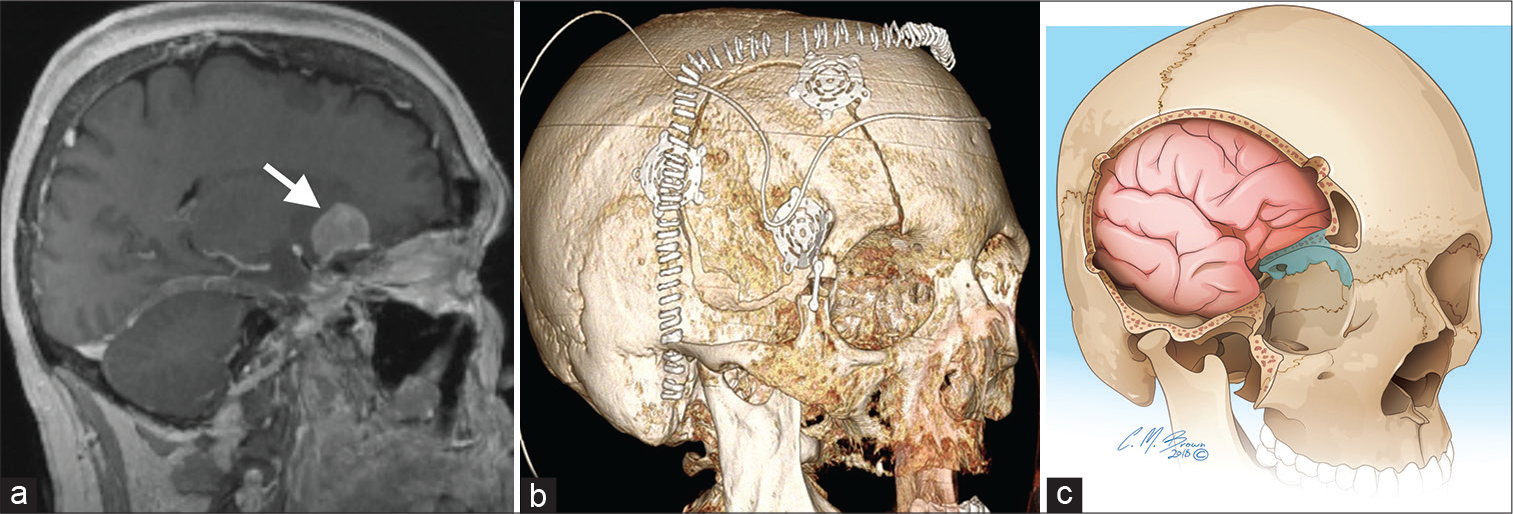
- A 55-year-old woman with the right paraclinoid meningioma (a – arrow) on sagittal T1 magnetic resonance imaging. Three-dimensional volume rendering (b) demonstrates post-operative orbitozygomatic craniotomy, the preferred approach for orbital apex, paraclinoid, cavernous sinus, basal cistern, or upper clivus lesions. An orbital rim fracture (c – blue highlight) should be avoided, as it can result in an optic nerve injury.
Retrosigmoid
The retrosigmoid or lateral suboccipital approach [Figure 6] was popularized by Woolsey and Krause in the early 1900s.[39] After several modifications, the lateral suboccipital approach evolved into what is now called the retrosigmoid approach.[26,39-42] This approach provides optimal access to the cerebellopontine and cerebellomedullary cisterns. The retrosigmoid approach uses a lateral suboccipital craniotomy combined with a partial mastoidectomy to enter the dorsolateral aspect of the posterior fossa.[43] This is the most widely used approach for vestibular schwannoma and other lesions which require exposure of the brainstem and cranial nerves. It is also used for aneurysms of the anterior inferior cerebellar artery, the posterior inferior cerebellar artery, and basilar trunk. Neurovascular decompression of the trigeminal nerve can also be treated through this approach. Small to medium size tumors of the internal auditory canal can be excised with preservation of hearing. Large tumors, which compress the brainstem and adjacent neurovascular structures, can also be resected. Larger tumors may cause neurological symptoms related to mass effect on the brainstem. These patients can be positioned in sitting, supine, or in lateral decubitus position, with lateral decubitus being the most common. Care should be taken to preserve the lesser occipital and greater auricular nerves to reduce postoperative headache and dysesthesia.[43] Post-operative complications include retraction injury to the cerebellum, venous sinus injury, damage to the cranial nerves and brainstem, as well as post-operative CSF leaks.[44] In particular, the mastoid emissary vein should be located during craniotomy exposure because it can cause substantial bleeding and be a source of air embolism.[43] Injury to the vertebral artery can also occur during osteotomy of the lower portion of the exposure at the occipital bone. In cases of intracanalicular extension of a vestibular schwannoma, further exposure of internal acoustic meatus is necessary. The posterior wall of the internal acoustic meatus is drilled, which results in exposure of the superior and inferior vestibular nerves. Hearing preservation is one of the main advantages of the retrosigmoid approach to resection of vestibular schwannoma treatment, as opposed to trans-labyrinthine approach in which the inner ear structures are sacrificed.[45]
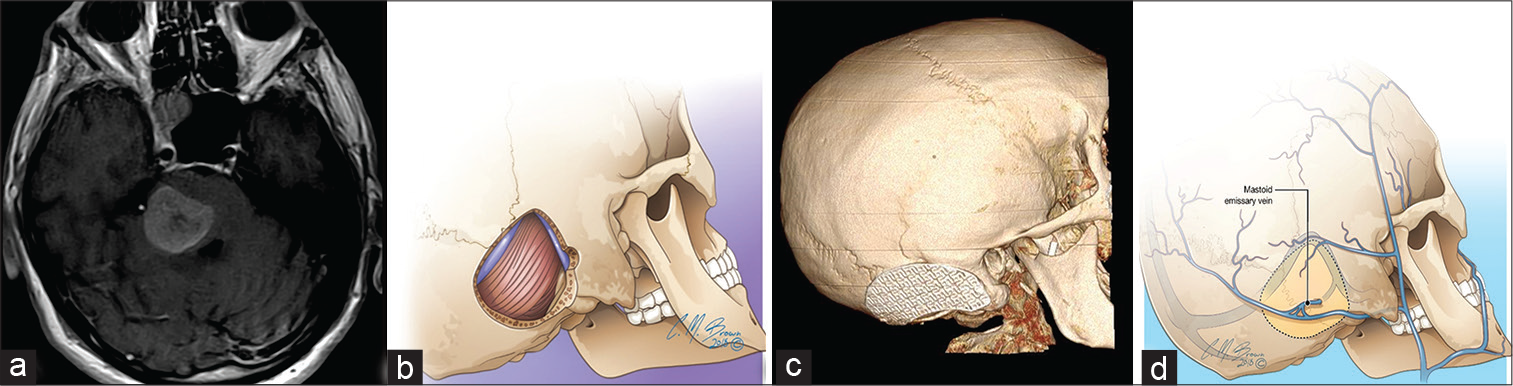
- A 59-year-old man with pontine melanoma metastasis (a) on axial T1 magnetic resonance imaging. Sagittal oblique illustration (b) and three-dimensional volume rendering (c) demonstrating retrosigmoid craniotomy, the preferred surgical approach for cerebellopontine or cerebellomedullary cistern lesions. The mastoid emissary vein (d) traverses through the surgical field of view and should be avoided to minimize hemorrhage or venous air embolism.
Suboccipital
Posterior fossa lesions carried a high operative mortality rate during the early 20th century.[45] Cerebellar lesions were initially considered as very risky or inoperable. Suboccipital craniotomy and craniectomy [Figure 7] are made by removing the caudal portion of the occipital bone.[44] Wide exposure to approach the posterior fossa was performed early in the disease course to avoid brainstem compression during surgery and to allow for posterior fossa decompression. Microsurgical techniques were developed by Rand and Yasargil to more safely navigate the posterior fossa.[46,47] Suboccipital craniectomy is the standard treatment for Chiari 1 malformation.
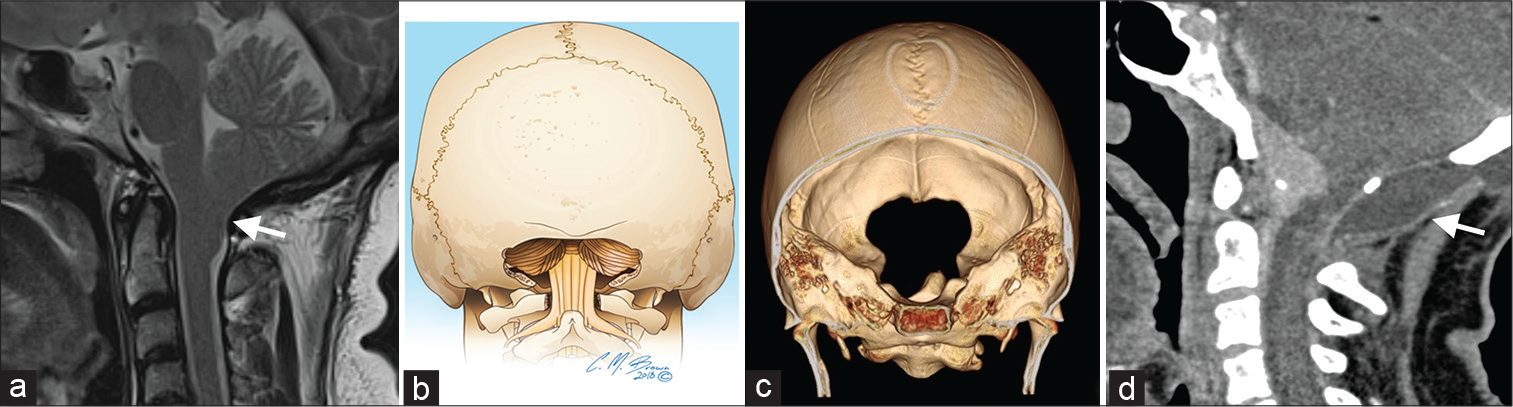
- A 45-year-old woman with Chiari I malformation (a – arrow) on axial and sagittal T2W magnetic resonance imaging. Coronal view illustration (b) and three-dimensional volume rendering (c) demonstrating suboccipital craniectomy, the gold standard for Chiari I malformations. Post-operative imaging may show pseudomeningocele (d – arrow), as seen in this sagittal view CT for a 61-year-old female with a craniocervical meningioma who underwent modified suboccipital craniotomy. This patient was treated with a ventriculoperitoneal shunt to manage the pseudomeningocele and hydrocephalus.
The posterior fossa is the deepest of the three cranial fossae, containing complex anatomy, including the cerebellar hemispheres and vermis, brainstem, cranial nerves, and vasculature. Neurophysiological monitoring is used during risky procedures. Access can be gained to the posterior fossa through the suboccipital approach allowing for the treatment of the cerebellar hemispheres, cerebellar tonsils and vermis, medulla, and fourth ventricle. In addition, lesions of the craniocervical junction and foramen magnum can be accessed. Patients with Chiari 1 malformation also undergo resection of the C1 posterior arch. Patients are usually placed in a prone, lateral decubitus, or sitting position. The prone position has a lower risk of venous air embolism, but blood may pool within the operative bed, limiting visibility. There is also more pressure placed on the face. The lateral position carries a decreased risk of venous air embolism than the sitting position, but the upper cerebellar hemisphere may fall into the surgical field and interfere with the approach. The sitting position carries a higher risk of cardiopulmonary instability, venous air embolism, and rapid CSF leak, which can result in brain herniation. Poor pre-operative planning can also result in difficulty in reaching deep lesions and/or injury to vital structures. For instance, injury to the transverse sinuses can occur during burr hole placement. In addition, the occipital sinus can be injured while cutting open the dura. In cases where opening of the dura is necessary, duraplasty is performed using the nuchal ligament, pericranium from the occipital bone, or a dural substitute. Surgery in the posterior fossa has been reported to carry a complication rate as high as 32%.[35] CSF leak is the most common surgical complication in the posterior fossa. Approximately 35% of children that undergo posterior fossa surgery require CSF diversion through ventriculoperitoneal shunting or third ventriculostomy. Cerebellar mutism is a rare complication occurring in less than 1% of patients, manifesting as slow or frank mutism. It is transient and thought to be secondary to edema or ischemia to the dentate nucleus or pathways of the dentatorubrothalamic tract.[35]
CONCLUSION
The history of craniotomy is colorful, beginning in the Neolithic period. The evolution from skull perforation from its mystical origins to the current advanced techniques has occurred over the course of several 1000 years. Advances in technology, diagnostic imaging, and operative technique have allowed for better exposure, reduced complications, and mortality. The most common craniotomies used by neurosurgeons should be understood in the setting of pre-procedural and post-operative image interpretations.
Acknowledgments
We would like to thank Mr. Chris Brown for his assistance with the illustrations.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- A history of neuroanesthesia In: Eger EI, Saidman LJ, Westhorpe RN, eds. The Wondrous Story of Anesthesia. New York: Springer; 2014. p. :871-85.
- [CrossRef] [Google Scholar]
- Sur les cranes artificiellement perforès á l'époque des dolmens [in French] Bull Mém Soc Anthropol Paris. 1874;9:185-205.
- [CrossRef] [Google Scholar]
- Trepanation of the cranium in ancient Peru. An Fac Med Lima. 1959;42:457-522.
- [CrossRef] [Google Scholar]
- Neurosurgical Classics Park Ridge, Illinois: American Association of Neurological Surgeons; 1992.
- [Google Scholar]
- From mystics to modern times: A history of craniotomy and religion. World Neurosurg. 2016;92:148-50.
- [CrossRef] [PubMed] [Google Scholar]
- Keyhole approaches in neurosurgery, Volume 1, Concept and surgical technique. J Korean Neurosurg Soc. 2009;45:132.
- [CrossRef] [Google Scholar]
- Intraoperative low-field MR-guided frameless stereotactic biopsy for intracerebral lesions. Acta Neurochir (Wien). 2013;155:721-6.
- [CrossRef] [PubMed] [Google Scholar]
- Frameless stereotactic surgery using intraoperative high-field magnetic resonance imaging. Neurol Med Chir (Tokyo). 2004;44:522-33.
- [CrossRef] [PubMed] [Google Scholar]
- Frameless image-guided stereotactic brain biopsy procedure: Diagnostic yield, surgical morbidity, and comparison with the frame-based technique. J Neurosurg. 2006;104:233-7.
- [CrossRef] [PubMed] [Google Scholar]
- The sitting position in neurosurgery: Indications, complications and results. A single institution experience of 600 cases. Acta Neurochir (Wien). 2013;155:1887-93.
- [CrossRef] [PubMed] [Google Scholar]
- Far lateral approach and transcondylar and supracondylar extensions for aneurysms of the vertebrobasilar junction In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :951-7.
- [CrossRef] [Google Scholar]
- Surgical management of cranial dural arteriovenous fistulas In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :959-76.
- [CrossRef] [Google Scholar]
- Fungal infections of the central nervous system In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :1691-732.
- [CrossRef] [Google Scholar]
- Stereotactic minimally invasive tubular retractor system for deep brain lesions. Neurosurgery. 2008;63:334-9.
- [CrossRef] [PubMed] [Google Scholar]
- The risk of cerebral damage during graded brain retractor pressure in the rat. J Neurosurg. 1985;63:608-11.
- [CrossRef] [PubMed] [Google Scholar]
- Minimally invasive transsulcal resection of intraventricular and periventricular lesions through a tubular retractor system: Multicentric experience and results. World Neurosurg. 2016;90:556-4.
- [CrossRef] [PubMed] [Google Scholar]
- Intracranial lesions, illustrating some points in connexion with the localization of cerebral affections and the advantages of antiseptic trephining. Lancet. 1881;3031:581-3.
- [CrossRef] [Google Scholar]
- Chirurgie im inneren gehörgang und an benachbarten strukturen In: Naumann HH, ed. Kopf und Hals-Chirurgie. Vol 3. Stuttgart, New York: Thieme; 1976. p. :457-543.
- [Google Scholar]
- Chirurgie des Gehirns und Rückenmarks Nach Eigenen Erfahrungen Berlin, Germany: Urban and Schwarzenberg; 1908.
- [Google Scholar]
- Surgical management of lesions of the clivus In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :486-500.
- [CrossRef] [Google Scholar]
- Olfactory groove meningiomas: Clinical outcome and recurrence rates after tumor removal through the frontolateral and bifrontal approach. Neurosurgery. 2007;60:1224-32.
- [CrossRef] [PubMed] [Google Scholar]
- History of pituitary surgery In: Greenblatt SH, Dagi TF, Epstein MH, eds. A History of Neurosurgery: In its Scientific and Professional Contexts. Park Ridge, Illinois: American Association of Neurological Surgeons; 1997. p. :373-400.
- [Google Scholar]
- Craniopharyngiomas In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :292-302.
- [CrossRef] [Google Scholar]
- Microsurgical treatment of intracranial saccular aneurysms. J Neurosurg. 1972;37:678-86.
- [CrossRef] [PubMed] [Google Scholar]
- Microsurgical pterional approach to aneurysms of the basilar bifurcation. Surg Neurol. 1976;6:83-91.
- [Google Scholar]
- Cerebellar tumors in adults In: Quinones-Hinojosa A, ed. Schmidek and Sweet Operative Neurosurgical Techniques: Indications, Methods, and Results (6th ed). Philadelphia, PA: Saunders; 2012. p. :169-77.
- [CrossRef] [Google Scholar]
- Neuropathology of traumatic brain injury In: Winn HR, ed. Youmans Neurological Surgery. Vol 4. Philadelphia, PA: Saunders; 2011. p. :3288-300.
- [CrossRef] [Google Scholar]
- Usefulness of the orbitofrontomalar approach associated with bone reconstruction for frontotemporosphenoid meningiomas. Neurosurgery. 1984;15:715-8.
- [CrossRef] [PubMed] [Google Scholar]
- The orbitozygomatic infratemporal approach: A new surgical technique. Surg Neurol. 1986;26:271-6.
- [CrossRef] [Google Scholar]
- History of acoustic neurinoma surgery. Neurosurg Focus. 2005;18:e9.
- [CrossRef] [PubMed] [Google Scholar]
- Surgical exposure of the internal auditory canal and its contents through the middle, cranial fossa. Laryngoscope. 1961;71:1363-85.
- [CrossRef] [PubMed] [Google Scholar]
- Zur operation der akustikusneurinome [in German] Z Hals Nasen Ohrenheilkd. 1937;42:237-9.
- [Google Scholar]
- Results of removal of acoustic tumors by the unilateral approach. Arch Surg. 1941;42:1026-33.
- [CrossRef] [Google Scholar]
- Neuroembryology In: Winn HR, ed. Youmans Neurological Surgery. Vol 1. Philadelphia, PA: Saunders; 2011. p. :78-98.
- [CrossRef] [Google Scholar]
- Retrosigmoid approach: Indications, techniques, and results. Otolaryngol Clin North Am. 2012;45:375-97.
- [CrossRef] [PubMed] [Google Scholar]
- Suboccipital approach In: Perneczky A, Reisch R, Kindel S, eds. Keyhole Approaches in Neurosurgery. Vienna, Austria: Springer; 2008. p. :179.
- [Google Scholar]
- Microneurosurgery: Application of the binocular surgical microscope in brain tumors, intracranial aneurysms, spinal cord disease, and nerve reconstruction. Clin Neurosurg. 1968;15:319-42.
- [CrossRef] [PubMed] [Google Scholar]






