Translate this page into:
Accuracy of Serial PET-CT Imaging in Systemic Sarcoidosis
Address for correspondence: Dr. Roberto G Carbone, MD FCCP Respiratory Unit, Department of Internal Medicine, Regional Hospital Aosta - Italy. E-mail: magister1@mail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Positron emission tomography (PET) in combination with computed tomography (PET-CT) is commonly used to identify malignant lesion in the lung. Despite there being only a few reports in literature, PET-CT imaging may have many advantages in the study of sarcoidosis, being useful in the diagnosis as well as in monitoring the response to treatment. The object of this case report is to highlight the clinical utility of integrated PET-CT imaging for evaluation of patients with systemic sarcoidosis and for comparing baseline findings to follow-up readings.
Keywords
Diagnosis
monitoring
PET-CT imaging
systemic sarcoidosis
therapy
INTRODUCTION

Positron emission tomography (PET) is commonly used to indentify malignant lesion in the lung. Today PET scans are performed using instruments with combined PET and computed tomography (CT) scanners. The combined PET-CT scans provide images that help pinpoint abnormal metabolic activity of anatomical structures seen in a CT scan, leading to a more accurate diagnosis. PET-CT imaging may have many advantages in the study of sarcoidosis, but very little data is available on this topic. Monitoring the metabolic activity of noncaseating granulomas by PET-CT can offer an accurate tool for the evaluation of response to treatment and disease activity.[12] Notably, PET-CT has been used with great success in identifying potential sites for biopsy and in obtaining histologically diagnostic samples.[2] The current report demonstrates that PET-CT imaging is evolving into an accurate tool for the diagnosis and monitoring of systemic sarcoidosis.
CASE REPORT
A 42-year-old male was admitted to the Respiratory department of the University of Genoa with fever, chest pain, and fatigue. Total body PET- imaging revealed hypermetabolic activity in multiple organs including lungs, liver, spleen, mediastinum, and lymph node chains above and below the diaphragm [Figures 1 and 2]. The standardized uptake value (SUV) in the lesion was calculated based on the activity concentration measured at the end of the scan and corrected for patient's body weight and dose injected as follows: SUV = Tissue Activity (measured counts/pixel/second) × Calibration Factor/Injected [F18] fluorodeoxy glucose (FDG) Dose (milliCuries per kilogram of body weight).[3] The SUV value in this patient was 11.2. Surgical biopsies from the thoracic lymph nodes revealed confluent noncaseating granulomas with multinuclear giant cells, mononuclear phagocytes, and lymphocytes [Figure 3]. No necrosis was identified, and subsequent cultures were negative for mycobacteria and fungi. The patient was diagnosed with sarcoidosis.
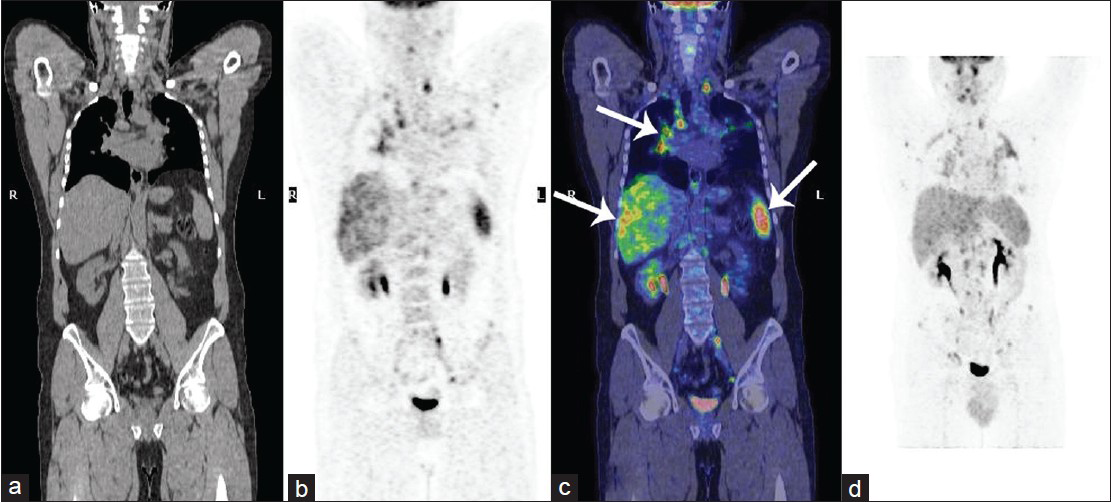
- 42-year-old male with fever, chest pain, and fatigue diagnosed with sarcoidosis. Pre-treatment 18F- FDG PET-CT total body scans of the patient from the base of the skull to the thighs: (a) CT scan, (b) PET scan, (c) Fusion coronal image, and (d) Maximum Intensity Projection (MIP) image show multiple abnormal uptakes of F18-FDG. In particular, some abnormal accumulations of radiotracer are present at the level of right pulmonary hilum, liver, and spleen (arrows in c).
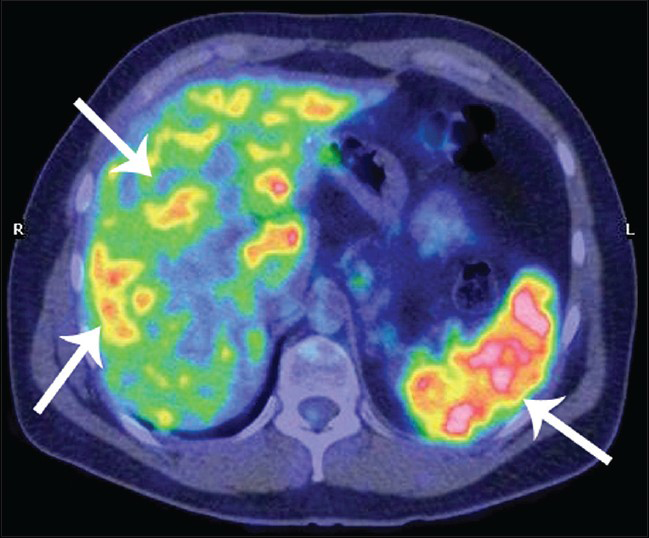
- 42-year-old male with fever, chest pain, and fatigue later diagnosed with sarcoidosis. Pre-treatment 18F-FDG PET- CT scan, transaxial fusion image at the level of the upper abdomen shows abnormal uptakes of radiotracer in the liver and spleen (arrows).
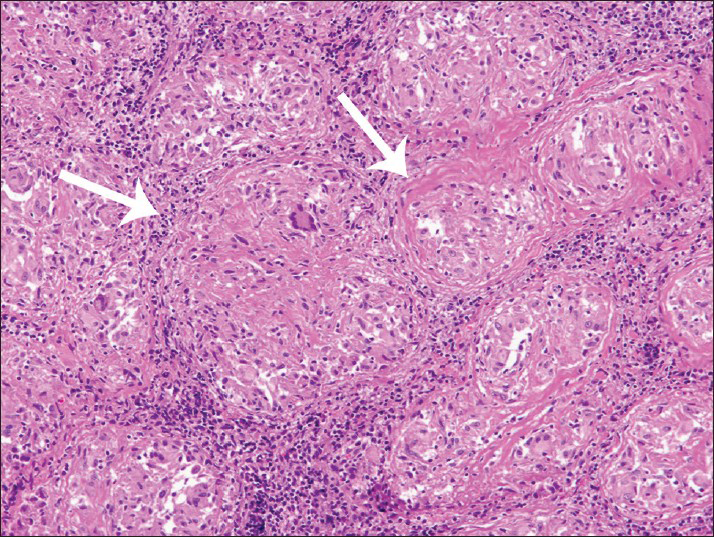
- 42-year-old male with fever, chest pain, and fatigue later diagnosed with sarcoidosis. Photomicrograph of the biopsy sample stained with hematoxylin and eosin shows confluent sarcoid granulomata pattern in thoracic lymph node (arrows). Tissue showed occasional multinuclear giant cells Langhans type, mononuclear phagocytes, and lymphocytes. No necrosis was present (×100).
After the diagnosis of sarcoidosis was confirmed, the patient was initially treated with prednisone 40 mg daily for one month. Over the next 5 months, the patient's dose of prednisone was gradually reduced to 10 mg. At this point, the patient continued to complain of fatigue and chest pain. Therefore, oral methotrexate at a dose of 10 mg once a week was added for the next 9 months. Fifteen months after initiating therapy, the patient reported total resolution of symptoms and his repeat PET-CT scan was now normal [Figure 4]. FDG scan images demonstrated a significant decrease in hypermetabolic activity in the lesion as shown by a decrease in the SUV value from an initial SUV of 11.2 to the follow-up study post-treatment showing a SUV of 2.2. Monitoring the metabolic activity of noncaseating granulomas by PET-CT can offer an accurate tool for the evaluation of response to treatment and disease activity.[23]
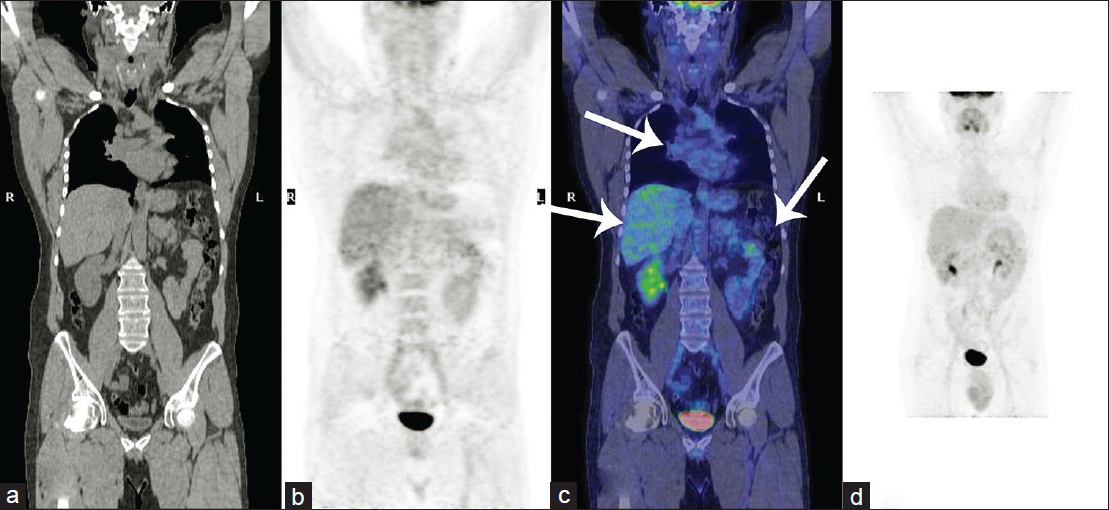
- 42-year-old male with fever, chest pain, and fatigue later diagnosed with sarcoidosis. Post treatment total body 18F-FDG PET/CT scan: (a) CT scan, (b) PET scan, (c) Fusion coronal image, and (d) MIP image show a physiological distribution of the radiotracer and the complete disappearance of abnormal uptakes at level of right pulmonary hilum, liver, and spleen (arrows in c).
DISCUSSION
There are only limited number of reports about the usefulness of PET-CT in sarcoidosis. The objective of this paper is to highlight the clinical utility of integrated [18F] FDG-PET-CT imaging, for evaluation of patients with systemic sarcoidosis by comparing baseline SUV values to follow-up PET-CT findings after treatment for active sarcoidosis, and by correlating changes in PET-CT with changes in clinical status. The SUV of the FDG accumulation is used as a quantitative index for differentiating between benign and malignant lesions. A SUV of less than 2.5 is generally considered to be indicative of a benign lesion. In this case the changes in PET-CT following treatment were in agreement with changes in clinical symptoms. The advantage of PET-CT technique is that it can visualize tracer's accumulation in activated inflammatory cells and simultaneously provide PET and CT images. PET-CT could be used to accurately assess inflammation activity in sarcoidosis patients with unexplained, persistent, disabling symptoms without serological inflammation activity, and is helpful in predicting pulmonary deterioration after 1 year in untreated patients or the pulmonary improvement expected after treatment. The PET-CT may demonstrate active inflammation in advanced stages of disease. Several studies confirm these findings.[45] In addition, one study, reported 100% sensitivity of PET-CT for pulmonary and systemic sarcoidosis compared with 81% sensitivity for 67Ga scintigraphy/single photon emission computed tomography (SPECT) and showed positive PET-CT results in cases with negative 67Ga scans.[1] Thus, in our study 67Ga scan was not done. The current case of systemic sarcoidosis found PET-CT useful for the staging and identification of occult sites for biopsy and for the assessment of inflammatory active sarcoidosis in patients with prolonged symptoms. PET-CT provides better visualization of extra-thoracic sites of active sarcoidosis, such as, when the lesion is in the mediastinum, liver, spleen, and lymph nodes and when the diagnosis is extremely difficult because only one organ that is initially involved is in an uncommon location, or when a biopsy is not possible, or an occult location for biopsy sample is found.[2] Therefore, PET-CT has been used in video-assisted thoracoscopic surgery procedures with great success in obtaining diagnostic biopsy samples.
PET-CT study also has a role in the clinical management of patients with chronic persistent sarcoidosis, since it may identify which patients still have active inflammation that may respond to therapy.[6] It may also identify which patients may require prolonged treatment with anti-inflammatory agents such as infliximab.[4] Thus, PET-CT is a valuable adjunct to clinical evaluation for monitoring the response to treatment in these patients.
Angiotensin-converting enzyme (ACE) was not performed because the diagnostic and prognostic usefulness of the serum ACE is questionable. In a study of 1941 patients with sarcoidosis, 1575 healthy control subjects, and 1355 patients with other diseases, the sensitivity of an elevated serum ACE for the diagnosis of sarcoidosis was 57%, specificity 90%, positive predictive value 90%, but negative predictive value was only 60%.[7] Therefore, this test is not adequately sensitive to be useful for diagnostic screening of sarcoidosis or for managing disease. Because of this, serial measurements of ACE were not performed in this case.
Cardiac sarcoidosis is a potentially life-threatening disease. PET-CT has proved to be of special interest in the detection of cardiac involvement. Inhibition of the physiological myocardial uptake with heparin injection or appropriate diets could enable the detection of cardiac sarcoidosis active lesions with total body PET-CT scan. PET has been reported to have a high sensitivity of 100% and a specificity of 81.5-90.9%.[8] Blankstein et al., found that the presence of focal perfusion defects and uptake on cardiac PET-CT identified patients at higher risk of death or ventricular tachycardia. These findings offer prognostic value beyond Japanese clinical criteria, the presence of extra-cardiac sarcoidosis, and left ventricular ejection fraction.[9] Our patient did not have cardiac involvement, thus avoiding serious complications. In fact an increased FDG in the myocardium is prognostically important as this finding is associated with increased rate of death/ventricular tachycardia.[9]
A major limitation of PET-CT is its lack of specificity. For example, PET-CT can be positive for both malignancy and sarcoidosis in a patient with both conditions, such as a patient with the “sarcoidosis-lymphoma syndrome”.[10] Other factors include cost and potentially excessive radiation exposure. Further prospective multicenter studies are needed to refine the clinical applications of PET-CT in patients with sarcoidosis and drive the field forward.
CONCLUSION
This report demonstrates that PET-CT imaging is evolving into an accurate tool for the diagnosis and monitoring of systemic sarcoidosis. Crucially, total body PET scanning must be suggested to: i) Provide a more comprehensive image of sarcoidosis activity closely related to clinical findings, ii) Assess response to antiinflammatory therapy in sarcoidosis, iii) Identify sites for diagnostic biopsy in patients without apparent lung involvement, and iv) Detect cardiac involvement.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/21/131645
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Imaging the inflammatory activity of sarcoidosis: sensitivity and inter observer agreement of (67) Ga imaging and (18) F-FDG PET. Q J Nucl Med Mol Imaging. 2011;55:66-71.
- [Google Scholar]
- Results of 188 whole-body fluorodeoxyglucose positron emission tomography scans in 137 patients with sarcoidosis. Chest. 2007;132:1949-53.
- [Google Scholar]
- 18F-FDG PET in sarcoidosis: An observational study in 12 patients treated with infliximab. Sarcoidosis Vasc Diffuse Lung Dis. 2008;25:143-9.
- [Google Scholar]
- Prediction of relapse after discontinuation of infliximab therapy in severe sarcoidosis. Eur Respir J. 2014;43:602-9.
- [Google Scholar]
- Severity of pulmonary involvement and (18) F-FDG PET activity in sarcoidosis. Respir Med. 2012;107:439-47.
- [Google Scholar]
- The utility of 18F-FDG PET/CT for diagnosis and adjustment of therapy in patients with active chronic sarcoidosis. J Nucl Med. 2012;53:1543-9.
- [Google Scholar]
- A Concise Review of Pulmonary Sarcoidosis. Am J Respir Crit Care Med. 2011;183:573-81.
- [Google Scholar]
- Cardiac Positron Emission Tomography Enhances Prognostic Assessments of Patients with Suspected Cardiac Sarcoidosis. J Am Coll Cardiol. 2014;63:329-36.
- [Google Scholar]
- Sarcoidosis lymphoma syndrome-the value of PET-CT in the diagnosis. World J Surg Oncol. 2013;11:235.
- [Google Scholar]






