Translate this page into:
Systemic sarcoidosis with pituitary adenoma

*Corresponding author: Francesco Puppo, Department of Internal Medicine, University of Genoa, Viale Benedetto XV, Genoa, Italy. puppof@unige.it
-
Received: ,
Accepted: ,
How to cite this article: Carbone RG, Daniele P, Monselise A, Puppo F. Systemic sarcoidosis with pituitary adenoma. J Clin Imaging Sci 2022;12:32.
Abstract
Involvement of the nervous system with sarcoidosis is seen clinically in approximately 5-15% of cases. In most cases, lesions are localized to the leptomeninges and cranial nerves, and rarely to the pituitary gland, leading to endocrinologic abnormalities. We report on an original clinical case demonstrating the effectiveness of 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) scan in the diagnosis and monitoring of systemic sarcoidosis with probable pituitary involvement.
Keywords
Sarcoidosis
Pituitary adenoma
18FDG-PET/CT
INTRODUCTION
Sarcoidosis unexpected and occulted lesions are not easily detectable. 18FDG-PET/CT is helpful to confirm MRI diagnosis of neurosarcoidosis especially in sites in which biopsy is at high risk and is a useful tool for monitoring treatment response.
CASE REPORT
A 42-year-old non-smoker male was admitted to the Department with fever, left chest pain, and fatigue. In addition, he referred to severe headache and joint pain lasting 3 months. Physical examination showed gynecomastia without secondary sexual characteristics. Blood tests showed severe hyperprolactinemia (10,000 ng/mL - normal value in men <70 ng/mL) while all other pituitary function studies were within the normal range. Brain magnetic resonance imaging (MRI) after enhancement with gadolinium identified a pituitary adenoma [Figure 1, panels a and b]. 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18FDG-PET/CT) scan showed multiple uptakes in the lungs, liver, and lymph nodes above and below the diaphragm [Figure 1, panel c]. In addition, a marked uptake was detected in the pituitary gland [Figure 1, panel d]. Surgical biopsies from thoracic lymph node and liver revealed confluent noncaseating granulomas with multinuclear giant cells, mononuclear phagocytes, and lymphocytes without necrosis [Figure 1, panels g and h]. Pituitary biopsy was not performed due to the anatomic site. The patient was diagnosed with systemic sarcoidosis with probable pituitary neurosarcoidosis according to the Neurosarcoidosis Consortium Consensus Group criteria.[1] Clinical and radiological remission [Figure 1, panels e and f] was achieved after a combined 18-month treatment with steroids, methotrexate, and bromocriptine, followed by normal prolactin levels.]
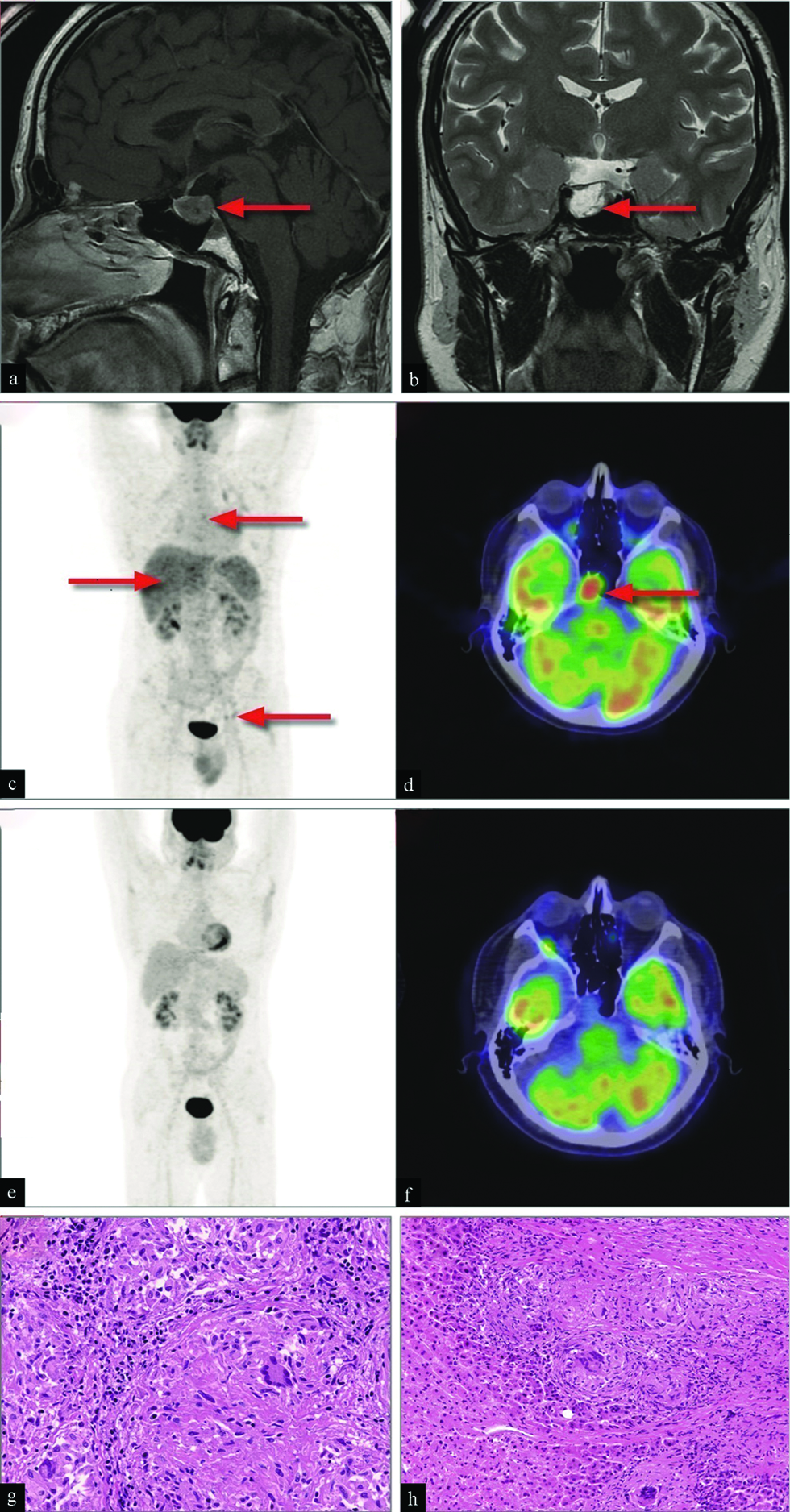
- A 42-year-old male with fever, left chest pain, fatigue, severe headache, joint pain, and gynecomastia was diagnosed with systemic sarcoidosis and probable pituitary neurosarcoidosis. Sagittal (panel a) and coronal (panel b) gadolinium enhanced brain MRI showing pituitary adenoma (arrows). PET/CT visualization with integrated maximum intensity projection and transaxial brain PET, fusion and CT imaging showing focal uptakes of 18F-FDG radiotracer in lungs, lymph nodes and liver (arrows) (panel c) and focal uptake in the pituitary gland (arrow) (panel d). PET/CT visualization with integrated maximum intensity projection and transaxial brain PET, fusion and CT imaging showing complete disappearance of lung, lymph nodes and liver uptakes (panel e) as well as the focal uptake in the pituitary gland (panel f) after treatment. Photomicrograph of biopsy samples stained with hematoxylin and eosin showing confluent sarcoid granulomata pattern with multinuclear giant cells Langhans type, mononuclear phagocytes, and lymphocytes in lymph node (panel g) and liver tissue (panel h), no necrosis was present (×100).
DISCUSSION
Sarcoidosis workup includes (1) preventing or limiting organ damage and improving the quality of life; (2) distinguishing the reversible granulomatous disease from irreversible fibrosis; (3) identifying the most suitable sites for biopsy to obtain an accurate evaluation of disease extension; (4) detect clinical silent lesions. In this setting, FDG-PET/CT is useful to identify active inflammation that indicates reversible granuloma, to detail anatomic sites of the body that are not possible to show with other imaging modalities, and to monitor response to treatment. The involvement of the nervous system is seen clinically in approximately 5-15% of cases of sarcoidosis and is estimated to be seen at autopsy in about 25% of cases.[2] Neurosarcoidosis is hard to diagnose as a bioptic confirmation of the lesion is difficult to obtain and the role of an 18FDG-PET/CT scan in the diagnosis of systemic sarcoidosis has been underlined in the 2021 European Respiratory Society guidelines.[3,4]
CONCLUSION
18FDG PET/CT has an important role in the early detection of unexpected and occulted lesions, especially in anatomical sites in which biopsy is at high risk, including neurosarcoidosis. The present case highlights that brain MRI and 18FDG-PET/CT scans may be helpful in the diagnosis of probable pituitary neurosarcoidosis when neurological involvement is accompanied by systemic symptoms characteristic of the disease.[1,5] Moreover, confirms that 18FDG PET/CT has an important role in the early detection of unexpected and occulted lesions, especially in a site in which biopsy is at high risk, and is a useful tool for monitoring treatment response.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Definition and consensus diagnostic criteria for neurosarcoidosis: From the Neurosarcoidosis Consortium Consensus Group. JAMA Neurol. 2018;75:1546-53.
- [CrossRef] [PubMed] [Google Scholar]
- PET/CT in the diagnosis and workup of sarcoidosis: Focus on atypical manifestations. RadioGraphics. 2018;38:1536-49.
- [CrossRef] [PubMed] [Google Scholar]
- ERS clinical practice guidelines on treatment of sarcoidosis. Eur Respir J. 2021;58:2004079.
- [CrossRef] [PubMed] [Google Scholar]
- Accuracy of serial PET-CT imaging in systemic sarcoidosis. J Clin Imaging Sci. 2014;4:21.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Incidental focal 18F-FDG uptake in the pituitary gland: Clinical significance and differential diagnostic criteria. J Nucl Med. 2011;52:547-50.
- [CrossRef] [PubMed] [Google Scholar]







