Translate this page into:
Unintended Thermal Injuries from Radiofrequency Ablation: Organ Protection with an Angioplasty Balloon Catheter in an Animal Model
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objectives:
The aim of this study was to investigate a novel approach of using a balloon catheter as a protective device to separate liver from the diaphragm or nearby bowel during radiofrequency ablation (RFA) of hepatic dome tumors in an animal model.
Materials and Methods:
All experimental procedures were approved by animal Institutional Review Board. Using a 3 cm RF needle electrode, 70 hepatic ablation zones were created using ultrasound in 7 pigs. 50 lesions were created using balloon interposition between liver and diaphragm; 20 lesions were created using the balloon device interposed posteriorly between liver and bowel. Additional 21 control lesions were performed. Animals were sacrificed immediately; diaphragm and bowel were then visually inspected and sectioned. Diaphragmatic and bowel injury was then classified according to the depth of thickness.
Results:
Control lesions caused full thickness injury, either to diaphragm or bowel. During ablation of lesions with balloon interposition, there was significantly less diaphragmatic injury, P < 0.001 and less bowel injury, P < 0.01.
Conclusion:
Using balloon interposition as a protective device has advantages over previous saline infusion or CO2 insufflation, providing a safe way to expand percutaneous RFA of liver tumors located on the undersurface of the diaphragm. In addition, this method may be used in protection of other organs adjacent to areas being ablated.
Keywords
Ablation
diaphragm
liver tumor
organ protection
INTRODUCTION

Percutaneous radiofrequency ablation (RFA) of liver tumors has been extensively described, and is considered safe and effective mode of treatment with minimal morbidity and mortality.[12345] Several authors have described minor and major complications as a result of RFA being between 2.2% and 9%, respectively.[346789] Besides bleeding and infection other complications may arise due to the treatment. Lesions must be carefully selected for treatment as unintended collateral thermal injury to nearby organs can occur and has been reported in percutaneous, laparoscopic, and open procedures.[210111213] Since the liver is closely associated with the diaphragm, injury to the diaphragm is a major concern in the ablation of superficial sub-capsular lesions. Clinically, diaphragmatic injury causes pain, diaphragmatic paralysis as well as other abdominal and thoracic complications.[4141516] Thermal injury to the diaphragm can also create significant scarring. This can lead to diaphragmatic weakening, which predisposes it to possible rupture after subsequent blunt abdominal trauma.[17] Furthermore, thermal injury to lesions near the diaphragmatic surface poses a risk for nearby structures such as the lung and pericardium. Another feared complication in the ablation of peripherally located lesions is injury to the bowel. Bowel injury has been associated with increased mortality resulting from sepsis and abscess formation.[9] This risk is markedly increased when the bowel is near the targeted tumor.[2]
Targeted CO2 insufflation has been described as a way to manipulate intra-abdominal organs to gain safe access routes.[41819] In particular, Raman et al.,[15] described CO2 insufflation as a way to minimize diaphragmatic injury in a porcine model during RFA. However, these authors stressed several drawbacks to this technique including possible increased risk of bleeding, limited role of ultrasound guidance and inherent risks of CO2 insufflation. Both Ginat and Saad[19] and Tsoumakidou et al.,[18] more recently stressed the difficulties in using CO2 dissection during ultrasound as the gas causes a ring-down artifact and obscures the image.[1819] Furthermore, since CO2 respects the gravity law in its distribution, the presence of adhesions can at times make the dissection difficult. Other studies have looked at using artificially created pleural effusion as a means to enhance the visibility of lesions in the hepatic dome under the diaphragm. However, this artificial effusion does not allow for separation of the diaphragm from the dome of the liver.[1720212223] Lee et al.,[2] described the use of artificially created ascites in a large animal study to increase the sonic window to the liver lesions, effectively creating an artificial space between the dome lesion and diaphragm. However, this study used normal saline as the infusant. Saline is an ionic fluid, able to conduct heat away from the ablation zone. Therefore, an increase in the electrical conductivity to adjacent structures can occur as a result of having this saline barrier. Other studies have compared the infusion of Dextrose 5% in 0.9% Sodium Chloride (D5) versus normal saline and found that D5 offers less heat conductivity and can be infused into the peritoneal cavity in fairly large volumes.[1424] However, the disadvantages to the use of D5 is that it increases the procedure time required to infuse these large volumes and has the potential for creating fluid overload. In addition, extra precautions need to be taken in diabetic individuals, who may be more sensitive to the administration of D5. Furthermore, the authors suggest the possibility that using peritoneal D5 could potentially isolate the liver from the body wall, reducing the surface area and potentially lead to burns of the liver ligamentous attachments.
Tsoumakidou et al., and Ginat and Saad in their study have highlighted the importance of considering the balloon interposition technique for bowel displacement, predominantly during renal tumor thermal ablation.[1819] Their work proposes that an angioplasty balloon catheter can be used as an effective protective device by serving as a barrier between potentially ablated liver and the diaphragm. This technique might provide a safe and effective way to expand the use of percutaneous RFA of liver tumors that are on the undersurface of the diaphragm. In addition, this technique could be used to protect other organs, such as the bowel adjacent to posterior liver tumors.
MATERIALS AND METHODS
All experimental procedures were performed in accordance with the National Institutes of Health guidelines for humane handling of animals and were approved by the animal Institutional Review Board at our institution during the performance of this study.
A total of seven Yorkshire-mix pigs weighing an average of 45 kg were used and all procedures were performed with the animals placed under general anesthesia. All vital signs including heart rate, respiratory rate, body temperature, oxygen saturation and CO2 were continuously monitored by a certified vet technologist. Induction was achieved with the use of an intramuscular injection of Zolazapam 2.2 mg/kg (Clearsynth Labs; Mumbai, India) and Xylazine 1.1 mg/kg (Bayer; Leverkusen, Germany). The animals were intubated and continuous ventilation of isoflurane continuous at 0-4% (Baxter; Deerfield, IL, USA) was administered via an endotracheal tube.
The pigs were placed in the supine position after adequate anesthesia was achieved. The entire upper abdomen was shaved, prepped, and draped in standard sterile fashion. Both thighs were also shaved and grounding pads were placed bilaterally. Transverse and longitudinal ultrasound scans was performed of the liver. Hepatic parenchyma within 1 cm of the hepatic dome (adjacent to the diaphragm) was chosen for ablation.
Radiofrequency ablation technique
A 15-gauge 3 cm RF probe LeVeen needle electrode (Boston Scientific; Natick, MA USA) was used for ablation. This needle electrode carries eight retractable curved tines (hooks), which when fully deployed assume an umbrella shape 3 cm in maximum diameter perpendicular to the axis of the probe. The authors followed the manufacturer's recommendation for ablation, including two roll-off cycles. A total of 21 control lesions were performed, 3/pig. These control lesions were created by advancing the probe under ultrasound guidance, into the hepatic parenchyma and deploying the prongs within 1 cm of the hepatic dome near the diaphragm, or posteriorly near the bowel. Ablation was created by the use of a RF generator as the energy source. Power output was titrated according to the standards set by the RF system for a 3 cm probe. Titration terminated when maximal power output had been achieved without a rise in impedance for 5 min. Average time for lesion creation was 12 min (range: 8-16 min) [Figure 1].
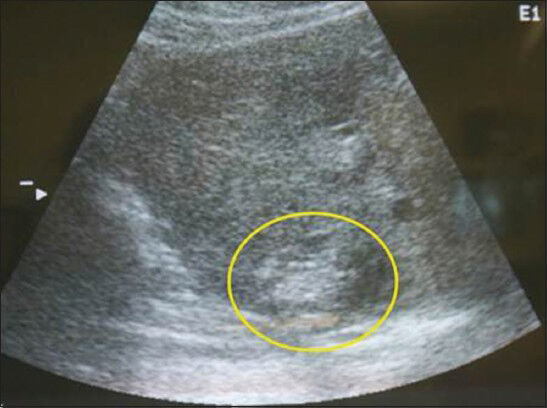
- Longitudinal ultrasound scan of the liver shows the characteristic hyperechoic lesion created with radiofrequency ablation. These lesions were created within 1 cm of the liver dome.
Protective balloon technique
Access to the peritoneum was made using the Seldinger technique. Under fluoroscopic guidance, access to the peritoneum was made through an intercostal mid-axillary approach with the use of a small 22 G micro puncture (Cook group, Bloomington, Indiana; USA) needle. A small amount of contrast was injected through the needle to observe appropriate localization within the peritoneum [Figure 2]. A 0.018 in wire was then advanced through the needle and was observed, under fluoroscopic guidance, passing over the dome of the liver for the diaphragm protected lesions. The needle was removed and a 5 French dilator was placed. A 0.035 in. guide wire was then manipulated under fluoroscopy over the dome of the diaphragm. The dilator was removed and an 18-4 Blue-Max balloon angioplasty catheter (Boston Scientific; Natick, MA USA) was advanced over the wire and placed into several positions along the dome of the diaphragm during lesion creation. For the bowel protected lesions, the balloon catheter was placed along the hepatic dome posteriorly. The balloon was inflated with a mix of Renograffin and saline and maintained insufflated during RF lesion ablation. Since the balloon contained saline, we expected it not to conduct adequate heat to cause damage to the adjacent organ. Using both fluoroscopic and sonographic guidance, the RF probe was advanced to within 1 cm of the hepatic dome and tightly opposed to the balloon catheter. RF lesions were then created as described above. Approximately 10 lesions/pig (total of 70 lesions) were created with the use of the protective balloon device [Figures 3a–c and 4].
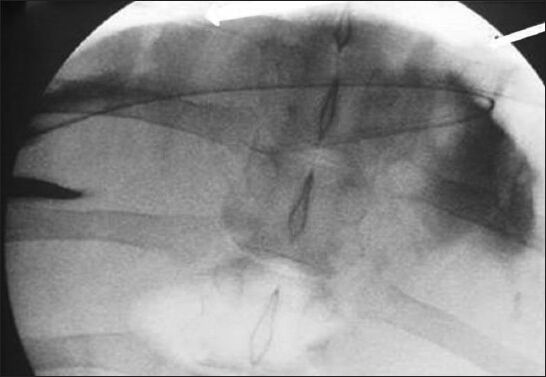
- Anteroposterior fluoroscopic image of the abdomen demonstrates the wire (arrows) passing over the hepatic dome from a right lateral percutaneous approach. Contrast is seen opacifying the correct location of the peritoneal cavity.
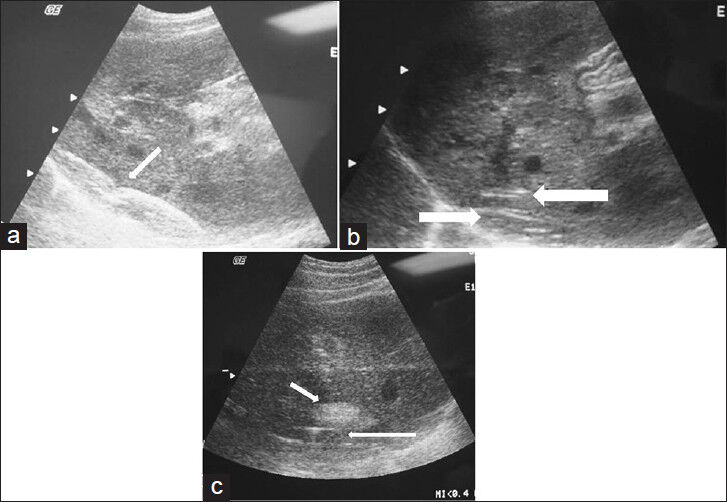
- (a) Longitudinal ultrasound view of the liver shows the positioning and placement of the inflated balloon catheter (arrow). (b) Longitudinal ultrasound view of the liver shows the radiofrequency ablation prongs deployed within 1 cm of the liver surface. The balloon catheter is seen interposed between the liver surface and the diaphragmatic edge (arrow). (c) Longitudinal ultrasound shows the characteristic echogenic focus corresponding to the ablated lesion (short arrow) created in close proximity to the inflated protective balloon (long arrow).
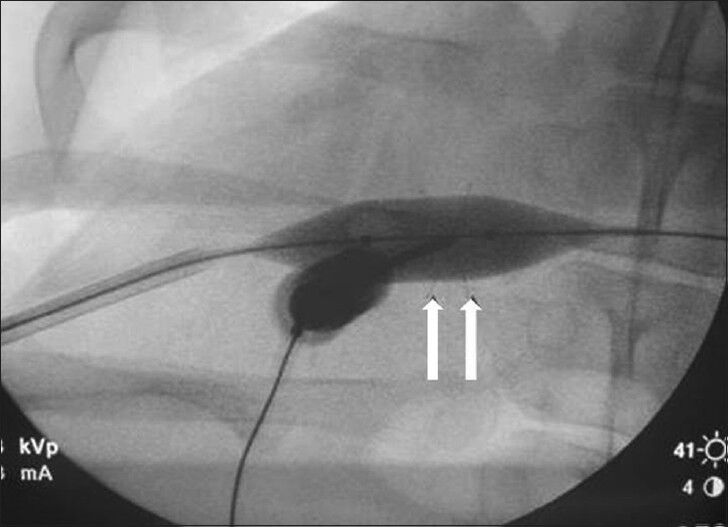
- Magnified anteroposterior fluoroscopic view of the liver demonstrates the balloon inflated during radiofrequency ablation prong deployment.
Pathology
The pigs were killed immediately following the procedure and the liver surface was inspected and sectioned to select lesions that have centers within 1 cm of the surface. Visual inspection of the ablation zones was performed, which were mostly peripheral (without heat sink effect from larger vessels more centrally); they roughly correlated to 3 cm on the axial dimension. At post-mortem examination, the diaphragmatic surface adjacent to the lesion was inspected and photographed. The diaphragm was injured if a discolored segment was seen extending from the pleural to peritoneal margin. The areas of diaphragm corresponding to the ablated hepatic lesions were marked with a nylon suture. Using the method as described by Raman et al.,[17] the areas of suspected diaphragmatic injury were graded on a four point scale: 0-being no injury; 1-mild injury up to one-third of the thickness; 2-moderate injury up to two-thirds of the thickness; and 3-representing severe full thickness injury. The injured areas of the diaphragm or bowel, along with the areas of the diaphragm that were marked with sutures, were then processed with paraffin sectioning and hematoxylin and eosin staining. An experienced pathologist performed the histopathologic analysis. The groups were then compared with respect to the severity of diaphragmatic or bowel injury.
RESULTS
All pigs tolerated the RFA procedure well. No changes in vital signs were observed. In total, 70 RFA zones were produced, 50 zones contiguous with the diaphragm and 20 adjacent to nearby bowel. All these 70 lesions created had visible extension to the hepatic surface. A 3 cm ablation zone was achieved in the axial direction (perpendicular to the needle path) but not in the direction of the needle in order to extend the ablation zone to the correspondent surrounding organ. A total of 15 diaphragmatic control lesions produced transdiaphragmatic injury (Grade 3), including one of which injured the nearby lung parenchyma [Figure 5]. The six bowel control lesions produced severe bowel injury, with injury extending into the mucosa [Figure 6]. The balloon protective catheter was successfully achieved with a single puncture in all pigs.
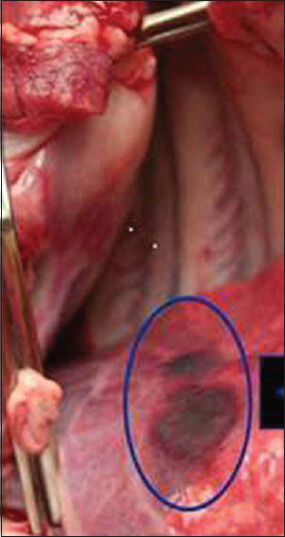
- Gross picture of the lung shows discolored segment of the lung caused from radiofrequency ablation of adjacent liver without protective balloon.
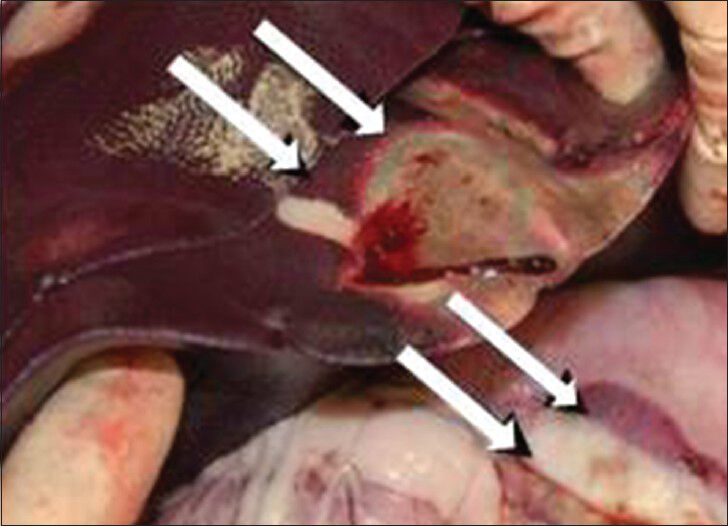
- Gross picture of the bowel shows discolored segment of bowel caused from radiofrequency ablation of adjacent liver.
Of the 50 diaphragm balloon protected lesions, 42 lesions (84%) had no associated diaphragmatic injury (Grade 0), five lesions (10%) had a minimal diaphragmatic injury. Three of these lesions had associated moderate diaphragmatic injury. These three lesions, however, had the complication in that the protective balloon device had ruptured during the ablation procedure. Therefore, the balloon had not been completely inflated and thereby the diaphragm not completely protected, during the creation of these lesions. The control lesions all resulted in nearby diaphragmatic injury. The frequency of thermal injury was significantly different when the two groups were compared.
Out of the 20 bowel balloon protected lesions, 12 (60%) lesions had no associated bowel injury, 2 lesions (10%) had minimal bowel injury and 5 lesions (25%) had moderate bowel injury [Figure 7]. One lesion created injury to nearby mesentery.
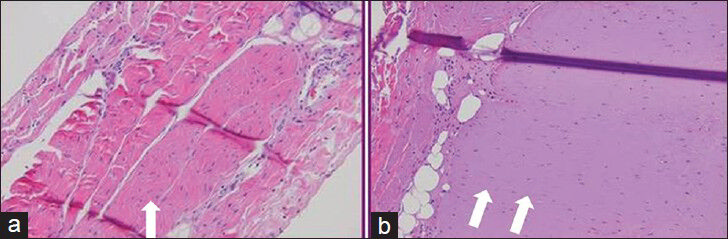
- Histologic specimens stained with hematoxylin and eosin staining (a) from balloon protected diaphragm shows no evidence of injury. (b) from lesion in the diaphragm created without protective balloon demonstrates Grade 3 type injury with a paucity of nuclei.
All control lesions had associated bowel injury. The frequency of thermal injury was significantly different between the two groups (Chi square = 6.686; P < 0.01). There were no complications, such as hemoperitoneum, related to the use of the balloon. Of those cases where the balloon ruptured, the balloon was still able to be withdrawn through the skin access site. No foreign balloon material was left behind in the peritoneal cavity in these cases. Precautions were also taken to ablate the tract if the probe was withdrawn and replaced for another nearby lesion creation.
DISCUSSION
RFA has become the widely accepted non-surgical percutaneous approach to treatment of patients with both primary and metastatic hepatic lesions. However, collateral thermal injury to the diaphragm or nearby bowel are possible drawbacks when percutaneous RFA is performed near the hepatic dome. Diaphragmatic injury has been reported in the literature. This injury leads to significant pain and in several cases, diaphragmatic paresis.[14] In addition, injury to the bowel has also been reported including colonic burns leading to perforation and fistula formation and jejunal perforation.[26721] These cases highlight the importance of carefully selecting lesions in which a sub-capsular tumor is adjacent to organs at risk of injury. We offer a technique of using a balloon angioplasty catheter as a means to protect the diaphragm or bowel during these RF procedures and offer future hope in extending RF treatment to those patients with lesions in these critical areas.
In our study, we show the feasibility of using this balloon catheter as a way to isolate the hepatic dome from the adjacent diaphragm and also to isolate nearby bowel from the posterior liver. Our model of using this balloon catheter offers several advantages. Firstly, interventional radiologists are very familiar with balloon catheter techniques. Secondly, the balloon catheter provides a protective barrier that stays in position, rather than saline infusion which has a tendency to dissipate. Thirdly, ultrasound guidance can be used for this technique as both the RFA probe and balloon catheter can be well visualized. Finally, the risk of bleeding is not increased because the point of entry into the liver remains closely opposed to the abdominal wall, as separation of the diaphragm and liver is localized and does not involve the region of probe entry.
Limitations
Our study had several limitations. First, there is a risk of puncturing the balloon during the deployment of the prongs. This happened with four of the created lesions. Continuous fluoroscopic guidance upon deployment is necessary to assess for this complication. Further work also needs to be performed to clearly delineate the appropriate size and shape of the balloon. For example, a balloon occlusion catheter may allow for more fine-tuned separation than the elongated version of the Blue Max catheter. It was noted that more moderate injury effects were noted to the bowel even if the protective balloon device was used. This is likely to have occurred as a result of difficulty in precise delivery of the balloon catheter in the posterior location. Secondly, further definition of ablation parameters needs to be defined, such as safe distance, type of electrode, and duration of ablation. With newer ablative technologies, such as the microwave ablation system, further studies need to be performed to see if significant differences exist between ablative technologies, i.e., single tine versus multiple tine systems.
As with Raman's earlier studies, the limitations also remain in the applicability of this method in humans. We performed these RFAs in normal pig livers and it is known that the conduction of heat in the hepatic parenchyma in normal liver is different from that of cirrhotic liver morphology. Therefore, these results may change when performed in cirrhotic individuals.
CONCLUSION
In summary, we describe a simple approach to protect the diaphragm and nearby bowel during percutaneous RF ablation of hepatic dome tumors using an angioplasty balloon catheter. We have clearly shown that an inflated angioplasty balloon catheter may help eliminate the magnitude of diaphragmatic or bowel injury. Further development of this technique can offer the possibility of not only protecting the diaphragm during RF ablation, but can also be adapted as a protective device of organs adjacent to areas being treated without the use of artificial ascites or CO2 insufflation.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/1/126018
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Percutaneous radiofrequency thermal ablation of primary and metastatic hepatic tumors: Current concepts and review of the literature. Semin Intervent Radiol. 2006;23:73-84.
- [Google Scholar]
- Effect of artificial ascites on thermal injury to the diaphragm and stomach in radiofrequency ablation of the liver: Experimental study with a porcine model. AJR Am J Roentgenol. 2008;190:1659-64.
- [Google Scholar]
- Treatment of focal liver tumors with percutaneous radio-frequency ablation: Complications encountered in a multicenter study. Radiology. 2003;226:441-51.
- [Google Scholar]
- Adverse events during radiofrequency treatment of 582 hepatic tumors. AJR Am J Roentgenol. 2003;181:695-700.
- [Google Scholar]
- Minimally invasive treatment of malignant hepatic tumors: At the threshold of a major breakthrough. Radiographics. 2000;20:9-27.
- [Google Scholar]
- Essential techniques for successful radio-frequency thermal ablation of malignant hepatic tumors. Radiographics. 2001;21:S17-35.
- [Google Scholar]
- Complications of radiofrequency coagulation of liver tumours. Br J Surg. 2002;89:1206-22.
- [Google Scholar]
- Outcome and long-term survival of patients with liver metastases from colorectal cancer treated with percutaneous cool-tip radiofrequency ablation. Radiology. 2001;221:625-6.
- [Google Scholar]
- Complications of radiofrequency ablation in hepatocellular carcinoma. Abdom Imaging. 2005;30:409-18.
- [Google Scholar]
- Radiofrequency ablation of 231 unresectable hepatic tumors: Indications, limitations, and complications. Ann Surg Oncol. 2000;7:593-600.
- [Google Scholar]
- Radiofrequency ablation of the liver: Current status. AJR Am J Roentgenol. 2001;176:3-16.
- [Google Scholar]
- Local recurrence after laparoscopic radiofrequency thermal ablation of hepatic tumors. Ann Surg Oncol. 2000;7:106-13.
- [Google Scholar]
- Intraoperative radiofrequency ablation or cryoablation for hepatic malignancies. Am J Surg. 1999;178:592-9.
- [Google Scholar]
- Complications of radiofrequency and laser ablation of liver metastases: Incidence and management (abstr) Radiology. 1999;213:509.
- [Google Scholar]
- Minimizing diaphragmatic injury during radiofrequency ablation: Efficacy of intraabdominal carbon dioxide insufflation. AJR Am J Roentgenol. 2004;183:197-200.
- [Google Scholar]
- Hepatic metastases: Percutaneous radio-frequency ablation with cooled-tip electrodes. Radiology. 1997;205:367-73.
- [Google Scholar]
- Minimizing diaphragmatic injury during radio-frequency ablation: Efficacy of subphrenic peritoneal saline injection in a porcine model. Radiology. 2002;222:819-23.
- [Google Scholar]
- Percutaneous thermal ablation: How to protect the surrounding organs. Tech Vasc Interv Radiol. 2011;14:170-6.
- [Google Scholar]
- Bowel displacement and protection techniques during percutaneous renal tumor thermal ablation. Tech Vasc Interv Radiol. 2010;13:66-74.
- [Google Scholar]
- Saline-enhanced radio-frequency tissue ablation in the treatment of liver metastases. Radiology. 1997;202:205-10.
- [Google Scholar]
- Radiofrequency ablation of the liver in a rabbit model: Creation of artificial ascites to minimize collateral thermal injury to the diaphragm and stomach. J Vasc Interv Radiol. 2006;17:541-7.
- [Google Scholar]
- Percutaneous ultrasound-guided radiofrequency ablation with artificial pleural effusion for hepatocellular carcinoma in the hepatic dome. J Gastroenterol. 2003;38:1066-70.
- [Google Scholar]
- Percutaneous sonographically guided radiofrequency ablation with artificial pleural effusion for hepatocellular carcinoma located under the diaphragm. AJR Am J Roentgenol. 2004;183:583-8.
- [Google Scholar]
- Unintended thermal injuries from radiofrequency ablation: Protection with 5% dextrose in water. AJR Am J Roentgenol. 2006;186:S249-54.
- [Google Scholar]






