Translate this page into:
The Utility of Dual Energy Computed Tomography in Musculoskeletal Imaging
Address for correspondence: Dr. Aakshit Goyal, Department of Radio-diagnosis, Era's Lucknow Medical College and Hospital, Sarfarazganj, Hardoi Road, Lucknow - 226 003, Uttar Pradesh, India. E-mail: aakshit11g@gmail.com
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The objective of this article is to review the mechanisms, advantages and disadvantages of dual energy computed tomography (DECT) over conventional tomography (CT) in musculoskeletal imaging as DECT provides additional information about tissue composition and artifact reduction. This provides clinical utility in detection of urate crystals, bone marrow edema, reduction of beam hardening metallic artifact, and ligament and tendon analysis.
Keywords
Bone marrow edema
dual energy computed tomography
metallic artifact
musculoskeletal imaging
urate crystals

INTRODUCTION
Dual energy computed tomography (DECT) or spectral imaging was first conceived in the 1970s;[1] however, its clinical utility has seen an increase in the last decade since the majority of literature has been published in this time frame. The potential areas where DECT provides added values and benefits include the field of neuroradiology, chest and cardiovascular system, vascular, gastrointestinal system, and renal and musculoskeletal imaging.[1] However, the current article focuses on its utility in musculoskeletal imaging.
DECT scanners are of three types:
-
Dual source dual energy
-
Single source dual energy with fast kilovoltage switching
-
Single source dual energy with dual detector layers.
Coupal et al.,[2] described the relative advantages and disadvantages of the three scanners.
Johnson described three commercial ways to obtain a DECT-dual source CT, rapid kilovoltage switching, and sequential acquisition.[3]
RAPID KILOVOLTAGE SWITCHING
In this method, the tube voltage alternates between high and low voltages with a small time gap of <0.5 ms. The data acquisition occurs twice, one for each projection, thus prolonging the acquisition time. Moreover, this setup offers limited photon output at lower energy levels, thus decreasing signal-to-noise ratio which increases the need for a higher tube current and a corresponding higher voltage. The spectral resolution for this system thus remains limited; however, good spatial resolution can be obtained.[3]
SEQUENTIAL ACQUISITION
This approach requires least hardware effort as two datasets at different tube voltages are acquired sequentially. There are two ways to scan the area of interest – either as two subsequent helical scans or as a sequence with subsequent rotations. Similar to the rapid kVp switching technique, this method also increases time to acquisition because of the delay between two simultaneous acquisitions which may lead to image artifact. However, this technique can be used for metal artifact removal.[3]
Dual source scanners are equipped with two X-ray tubes (80/140 kVp) within a single gantry. The tubes can operate at 80, 100, 120, and 140 kVp allowing simultaneous acquisition of images at these two energy levels.[456] DECT uses an additional attenuation measurement that allows the differentiation of materials with different chemical composition to be distinguished from each other.[1]
Grasruck et al.,[7] in their study compared dual source dual energy scanner performance with that of single source dual energy in terms of contrast-to-noise ratio and concluded higher performance of the former system. Moreover, the latter acquisition system showed a poor temporal resolution.
The utility of DECT in musculoskeletal imaging is used to confirm gout as it is able to detect monosodium urate crystals (MSU),[8] evaluate bone marrow,[9] especially in posttraumatic conditions by generating virtual noncalcium images (VNCa), minimize metallic artifacts from prosthesis by minimizing beam hardening artifacts,[10] and assessment of tendons and ligaments using collagen decomposition algorithm. However, the most promising use has been in diagnosis and follow-up of patients with gout.[11]
Radiation exposure for DECT depends on the type of approach. Ho et al.,[12] showed two to three times higher doses for DECT based on single source rapid voltage switching technique. However, their study showed certain pitfalls as they did not normalize image noise or dose. In dual source scanners, the tube currents can be adjusted so that the radiation exposure is similar to that of single energy CT. Schevzle et al.,[13] showed improved contrast-to-noise ratio for DECT at equivalent dose.
However, the studies take into account thoracic and abdominal imaging. There exists paucity of literature regarding the radiation dosage in DECT musculoskeletal imaging. Studies by De Cecco et al., not only state that the radiation dose for imaging in gout for peripheral joints to be around 3 mSv but also state peripheral joints being less radiosensitive.[14] Pache et al., used radiation dose of about 11.15 mGy for bone marrow edema detection and tendon imaging. Bamberg et al., used radiation dose of 11 mGy and 15.4 mGy for metal artifact reduction from extremities and trunk, respectively. DECT exposes the patient to excessive radiation; however, it is way <100–200 mSv, the proposed values below which there is no statistical significance of increased risk of radiation induced malignancy.[15]
At our institution, we use 384-slice dual source DECT scanner (Somatom Force, Siemens Healthcare). Scan parameters include two tubes with peak kilovoltages of 150 kVp with a tin filter and 70 kVp with their corresponding detectors and automated tube current settings. The increased gap between peak kilovoltages in our scanner helps in better characterization of tissues as compared to previous generation scanners having tubes with peak kilovoltages of 80 and 140 kVp. Postprocessing occurs on a multimodality workstation (SYNGO-VIA) and images are studied using color-coded multiplanar reformations and three-dimensional volume rendered techniques.
GOUT
Deposition of MSU crystals in the joints, tendon, bursae, or surrounding tissues elicits an intense immune inflammatory response as seen in gouty arthritis. This eventually would lead to joint destruction and weakening of tendons and ligaments making them prone to rupture.[16] Gout currently affects approximately 8.3 million (4% population) people in United States and its prevalence has seen an increase in the last decade.[17]
Acute gout can involve almost any joint; however, the first metatarsal phalangeal joint of the foot is the most commonly affected. Although gout is more often a peri-arthritis in the soft tissues than actual joint involvement. The higher the blood urate levels, longer the duration of disease, greater is its severity. If left untreated, the urate deposition can lead to joint inflammation and subsequently joint destruction. This necessitates the need for diagnosis of gout with high accuracy as it would help in intervening in the disease process early on its course and prevent associated morbidity.[16]
Certain inflammatory joint diseases such as rheumatoid arthritis, osteoarthritis, septic arthritis, psoriasis, and calcium pyrophosphate dehydrate crystal deposition disease can mimic gouty arthritis, but certain clinical features can aid in its diagnosis. However, confirmatory diagnosis is established by microscopic analysis of the aspirated joint fluid, which reveals negatively birefringent MSU crystals. Joint aspiration is a technically challenging invasive procedure, especially in an acute inflammatory episode involving a small joint. Moreover, it carries the risk of bleeding, infection and can be negative in about 25% of patients.[9]
Multiple imaging techniques, such as radiographs, sonography, magnetic resonance imaging (MRI), and single energy CT, have been used for diagnosis of gout; however, the studies have suggested low sensitivity and specificity to diagnose the presence of urate crystals.[18] DECT helps to address this issue. The use of two X-ray tubes with different energy levels helps in characterization of compounds with different molecular weights;[19] in this mechanism, it differentiates crystals based on the atomic weight and electron density which changes X-ray absorption at the different energies.[20] Two different tubes at peak kilovoltage of 80 and 150 kVp are used to scan the area of interest, and an image is created which is viewed on a multimodality workstation. For dual source energy scanners, the two tubes operate simultaneously. Our scanner operates tubes at 80 kVp at 100 mA and 150 kVp at 67 mA, but the values may vary depending on the patient characteristics. Separate detector layers allow simultaneous data acquisition that helps in superior spectral contrast acquisition. On the contrary, for single source scanners, the tube operates at different energies with a small time gap of <0.5 msec for fast kVp switching between 140 and 80 kVp in a single acquisition. Optimized spectral filtration (tin filtration) is not provided in this setup.[20]
Postprocessing takes place using a two-material decomposition algorithm, which distinctively separates and color codes calcium and urate crystals separately owing to their different molecular weights. The uric acid crystals are color-coded in green, different from calcium and bone marrow - cortical bone is coded blue, medullary bone is coded pink.[21] The presence of color-coded urate crystals in joints and/or periarticular tissues is considered a positive scan [Figure 1].
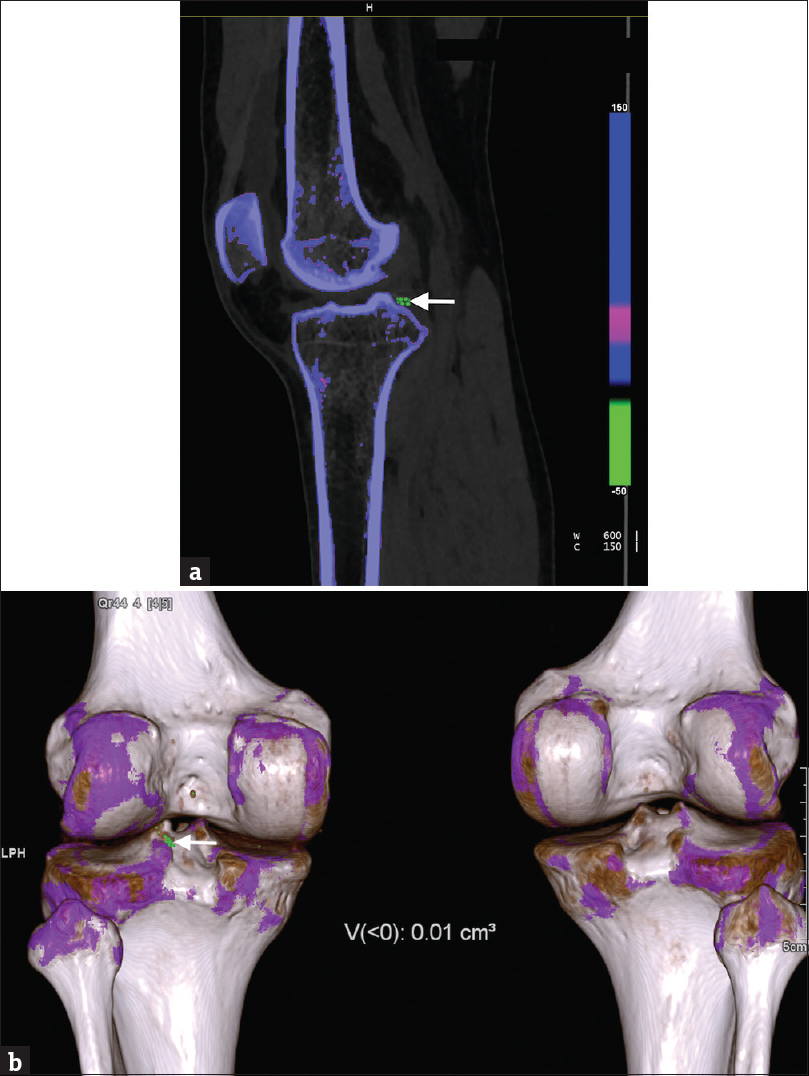
- (a) A 28-year-old man with gout depicting monosodium urate crystal deposition in the left knee joint on dual energy computed tomography. A cross-sectional (sagittal) color-coded image of the left knee joint using multiplanar reconstruction demonstrates presence of urate deposits (green) in periarticular soft tissue (arrow). The uric acid deposits appear different from osseous structures containing calcium (blue). (b) Volume-rendered color-coded image of both the knee joints depicts presence of monosodium urate crystals in periarticular soft tissue of the left knee joint (arrow). The automated software also calculates the volume of the crystals which was 0.01 cm3 in this patient (as mentioned in figure). The volume-rendered images depict the anatomic relationship between the uric acid deposits and the osseous structures (blue and white).
Once urate crystals are recognized on DECT, automated volume software can be used for quantification of crystal burden in that particular tophus.[16] This function provides additional utility as it can be used to monitor a patient's response to treatment. Choe et al.,[16] in their study proved the high accuracy and reliability of the automated software to determine the volume as compared to physical measurements. They showed 84% sensitivity and 93% specificity of DECT for diagnosis of gout and its high reproducibility for volume measurements.
Multiple studies have pointed to high sensitivity and specificity of DECT in diagnosis of gout.[9] Bongartz et al.,[21] in 2015 conducted a study to assess the same and its potential role in clinical decision making beyond joint aspiration. They concluded a 0.90 and 0.83 sensitivity and specificity, thus depicting a good diagnostic accuracy. Glazebrook et al.,[22] examined 94 patients to estimate the utility of DECT in diagnosis of gout. Two musculoskeletal radiologists with different years of experience examined the images. They reported a sensitivity of 100% for both the readers and a specificity of 89% and 79% for both the readers with near-perfect agreement between the readers. However, the utility of DECT in diagnosis of gout in early stages still remains debatable with studies showing variable results.[2324] Dalbeth et al.,[24] compared the frequency and volume of DECT urate deposits in gout patients depending on the stage of disease. They found increased volume of deposits in later stages; however, the study did not discuss the sensitivity or specificity of DECT for urate crystal detection.
Thus, DECT plays an important role in certain acute-care settings when it is difficult to differentiate an acute gout attack from that of other clinically similar pathologies. Detection of urate crystals on DECT can help in early diagnosis and thus early management of such patients. However, DECT has low sensitivity in the diagnosis of recent onset gout.[21]
BONE MARROW EVALUATION
MRI has been used as a modality of choice for detection of bone marrow edema. The affected areas show altered signal intensity hypointense on T1-weighted images and hyperintense on T2-weighted and short tau inversion recovery sequences.[25] This is referred to as the “bone bruise pattern” of bone edema.[26] MRI has certain limitations in an emergency setting such as long acquisition times and painful patient positioning. Conventional CT, on the other hand, is not able to detect the bone bruise pattern because of its limited ability to assess the bone marrow.[27]
DECT by virtue of its VNCa technique has been shown to be a useful tool in assessing bone marrow edema. VNCa is a type of material decomposition technique that helps in superior material differentiation. The edema detected can be viewed as gray-scale overlay, color-coded pattern, or three dimensional depending on availability of software and user convenience [Figure 2].[27] In our scanner, we use protocols depending on the area of interest to detect bone marrow edema. For spinal imaging, we use tubes at 90 kVp at 220 mA and 150 kVp at 140 mA, and for extremities, we use 80 kVp at 150 mA and 150 kVp at 100 mA.
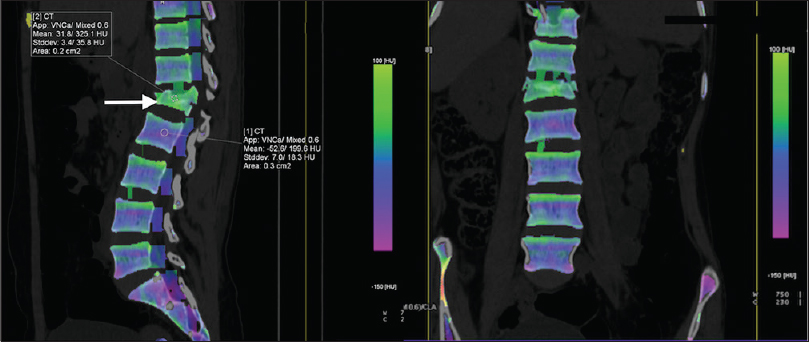
- An 18-year-old female with vertebral compression fracture and bone edema. Cross-sectional (sagittal and coronal) color-coded three-material decomposition (water, fat and calcium) images obtained using virtual noncalcium technique show collapse of L1 vertebral body with presence of bone marrow edema (arrow) as high attenuation area appearing dense green. The region of interest (arrow) shows high mean HU value as compared to the normal (as indicated in L2 vertebra), indicating bone edema.
Wang et al.,[27] in their study assessed the use of this technique for detection of bone marrow edema in vertebral compression fractures. Their results proved that DECT was able to detect marrow edema in collapsed vertebral bodies, as VNCa technique is able to subtract calcium from cancellous bones allowing assessment of the bone marrow. Pache et al.,[9] also used DECT VNCa technique for assessment of bone marrow edema in post-traumatic bone bruises of the knee. They reported sensitivity of 86.4% and specificity of 94.4% and 95.5% for the two observers as they compared the values against MRI. The authors suggested that lower sensitivity rate would improve with experience, as this was a relatively new technique. They advocated the use of color-coded maps for detection of edema.
Bone marrow involvement of spine in multiple myeloma was studied by Thomas et al.,[28] using DECT-VNCa technique. They reported improved sensitivity for detection of diffuse bone marrow infiltration of spine using CT, especially in the cases with high-grade infiltration.
However, DECT does have certain limitations.[15] Inadequate parameter optimization and extensive degenerative changes may make it difficult to detect bone edema. Furthermore, a certain mixture of red and yellow marrow may simulate edema, especially in young patients undergoing transition from red to yellow marrow.
Nonetheless, DECT has the potential to be an important imaging tool in emergency radiology, especially in patients with contraindications to MRI.
METAL ARTIFACT REDUCTION
The use of CT in patients with metal hardware remains an important diagnostic challenge. Table 1 lists some of the causes of artifact and the potential suggested techniques to reduce them.[29]

Newer methods such as projection interpolation, adaptive filtering methods, iterative reconstruction, and reconstruction with thicker slices have also been suggested to reduce the artifact.[30]
Recent literature has suggested the use of DECT in metal artifact reduction. Virtual monochromatic image spectrum is derived from DECT dataset, which is used to suppress the metallic artifacts.[31] Monochromatic DECT images are less affected by beam-hardening artifacts as they are generated from projection space data. The CT images are generated from any photon energy level between 40 and 140 keV. We operate our scanner at 100 kVp at 190 mA and 150 kVp at 100 mA for optimum image visualization. Set of images can be created that can help to study contrast differences between two adjacent structures.[32] On the contrary, single-energy CT uses a polychromatic X-ray spectrum. The lower energy photons are absorbed and the high energy photons from the detected X-ray beam thus resulting in dark streaks from metallic implants.[3334] Higher energies (i.e., 140 keV) are used to study the metal-bone or cement-bone interfaces, but it reduces contrast between materials. Soft-tissue detail is better appreciated on low energy images as the contrast-to-noise ratio is increased [Figure 3].[3536]
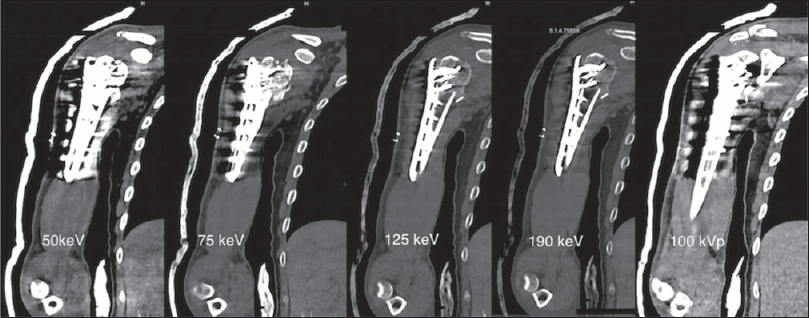
- A 30-year-old man with open reduction and internal fixation of the head and the neck of right humerus. Shown are coronal dual energy computed tomography images reconstructed using monoenergetic spectrum application on a multimodality workstation at monoenergies as mentioned on the respective images (left to right). Reduction of beam hardening artifact is seen at 125 and 190 keV as compared to simulated conventional CT image obtained at 100 kVp (on extreme right). The visibility of lateral chest wall, adjacent soft tissues is markedly improved at higher keV.
Bamberg et al.,[37] pointed that monochromatic images obtained at high energy level were superior from those at low energy level in quality and diagnostic value. Although complete elimination of artifact could not be achieved, the quality and diagnostic value improved by 49% and 44%, respectively. However, the radiation dose to the patient was not significantly altered as compared to single energy CT, thus proving the potential benefit of DECT in metallic artifact reduction with no increased radiation exposure. Bamberg et al.,[37] examined 31 patients with metallic implants to assess the performance and diagnostic value of DECT to reduce metal artifacts. Image quality and diagnostic value were rated superior by approximately 93% and 87%, respectively.
Sverzut et al.,[33] further highlighted the use of gem stone spectral imaging metal artifact reduction software (GSI-MARS) to improve soft tissue details at lower energy levels. However, GSI-MARS CT is more effective in reduction of artifacts from dense metal implants such as stainless steel and cobalt-chrome as compared to less dense metals such as titanium, thus signifying material composition as an important aspect.
There exists paucity of literature regarding direct comparison of dual source CT scanners with those of rapid kV switching; however, currently, the scanners with rapid kV switching are considered more effective since those with dual source tend to lose spatial resolution.[21]
TENDONS AND LIGAMENTS
Visualization of collagenous structures is most commonly done on MRI whereas CT is primarily used for assessing the osseous anatomy. However, due to certain limitations of MRI as discussed before and limited soft-tissue assessment ability of CT imaging, there has been a recent interest to explore a potential role of DECT in assessing these soft tissue structures. Tendons and ligaments are composed of densely packed collagen, elastin, glycoprotein, and glycosaminoglycan[38] that render them spectral properties. The set of images obtained at two different energy levels can be made to undergo three-material collagen decomposition algorithm on a multimodality workstation.[39] However, the use of DECT in imaging of tendons and ligaments still remains debatable.[404142]
Deng et al.,[41] in 2008 assessed the feasibility of visualizing hand and foot tendons by DECT. They concluded that it offers a potentially new imaging method to visualize tendons and ligaments, as they were able to visualize the size, shape, and insertion of flexor and extensor tendons of the hand and feet. Some of the lesions such as thickening and adherence could also be depicted. It is important to note that radiation neutrality was maintained as compared to single energy CT. Sun et al.,[42] in 2008 studied the clinical application of DECT in assessment of ligaments knee and proposed that it is a potential tool to display the main ligaments such as anterior cruciate ligament (ACL), posterior cruciate ligament, patellar ligament, and fibular collateral ligament.
However, in the same year, Lohan et al.,[40] pointed out that DECT yielded multiplanar reformations inferior to those of single energy CT that had better signal-to-noise and contrast-to-noise ratio. In addition, the patients were exposed to higher radiation dose. Fickert et al.,[43] studied the utility of DECT to visualize ACL in swine models, which showed good depiction of tears but with lower sensitivity as compared to MRI.
On the basis of these contradictory studies, it is difficult to establish the true utility of DECT for visualization of tendons and ligaments. Besides, window width and level adjustments are required for proper imaging through collagen decomposition algorithm.[19] This necessitates the need for new prospective studies to establish the significance of this imaging technique. However, at our institution, we do not use DECT as a potential tool to visualize tendons and ligaments in symptomatic patients. MRI is still the preferred modality.
CONCLUSION
DECT provides promising applications and benefits in musculoskeletal imaging. Some early promising results are seen in detection of bone marrow edema, reduction of metallic artifact, and visualization of tendons and ligaments. However, its utility in detection of urate crystals is currently in use in clinical practice. Although MRI has been the modality of choice to assess the above-mentioned issues, patients with contraindications to MRI can benefit from this noninvasive technique. Some of the future applications include its potential use in arthrography, assessment of metastasis and bone densitometry.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2017/7/1/34/213658
REFERENCES
- Dual- and multi-energy CT: Principles, technical approaches, and clinical applications. Radiology. 2015;276:637-53.
- [Google Scholar]
- Latest Advances in Musculoskeletal Dual Energy Computed Tomography (DECT) Current Radiology Reports. 2015;3
- [Google Scholar]
- Dual-energy computed tomography of the knee, ankle, and foot: Noninvasive diagnosis of gout and quantification of monosodium urate in tendons and ligaments. Semin Musculoskelet Radiol. 2016;20:130-6.
- [Google Scholar]
- Clinical utility of dual-energy CT for gout diagnosis. Clin Imaging. 2015;39:880-5.
- [Google Scholar]
- Dual energy with dual source CT and kVp switching with single source CT: A comparison of dual energy performance. Proc SPIE. 2009;7258:72583R.
- [Google Scholar]
- 2015 Gout classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis. 2015;74:1789-98.
- [Google Scholar]
- Dual-energy CT virtual noncalcium technique: Detecting posttraumatic bone marrow lesions – Feasibility study. Radiology. 2010;256:617-24.
- [Google Scholar]
- Multiparametric evaluation of head and neck squamous cell carcinoma using a single-source dual-energy CT with Fast kVp switching: State of the art. Cancers (Basel). 2015;7:2201-16.
- [Google Scholar]
- Gouty arthritis: The diagnostic and therapeutic impact of dual-energy CT. Eur Radiol. 2016;26:3989-99.
- [Google Scholar]
- Dual energy versus single energy MDCT: Measurement of radiation dose using adult abdominal imaging protocols. Acad Radiol. 2009;16:1400-7.
- [Google Scholar]
- White Paper of the society of computed body tomography and magnetic resonance on dual-energy CT, Part 3: Vascular, cardiac, pulmonary, and musculoskeletal applications. J Comput Assist Tomogr. 2017;41:1-7.
- [Google Scholar]
- The role of the medical physicist in managing radiation dose and Communicating risk in CT. AJR Am J Roentgenol. 2016;206:1241-4.
- [Google Scholar]
- Dual energy computed tomography in tophaceous gout. Annals of the Rheumatic Diseases. 2009;68:1609-12.
- [Google Scholar]
- Dual-energy computed tomography has limited sensitivity for non-tophaceous gout: A comparison study with tophaceous gout. BMC Musculoskelet Disord. 2016;17:91.
- [Google Scholar]
- Energy-selective reconstructions in X-ray computerized tomography. Phys Med Biol. 1976;21:733-44.
- [Google Scholar]
- Dual-energy CT: A promising new technique for assessment of the musculoskeletal system. AJR Am J Roentgenol. 2012;199(5 Suppl):S78-86.
- [Google Scholar]
- Dual-energy CT in gout – A review of current concepts and applications. J Med Radiat Sci. 2017;64:41-51.
- [Google Scholar]
- Dual-energy CT for the diagnosis of gout: An accuracy and diagnostic yield study. Ann Rheum Dis. 2015;74:1072-7.
- [Google Scholar]
- Identification of intraarticular and periarticular uric acid crystals with dual-energy CT: Initial evaluation. Radiology. 2011;261:516-24.
- [Google Scholar]
- Urate crystal deposition in asymptomatic hyperuricaemia and symptomatic gout: A dual energy CT study. Ann Rheum Dis. 2015;74:908-11.
- [Google Scholar]
- Bone marrow edema in vertebral compression fractures: Detection with dual-energy CT. Radiology. 2013;269:525-33.
- [Google Scholar]
- Dual-energy CT: Virtual calcium subtraction for assessment of bone marrow involvement of the spine in multiple myeloma. AJR Am J Roentgenol. 2015;204:W324-31.
- [Google Scholar]
- Applications of dual-energy CT in emergency radiology. AJR Am J Roentgenol. 2014;202:W314-24.
- [Google Scholar]
- Dual-energy CT with single- and dual-source scanners: Current applications in evaluating the genitourinary tract. Radiographics. 2012;32:353-69.
- [Google Scholar]
- Reduction of metal artifact with dual-energy CT: Virtual monospectral imaging with fast kilovoltage switching and metal artifact reduction software. Semin Musculoskelet Radiol. 2015;19:446-55.
- [Google Scholar]
- Virtual monochromatic spectral imaging with fast kilovoltage switching: Reduction of metal artifacts at CT. Radiographics. 2013;33:573-83.
- [Google Scholar]
- Virtual monochromatic spectral imaging with fast kilovoltage switching: Improved image quality as compared with that obtained with conventional 120-kVp CT. Radiology. 2011;259:257-62.
- [Google Scholar]
- Metal artifact reduction by dual energy computed tomography using monoenergetic extrapolation. Eur Radiol. 2011;21:1424-9.
- [Google Scholar]
- Structure relates to elastic recoil and functional role in quadriceps tendon and patellar ligament. Micron. 2009;40:370-7.
- [Google Scholar]
- Material differentiation by dual energy CT: initial experience. European Radiology. 2006;17:1510-7.
- [Google Scholar]
- Does dual-energy CT of lower-extremity tendons incur penalties in patient radiation exposure or reduced multiplanar reconstruction image quality? AJR Am J Roentgenol. 2008;191:1386-90.
- [Google Scholar]
- Initial experience with visualizing hand and foot tendons by dual-energy computed tomography. Clin Imaging. 2009;33:384-9.
- [Google Scholar]
- An initial qualitative study of dual-energy CT in the knee ligaments. Surg Radiol Anat. 2008;30:443-7.
- [Google Scholar]
- Assessment of the diagnostic value of dual-energy CT and MRI in the detection of iatrogenically induced injuries of anterior cruciate ligament in a porcine model. Skeletal Radiol. 2013;42:411-7.
- [Google Scholar]






