Translate this page into:
The Potential Utility of Arterial Spin Labeling in Detecting and Localizing Posterior Circulation Occlusions in Every Day Practice: A Clinical Report of Selected Cases

*Corresponding author: Vivek Yedavalli, Department of Radiology, Johns Hopkins University, Baltimore, Maryland, United States. vyedava1@jhmi.edu
-
Received: ,
Accepted: ,
How to cite this article: Yedavalli V, Tong E. The potential utility of arterial spin labeling in detecting and localizing posterior circulation occlusions in every day practice: A clinical report of selected cases. J Clin Imaging Sci 2020;10:78.
Abstract
Arterial spin labeling (ASL) is a non-contrast, non-invasive method used for the evaluation of cerebral perfusion, which is now increasingly utilized in everyday clinical practice. As a marker of cerebral blood flow at the capillary level, it has particular utility in stroke assessment. One rarer stroke subtype with non-specific symptomatology that can lead to significant morbidity is the posterior circulation (PC) infarct. As with the more common anterior circulation infarcts, ASL has shown benefit in PC infarcts as well, but has not been extensively explored in the literature nor been directly compared to bolus perfusion techniques. This clinical report of selected cases shows the utility of ASL in localization and detection of PC infarcts both in conjunction with and in the absence of bolus perfusion.
Keywords
Stroke
Perfusion imaging
Arterial spin labeling
Dynamic susceptibility contrast
INTRODUCTION
Arterial spin labeling (ASL) is a method of assessing cerebral perfusion at the capillary level.[1] It is increasingly being used in clinical practice as a non-invasive, non-contrast sequence that can evaluate cerebral blood flow (CBF).[2] Several studies have noted the efficacy of ASL in detecting decreased perfusion within strokes and perfusion/diffusion mismatches and its good agreement with dynamic susceptibility contrast (DSC) bolus perfusion.[3,4]
One stroke subtype that can lead to significant morbidity and mortality is posterior circulation (PC) infarcts, which account for approximately 5–15% of cases.[5] Clinical presentations can vary with dizziness, limb weakness, and dysarthria seen most commonly.[6] In addition, vessel occlusion location plays a pivotal role in prognosis, with distal basilar infarcts being associated with the poorest outcomes.[6] Occlusions in the PC – which can lead to infarcts – are most often caused by thromboembolism related to either large vessel atherosclerosis or cardiac sources.[7] Therefore, with the non-specific symptomatology, potentially devastating consequences, and debate regarding invasive management of PC infarcts, neuroimaging, in particular magnetic resonance imaging (MRI), plays a key role in prompt detection.[7]
The potential role of ASL in detecting PC infarcts, however, remains an area of continued research. While the past studies have incorporated ASL or DSC bolus perfusion (DSC PWI) for PC infarcts into larger studies, none have exclusively explored both together. In this clinical report, we discuss selected cases which explore the promising role of ASL in detecting and localizing PC occlusion and infarcts both with and without DSC bolus perfusion.
MATERIAL AND METHODS
IRB approval was obtained for this retrospective report. We searched for MRI stroke cases performed on 1.5 or 3T from September 2018 to May 2020 within our radiology database which yielded 144 patients. Inclusion criteria consisted of patients with (a) diffusion weighting imaging (DWI) evidence of acute PC infarcts, (b) time of flight (TOF) or contrast-enhanced magnetic resonance angiography (MRA), (c) gradient echo (GRE), (d) ASL, and (e) DSC PWI. ASL parameters on both 1.5 and 3T were as follows: Repetition time (TR): 5500 ms; echo time (TE): 10.5 ms; number of slices: 24–40; label time 1500 ms; post-label delay time: 2000 ms; in-plane spatial resolution 3 mm, through-plane spatial resolution 4 mm; and scan time: 4–6 min. Based on these criteria, among the 144 cases, 36 were suspected of PC infarcts based on symptomatology and nine total patients were selected (six males and three females; 48–89 years of age). Among these nine patients, there were five proximal and four distal PC occlusion/infarct cases.
CASE SERIES
Proximal occlusions with DSC
Case 1
A 53-year-old male with a history of prior right cerebellar infarct, diabetes, and hypertension presented with headache, acute left extremity weakness and numbness, and abnormal gait. Physical examination revealed decreased sensation to pinprick along the left forearm and dysmetria. On prior imaging, the patient was known to have focal severe left V4 stenosis. Computed tomography perfusion (CTP) was unremarkable. Subsequent MR [Figure 1] performed revealed bilateral acute cerebellar infarcts, right greater than left, with diminished related enhancement in the left V4 segment on MRA. ASL exhibited increased intravascular signal within the high-grade stenotic segment. No clear perfusion abnormality was seen on Tmax. Notably, no abnormality was detected on GRE.

- A 53-year-old male found to have small wedge-shaped acute cerebellar infarcts (not shown). (a) TOF MRA images show partial occlusion and severe stenosis of the left V4 segment (white arrow). (b) ASL shows increased intravascular signal corresponding to the occlusion and stenosis (white arrow). (c) Tmax does not show any definite perfusion abnormality.
Case 2
A 64-year-old male without significant medical history presented with central vertigo. Physical examination revealed dysmetria. The patient was sent directly to MRI, which demonstrated an acute left PICA territory infarct [Figure 2] with lack of flow-related enhancement in the vertebral artery on MRA and intravascular clot noted in the distal PICA on GRE. Tmax delay was seen on DSC bolus perfusion. ASL showed corresponding decreased CBF in the infarct territory and increased intravascular signal of the proximal PICA.

- A 64-year-old male found to have an acute left PICA infarct. (a) DWI shows the extent of the left PICA infarct. (b and c) TOF MRA shows occlusion of the left V4 segment (red arrows). (d) ASL shows increased intravascular signal at or immediately distal to the occlusion (red arrow) in addition to decreased signal corresponding to the core infarct (yellow dashed circle). (e) GRE shows intravascular clot within the mid to distal left PICA (red arrow). (f) Tmax image shows a perfusion delay within the left PICA territory (red arrow).
Case 3
An 89-year-old female with a history of hypertension and hypothyroidism presented with dizziness and postural instability after neck manipulation 10 days prior. Physical examination revealed left-sided dysmetria. Non-contrast computed tomography (NCCT) was negative. MR showed acute left cerebellar infarcts with intravascular clot on GRE and occlusion of the left vertebral artery on MRA [Figure 3]. Tmax delay was seen in the left PICA territory. ASL showed decreased CBF corresponding to the region of the infarct.

- An 89-year-old female with an acute left cerebellar and medullary infarct. (a) DWI shows infarcts within the left posterior medulla and cerebellum (white arrow). (b) TOF MRA shows occlusion of the left v4 segment (white arrow). (c) GRE shows corresponding intravascular clot (yellow arrow). (d and e) ASL shows increased intravascular signal within the occluded vessel (red arrow). (f) Tmax shows perfusion delay within the visualized left cerebellum.
There is increased intravascular signal in the left vertebral artery occlusion, corresponding to the clot seen on GRE and in the distal left PICA, suggesting another downstream thrombus, which was not apparent on other sequences.
Case 4
A 68-year-old male with a history of metastatic nasopharyngeal carcinoma and hypertension presented with acute onset vertigo and nausea. NCCT showed a new hypodensity within the right inferior cerebellum that favored metastasis. Physical examination showed left dysmetria. MR revealed an acute right cerebellar infarct with both intravascular clot and occlusion of the right vertebral artery and distal right PICA [Figure 4]. Tmax delay was in the right PICA distribution, corresponding to the areas with restricted diffusion. ASL showed CBF corresponding to the region of the infarct as well as increased intravascular signal just proximal to the right PICA thrombus seen on GRE.

- A 68-year-old male an acute right PICA infarct. (a) DWI shows the extent of the infarct. (b) TOF MRA shows occlusion of the right V4 segment (red arrow). (c) ASL shows increased intravascular signal immediately proximal to the occlusion (yellow arrow). (d and e) GRE shows intravascular clot corresponding to the occlusion (blue arrow). (f) Tmax shows a corresponding right cerebellar perfusion delay.
Distal occlusions with DSC
Case 5
A 48-year-old female presented with transient right visual field cut. NCCT was unremarkable. Physical examination, however, demonstrated intact visual fields. Subsequent MRI revealed small acute left occipital infarcts with a faint susceptibility corresponding to the medial occipital branch of the left PCA on GRE, representing tiny clot [Figure 5]. Lack of flow-related enhancement was noted within this small branch on MRA, although not definitive. Tmax delay was seen in the region of the infarcts. ASL demonstrated both decreased CBF corresponding to the left occipital infarct and focally increased signal proximal to the suspected medial occipital branch occlusion.
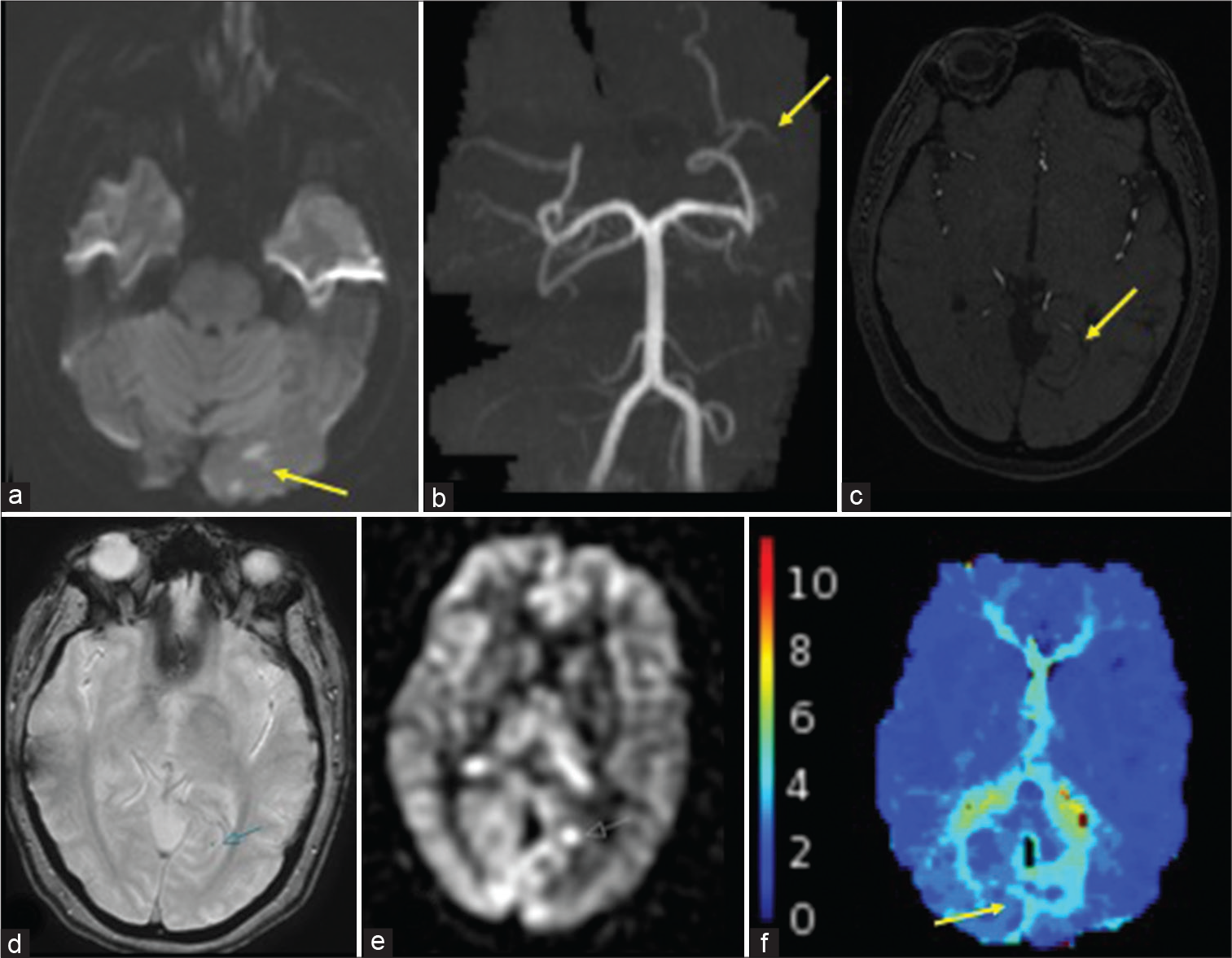
- A 48-year-old female with the left occipital infarcts. (a) DWI shows multiple infarcts within the left occipital lobe (yellow arrow). (b and c) TOF MRA shows the occlusion of a distal left P3 branch (yellow arrows). (d) GRE shows a punctate focus thought to represent intravascular clot (blue arrow). (e) ASL shows a focus of increased signal at the occlusion site (white arrow). (f) Tmax delay is noted in the medial left occipital lobe (yellow arrow).
Case 6
A 79-year-old male with hypertension and lung adenocarcinoma presented with the right extremity weakness and slurred speech. Physical examination was negative. NCCT noted a hypodensity within the left dorsolateral thalamus concerning for a lacunar infarct. MRI was then performed for further evaluation, which revealed an acute infarct corresponding to the CT hypodensity. Intravascular clot and lack of flow-related enhancement were both detected and consistent with partial occlusion of the parietooccipital branch of the left PCA [Figure 6]. Tmax delay was seen in the left occipital lobe. ASL also revealed decreased CBF within the left occipital lobe in addition to increased intravascular signal distal to the partial occlusion.
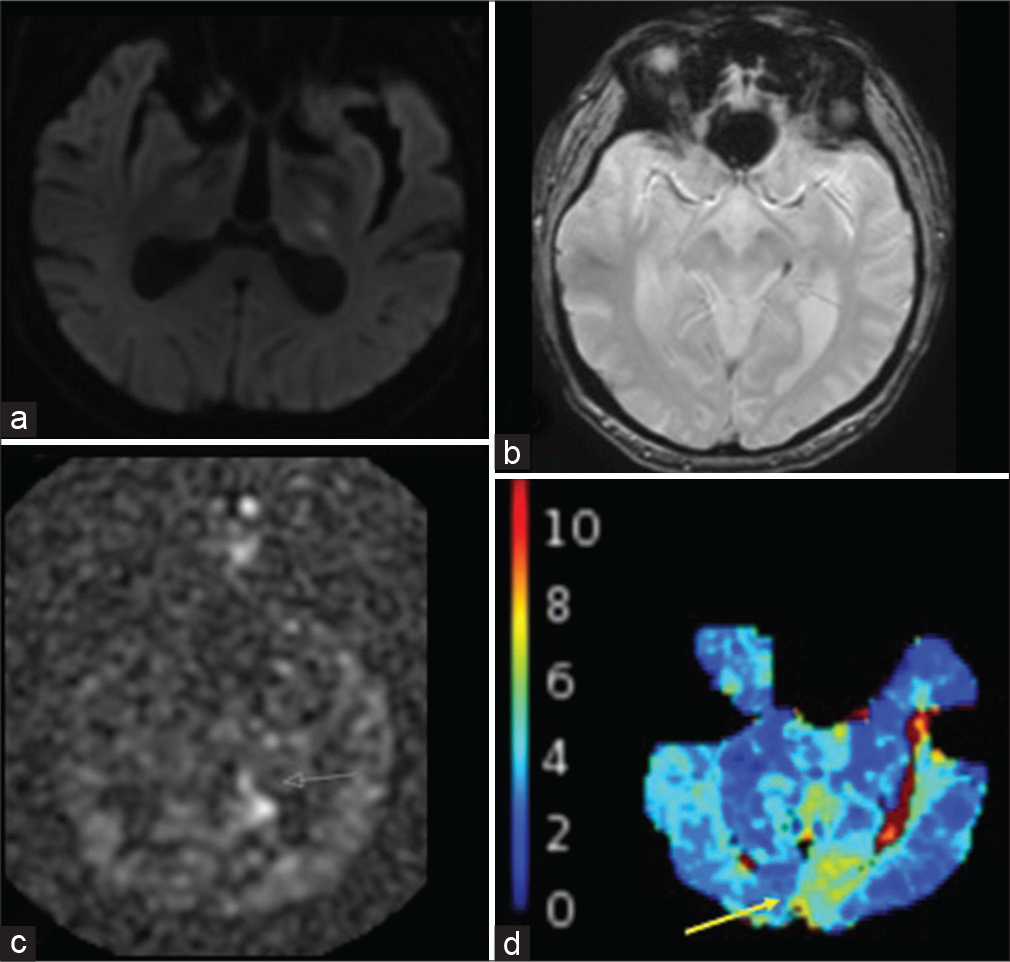
- A 79-year-old male with the left thalamic infarcts. (a) DWI shows the punctate infarcts within the left dorsolateral thalamus. (b) GRE shows intravascular clot within the left P2 segment (white arrow). (c) ASL shows increased intravascular signal distal to the left P2 occlusion (white arrow). (d) Tmax delay is demonstrated within the left medial occipital lobe in the left PCA territory (yellow arrow).
ASL without DSC
Case 7
A 59-year-old male with a history of coronary artery disease and hypertension initially presented with the left arm weakness and was diagnosed as a right MCA infarct. The patient’s hospital course was complicated by hemorrhagic transformation and superimposed infection outside of the infarcted region. A month after the initial admission, the patient presented with acute altered mental status. Physical examination showed worsening extremity weakness. NCCT was negative. MRI, however, demonstrated bilateral inferior cerebellar acute infarcts [Figure 7]. Complete occlusion of the right vertebral artery and severe stenosis/partial occlusion of the left vertebral artery was noted. There was a suggestion of intravascular clot on GRE, however, motion degradation makes this finding difficult to definitively appreciate. DSC was of limited diagnostic value due to the motion degradation, but showed delay in area corresponding to the infarcts. However, ASL demonstrated increased intravascular signal within the right vertebral artery proximal to the occlusion and distal to the left vertebral artery corresponding to the partial occlusion.
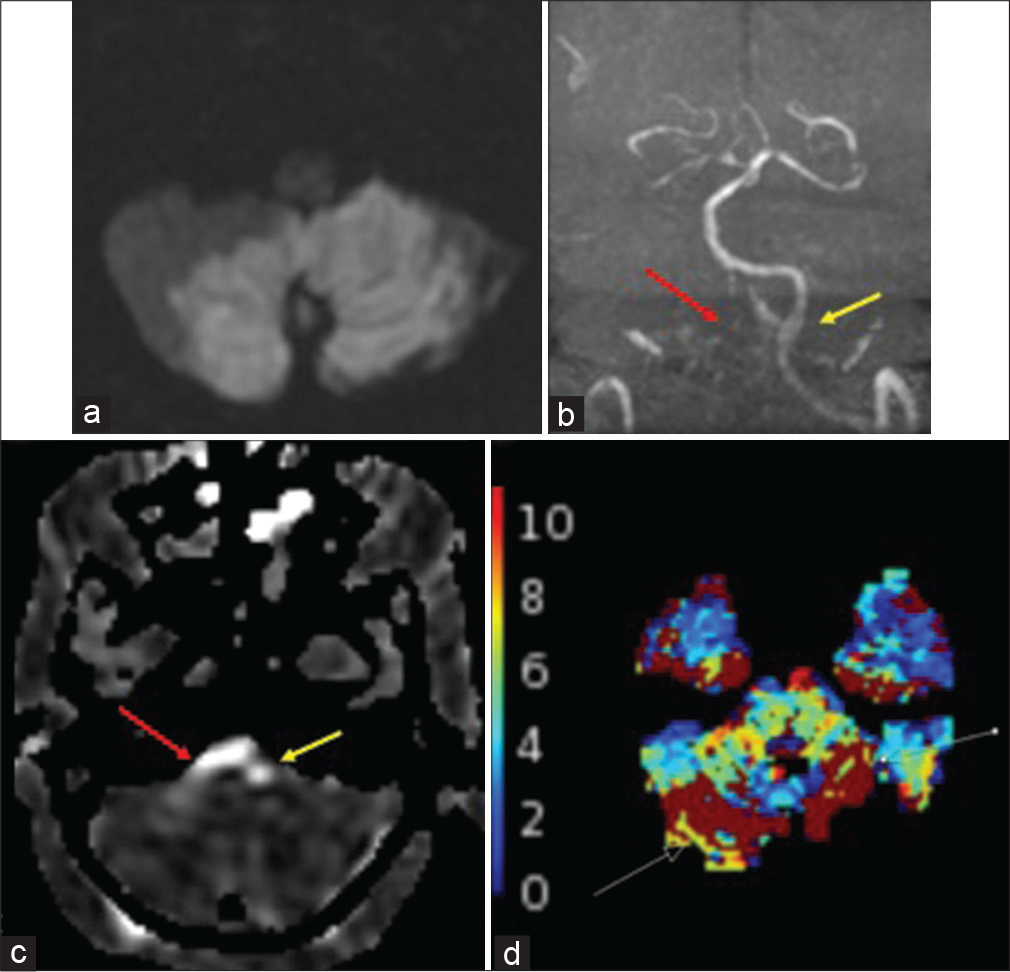
- A 59-year-old male with infratentorial infarcts. (a) DWI shows infarcts involving the cerebellar hemispheres bilaterally and vermis. (b) TOF MRA shows compete and partial occlusions (red arrow) of the right and left (yellow arrow) V4 segments, respectively. (c)ASL shows increased intravascular signal within the bilateral V4 segments (right V4, red arrow; left V4, yellow arrow). (d) Tmax was motion degraded and partially evaluated the infratentorial brain, limiting diagnostic evaluation, but showed questionable abnormality in the infarcted regions (white arrows).
Case 8
A 60-year-old female with a history of hypertension, diabetes, and endometrial adenocarcinoma presented with acute right extremity weakness. Physical examination noted semantic aphasia with retained repetition and reading (thalamic aphasia) and ataxia. NCCT showed a hypodensity within the left thalamus involving the posterior limb of the internal capsule. MRI then demonstrated acute left thalamic and hippocampal tail infarcts [Figure 8]. Intravascular clot was noted in the left P1 segment of the PCA on GRE, corresponding to the abrupt cutoff on MRA. ASL showed decreased CBF in the left PCA territory with increased intravascular signal noted proximal and distal to the occlusion, suggestive of a partially occlusive thrombus. DSC was not performed due to patient discomfort.
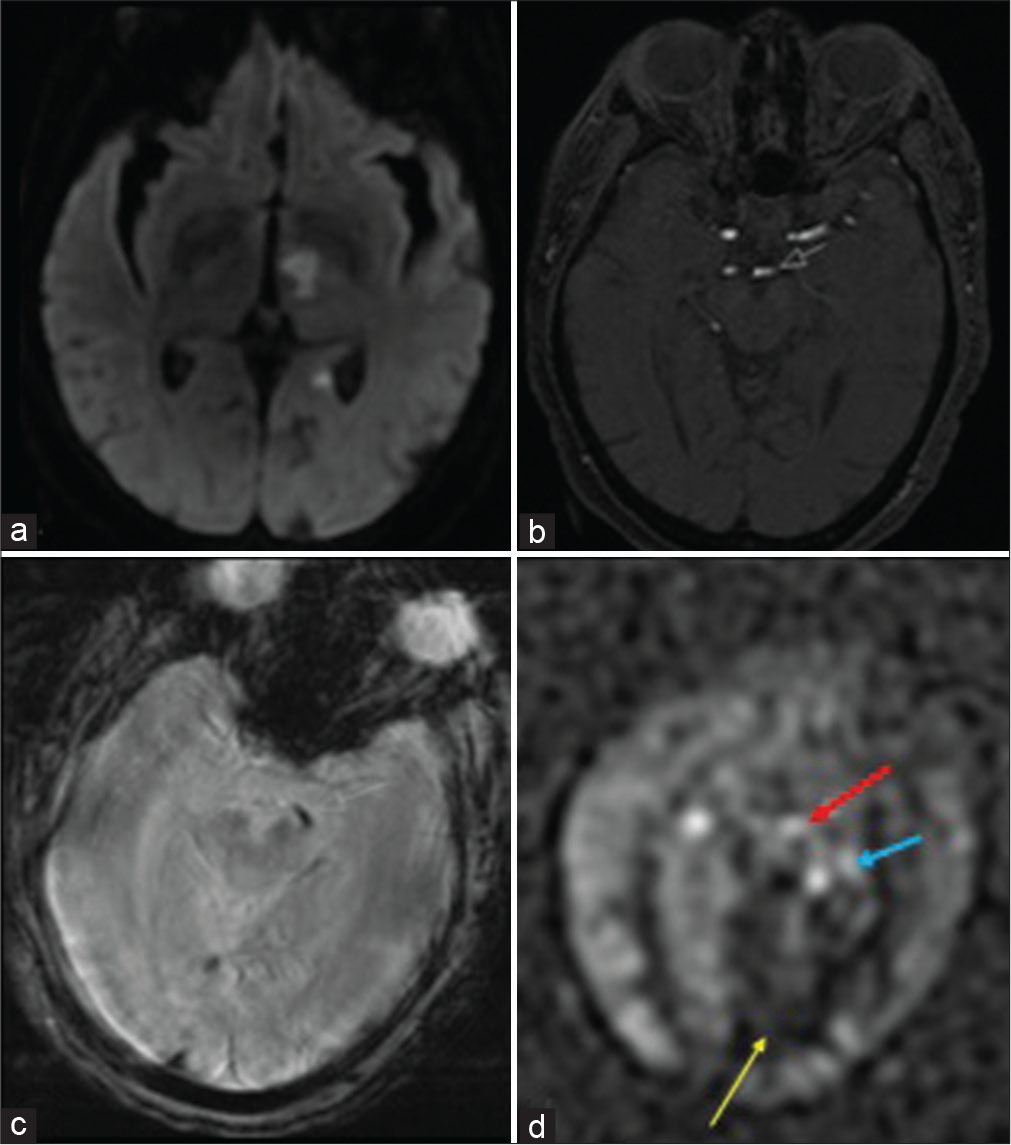
- A 60-year-old female with acute kidney injury who was found to have a left PCA infarct. (a) DWI shows acute infarcts in the left anterior thalamus with involvement of the posterior limb of the internal capsule as well as the left hippocampal tail. (b) TOF MRA shows an abrupt cutoff representing the occlusion. (c) GRE shows corresponding intravascular clot. (d) ASL shows increased intravascular signal both proximal and distal to the left P1 occlusion (red/white and blue arrows respectively) in addition to decreased signal in the left medial occipital lobe (yellow arrow).
Case 9
A 54-year-old male with a history of hypertension presented initially with quadriparesis and was subsequently found to have a basilar thrombosis. She underwent a thrombectomy with full restoration of flow (TICI 3). On the following day, the patient acutely presented with vision loss. Physical examination was remarkable for the right homonymous hemianopsia. CT was positive for the left PCA infarct. Due to acute kidney injury, MRI was performed without IV contrast and revealed an acute left PCA territory infarct with intravascular clot and occlusion of the P2 segment of the left PCA on GRE [Figure 9]. ASL showed increased signal within the left PCA immediately proximal to the occlusion.
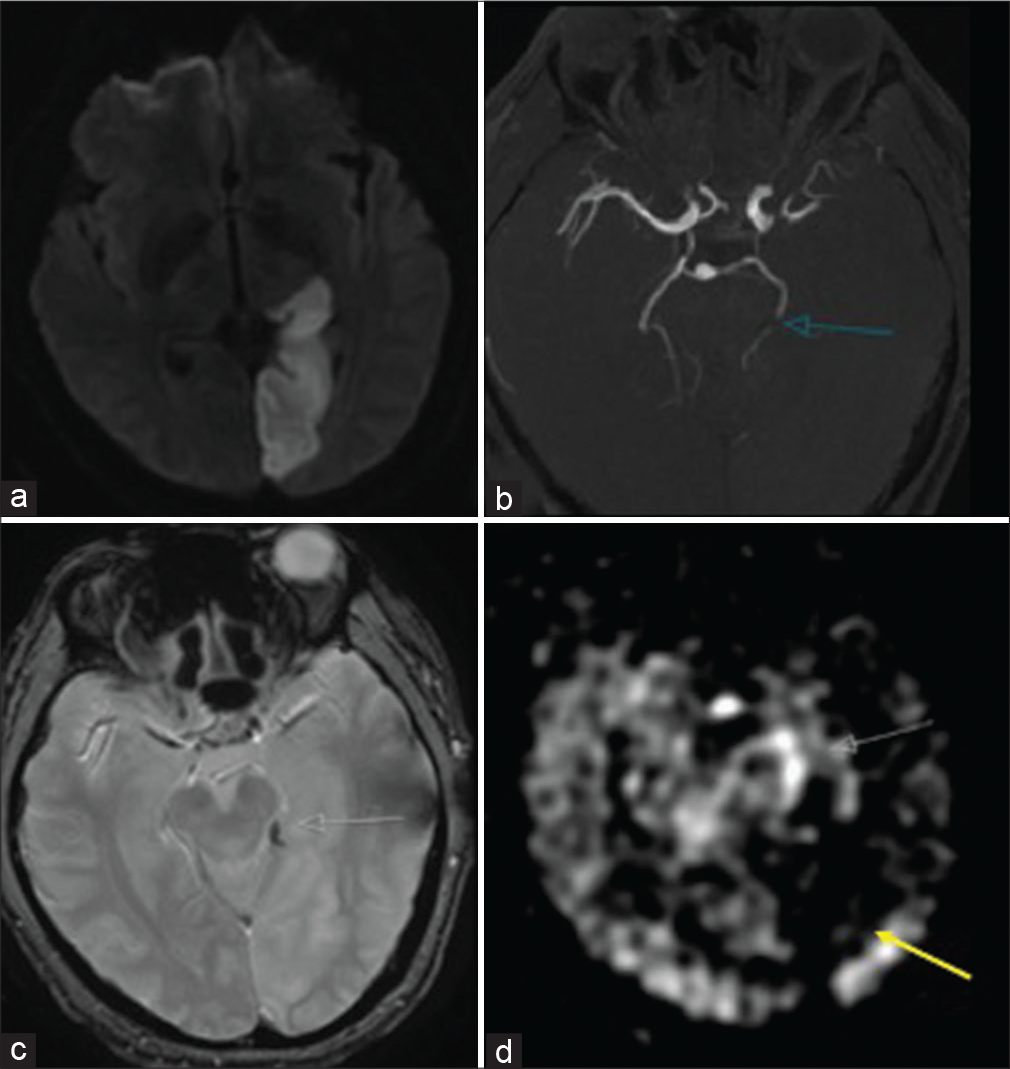
- A 54-year-old male with acute kidney injury found to have a left PCA infarct. (a) DWI shows the extent of the left PCA infarct. (b) TOF MRA shows the abrupt cutoff of the left P2 segment compatible with occlusion (blue arrow). (c) GRE shows corresponding intravascular clot (white arrow). (d) ASL shows increased intravascular signal proximal to the left P2 occlusion (white arrow) with decreased signal in the left medial occipital lobe (yellow arrow).
DISCUSSION
This case series describes the potential utility of ASL in detecting PC vessel occlusion and infarcts for everyday workflow. As a non-invasive and non-contrast method, ASL is especially useful in patients with kidney dysfunction, pediatric patients, gadolinium contrast allergy, and cases where DSC perfusion cannot be performed.[1,8]
However, ASL has some important drawbacks which must be considered. At present, ASL can be altered significantly based on technical and physiological variables. For example, there are a number of subtypes of ASL leading to variance in acquisition, with the pseudocontinuous form now recognized as the preferred method.[9] Physiological variables such as cardiac output can also change the acquisition parameters of ASL.[9] These are important parameters to keep in mind when assessing ASL. Clinically, the main limitations of ASL are the decreased signal-to-noise ratio and longer acquisition times compared to DSC.[1]
Many studies have shown the added value of ASL in stroke detection but its role in PC occlusion and infarcts has not been exhaustively explored. The landmark trials in stroke focused on anterior circulation large vessel occlusion for potential thrombectomy, but PC occlusion still warrants further exploration. Yoo et al. were one of the first groups to use the “bright vessel appearance” on ASL to detect occlusion with 94% sensitivity – compared with using susceptibility artifact on GRE with 66% sensitivity – when MRA was used as the gold standard.[3] They postulated that this bright vessel appearance is a form of arterial transit artifact (ATA) which can be seen proximal or distal to the occlusion.[3] Increased signal intensity proximally is attributed to slow flow within the occluded vessel causing accumulation of tagged blood, while similar findings seen distally are due to delayed filling from collateral vessels.[3] However, while Yoo et al.’s cohort did include PC occlusions, it did not explore it separately nor did it includes bolus perfusion as a comparison in their analysis. Our series shows multiple examples of the bright vessel appearance – either proximally or distally – in detecting PC occlusion, showing concordance with Yoo et al.’s analysis.
Cereda et al. explored both CTP and DSC in detecting PC occlusion, where they deemed both perfusion modalities combined to have 85% accuracy.[10] However, their cohort did not include ASL in their analysis. To date, no studies have compared ASL and DSC in detecting PC occlusion and infarct.
This clinical report demonstrates the additive benefit of using ASL in corroboration with DSC or using ASL independently when DSC cannot be performed. We show six total cases of PC occlusions (both proximal and distal) which corroborated findings seen on DSC. In some cases, the ATA on ASL helped identify and confirmed that the susceptibility artifact on GRE corresponds to a thrombus. We also show the independent utility of ASL in both detection and localization of PC occlusion and infarct in three cases where DSC could not be performed. While prior studies have investigated ASL and DSC separately for detecting PC occlusion with strong potential for both, none have done so in comparing both and assessing the possible increased sensitivity of using both in conjunction. Our selected cases suggest that ASL could be a highly sensitive diagnostic marker, both independently and with DSC, in localizing and detecting PC occlusion and infarct. Therefore, it necessitates the importance of future studies in using both modalities in analysis in detecting these types of strokes.
CONCLUSION
ASL is a noninvasive MR perfusion sequence that is increasingly being used in everyday practice. It adds benefit as a noncontrast and repeatable method with utility in stroke imaging. One such stroke subtype that ASL can also have utility in is PC infarcts. This clinical report highlights cases where ASL can detect and localize PC occlusions and infarcts in conjunction with and in the absence of bolus perfusion. This series suggests that ASL’s role in PC occlusion and infarct is worth exploring in larger studies.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Arterial spin label imaging of acute ischemic stroke and transient ischemic attack. Neuroimaging Clin N Am. 2011;21:285-301.
- [CrossRef] [PubMed] [Google Scholar]
- Multi-delay multi-parametric arterial spin-labeled perfusion MRI in acute ischemic stroke-comparison with dynamic susceptibility contrast enhanced perfusion imaging. Neuroimage Clin. 2013;3:1-7.
- [CrossRef] [PubMed] [Google Scholar]
- Bright vessel appearance on arterial spin labeling MRI for localizing arterial occlusion in acute ischemic stroke. Stroke. 2015;46:564-7.
- [CrossRef] [PubMed] [Google Scholar]
- The value of arterial spin-labeled perfusion imaging in acute ischemic stroke: Comparison with dynamic susceptibility contrast-enhanced MRI. Stroke. 2012;43:1018-24.
- [CrossRef] [PubMed] [Google Scholar]
- Predicting outcome after acute basilar artery occlusion based on admission characteristics. Neurology. 2012;78:1058-63.
- [CrossRef] [PubMed] [Google Scholar]
- Symptoms and signs of posterior circulation ischemia in the New England medical center posterior circulation registry. Arch Neurol. 2012;69:346-51.
- [CrossRef] [PubMed] [Google Scholar]
- Ischemic posterior circulation stroke: A review of anatomy, clinical presentations, diagnosis, and current management. Front Neurol. 2014;5:30.
- [CrossRef] [PubMed] [Google Scholar]
- Arterial spin labeling perfusion of the brain: Emerging clinical applications. Radiology. 2016;281:337-56.
- [CrossRef] [PubMed] [Google Scholar]
- Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: A consensus of the ISMRM perfusion study group and the European consortium for ASL in dementia. Magn Reson Med. 2015;73:102-16.
- [CrossRef] [PubMed] [Google Scholar]
- Abstract TP72: Localization of Large Vessel Occlusion (LVO) Stroke in the Posterior Circulation Based on Perfusion Imaging. 2020. Stroke. Available from: https://www.ahajournals.org/doi/abs/10.1161/Str.51.suppl_1.tp72 [Last accessed on 2020 May 21]
- [Google Scholar]






