Translate this page into:
T1 hyperintensity in the spinal cord: A diagnostic marker of amyotrophic lateral sclerosis?

*Corresponding author: Saravana Kumar Swaminathan, Department of Neuroradiology, National Neuroscience Institute, Singapore. saravana.kumar.s@singhealth.com.sg
-
Received: ,
Accepted: ,
How to cite this article: Pai V, Trivedi CR, Pai B, Swaminathan SK. T1 hyperintensity in the spinal cord: A diagnostic marker of amyotrophic lateral sclerosis?. J Clin Imaging Sci 2022;12:20.
Abstract
Amyotrophic Lateral Sclerosis (ALS) is a rare, devastating motor neuron disease characterized by the degeneration of upper and lower motor neurons causing muscular weakness, paralysis, and eventual death. MRI plays a supportive role in the diagnosis; its primary role is to exclude other clinical mimics. Some of the imaging features associated with ALS include hypointense signal along the motor cortices on susceptibility or T2*-weighted imaging and hyperintensity along the corticospinal tracts (CST) within the cerebral hemispheres, brainstem, and spinal cord on the T2 weighted imaging. In this report, we discuss the value of T1 hyperintensity along the CST, especially in the spinal cord.
Keywords
Amyotrophic lateral sclerosis
Corticospinal tracts
INTRODUCTION
Amyotrophic Lateral Sclerosis (ALS) is a progressive, potentially fatal, neurodegenerative disorder.[1-3] It is characterized by insidiously worsening voluntary muscle weakness because of degenerating upper and lower motor neurons, eventually leading to respiratory paralysis and death.[3-5] The loss of neuromuscular connection and axonal retraction culminating in neuronal cell death are considered pathological hallmarks of ALS.[6] The annual incidence of ALS is approximately 4–8 per 100,000 with the vulnerable age ranging from 45–75 years.[6] Males are affected more frequently (1.2–1.5:1).[5] ALS is predominantly sporadic, but a familial form of ALS has also been identified accounting for 10–15% of cases.[1,4,5] The diagnosis is made using a combination of clinical and electromyography (EMG) findings; MRI is a supportive diagnostic modality that helps to rule out other mimics of this condition. Changes along the motor cortices and the corticospinal tracts (CST) have been described, however, literature on the changes in the T1-weighted imaging, especially in the spinal cord is sparse. In this report, we discuss the MRI findings in a young patient with clinically and radiologically suspected ALS with emphasis on T1-weighted imaging.
CASE REPORT
A 38-year-old male patient presented with the complaints of slowly progressive weakness of the upper and lower extremities over a period of 12 months. This was associated with weight loss. There was no relevant family history, past medical history, or toxic exposure. Clinical examination revealed diffuse muscular wasting, hypertonia, brisk reflexes, and ankle clonus along with positive Hoffman and Babinski signs. These findings suggested a mixed upper and lower motor neuron disease. MRI of the brain and cervical spine was performed for the evaluation of underlying organic or structural etiologies to account for the clinical presentation.
3T-MRI of the brain revealed symmetric susceptibility artifacts on the susceptibility weighted imaging (SWI) sequence along the posterior aspects of the precentral gyri [Figure 1]. Linear, symmetric, contiguous T1, and T2 hyperintense signal abnormality was also observed extending from the precentral gyri to the cerebral peduncles, across the centrum semiovale, corona radiata, and posterior limbs of the internal capsules i.e., the expected course of the corticospinal tracts (CST) [Figure 2]. These signal changes were not associated with restricted diffusion. On evaluation of the cervical spine, further extension of the T1 hyperintense signal abnormality was noted along the anterolateral aspects of the cervical cord along the expected course of the CST, however, there was no convincing T2 signal abnormality [Figure 3]. No cortical or spinal cord volume loss was detected. No organic intracranial lesion, mass effect, or spinal cord compression was identified. Considering the constellation of clinical and radiological findings, the diagnosis of Amyotrophic Lateral Sclerosis (ALS) was suggested. Unfortunately, the patient was lost to follow up.
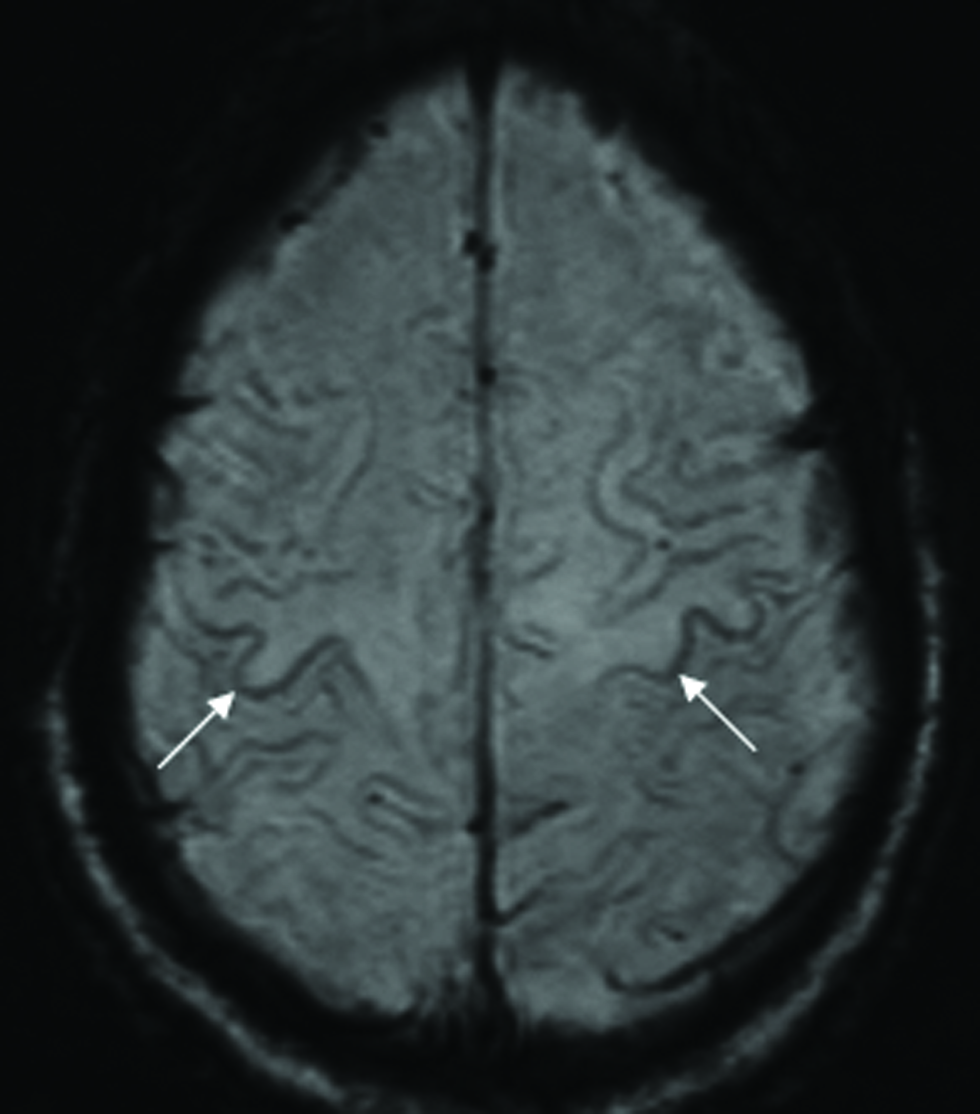
- MRI of the 38-year-old male patient who presented with slowly progressive weakness of the upper and lower extremities. Axial SWI sequence of the brain reveals symmetric gyriform susceptibility exclusively along the posterior cortex of the precentral gyrus in keeping with the “motor band sign.”
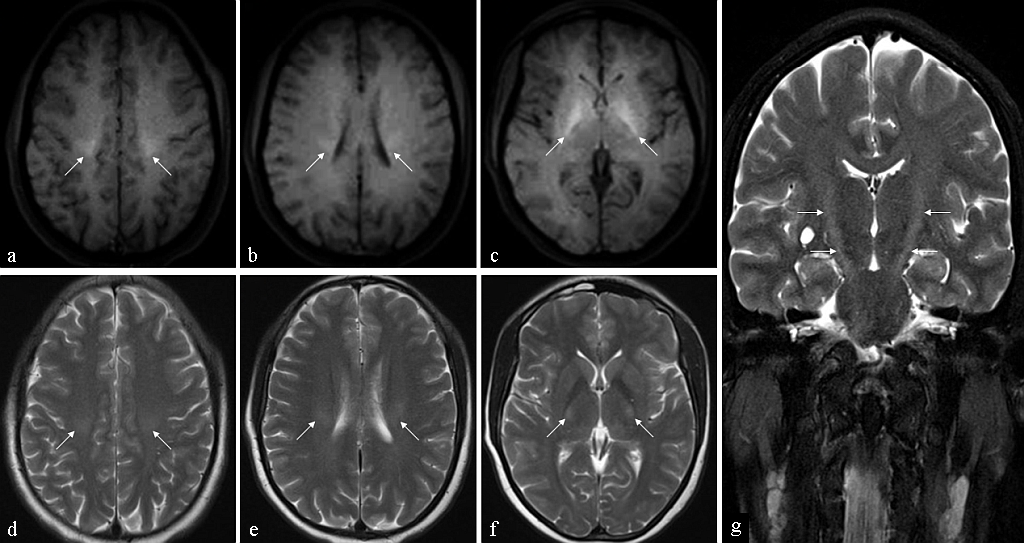
- MRI of the 38-year-old male patient who presented with slowly progressive weakness of the upper and lower extremities. Axial T1 (a–c) and axial T2 (d–f) images of the brain reveal symmetric hyperintensity in the centra semiovale (a and d), coronae radiatae (b and e) and the posterior limbs of the internal capsules (c and f), along the expected course of the CSTs. Coronal T2 image (g) reveals the entire extent of the signal abnormality.
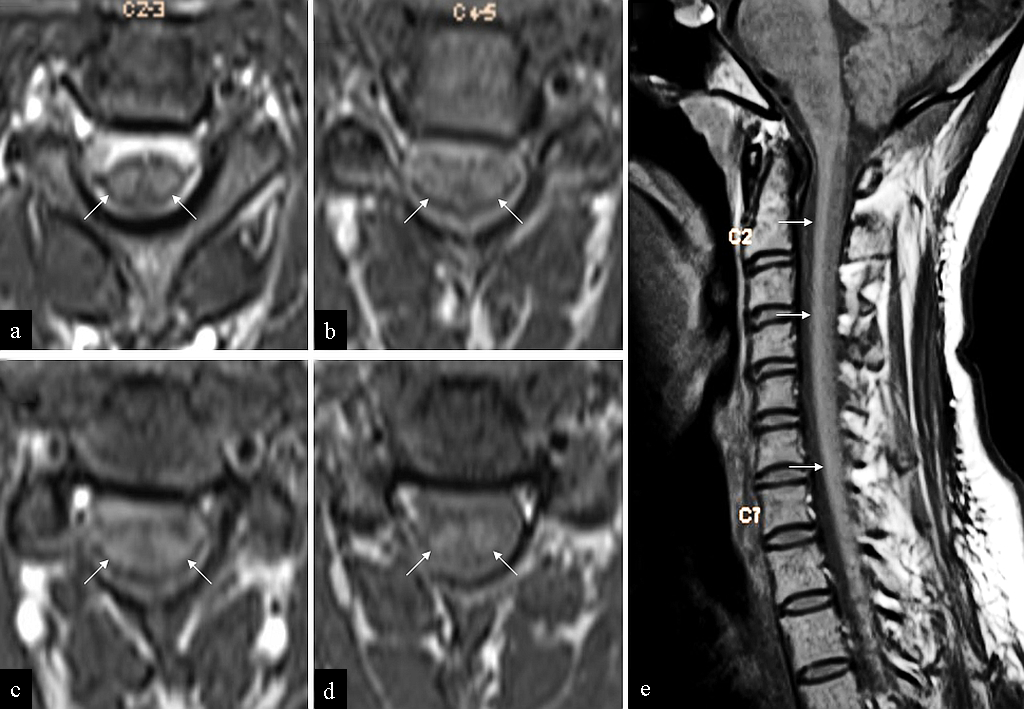
- MRI of the 38-year-old male patient who presented with slowly progressive weakness of the upper and lower extremities. Axial T1 images (a–d) of the cervical spine demonstrate symmetric T1 hyperintensity along the anterolateral columns of the cervical cord. Note the absence of cord swelling or atrophy. T1 hyperintensity within the perimedullary CSF is artifactual (attributed to “entry slice phenomenon”). Sagittal T1 (e) reveals the entire extent of the signal abnormality.
DISCUSSION
The diagnosis of ALS is based on a combination of clinical examination and electromyography (EMG). Clinical examination generally reveals features of upper motor neuron (UMN) signs including hyperreflexia, increased tone, and slow voluntary movements.[6] Babinski and Hoffman signs, in addition to emotional lability, have also been identified as UMN signs of ALS.[8] Lower motor neuron (LMN) involvement is characterized by atrophy of muscles and fasciculations.[6] Needle EMG is one of the fundamental diagnostic tests in the workup of ALS. Changes in acute denervation (positive sharp waves, fibrillation, and/or fasciculation potentials) and chronic neurogenic changes (reduced recruitment, rapidly firing and large amplitude, long duration motor unit potential) are indicative of ALS.[9]
Imaging plays a supportive role in the diagnosis of ALS. Conventional MRI is primarily used to exclude ALS mimics including other motor neuron diseases, neuroinflammatory diseases, skull base lesions, cervical spondylotic myelopathy, and other compressive spinal cord lesions.[1,10] Multiple advanced MRI techniques have been used to detect ALS and quantify the extent of the disease. However, these are beyond the scope of this case report.
At conventional MR imaging, hypointensity along the precentral cortex may be observed on T2*-weighted or susceptibility-weighted imaging (SWI), a finding termed the “motor band sign” (MBS).[10,11] The finding may be unilateral or bilateral and is thought to result from excessive deposition of iron, fibrillary gliosis, and/or macrophage infiltration.[10,12] Based on a study by Roeben et al., a total of 157 patients from an ALS cohort were evaluated for the presence of the MBS.[11] They observed MBS in 78% (7/9) of patients with SWI included in the MR imaging study but only in 5% of patients without SWI added to the imaging protocol.[11] Using this data, they suggested the use of SWI in patients with suspected ALS, given its higher resolution and sensitivity to susceptibility effects as compared to other sequences including T2*-weighted imaging.[11] The exact sensitivity of MBS is unknown. Reports suggest cortical hypointensity in the elderly as well as in patients suffering from other neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease.[11,12] However, the specific involvement of the motor cortices and consequent UMN findings have been thought to be more suggestive of ALS.[11,12] It must be acknowledged, however, that larger prospective trials are warranted to evaluate the incidence, sensitivity, and specificity of MBS as a marker of ALS and its role in prognostication.[12] Global volume loss, especially of the grey matter in the frontal and parietal lobes has also been described.[1]
Another finding in ALS is hyperintensity along the corticospinal tracts (CST) on T2-weighted and/or fluid-attenuated inversion recovery (FLAIR) sequences. The finding is commonly observed along the posterior limb of the internal capsules but may also involve contiguous portions of the centrum semiovale, cerebral peduncles, and ventral brainstem.[10,13] Pathophysiology of this signal abnormality is thought to be related to neuronal death, degeneration, and/or demyelination.[14] The signal changes may further extend caudally into the anterolateral columns of the spinal cord.[4] The anatomic course of the CST along the anterior paramedian (uncrossed fibers) and posterior portion of lateral funiculus (crossed fibers) forms the anatomic basis of this signal distribution in the spinal cord.[15] Atrophy of the cord may be seen with flattening of its anterior and lateral contours, due to loss of motor neuron bulk, imparting an inverted horseshoe appearance.[4,15]
These findings have been deemed non-specific and poor biomarkers for ALS. T2/FLAIR hyperintensity has been described along the CST in normal subjects when imaged in high field strengths (3.0 T).[13] Primary lateral sclerosis may result in similar imaging abnormality along the intracranial CST.[1,13] Wallerian degeneration, due to a remote insult, also demonstrates similar signal abnormality, though the cause is usually apparent and not frequently symmetric.[15] Other diseases such as inflammatory-demyelinating disorders, acute hypoglycemia, AIDS, syphilitic meningomyelitis, and deficiency of vitamin B12 can affect the CST, but with clinically distinct manifestations and other associated imaging features.[13,15]
An important, but seldom discussed MR finding in ALS, is abnormal hyperintense signals along the CST on T1-weighted images and T1-Magnetization Transfer Contrast (MTC) sequence.[16] MTC relies on longitudinal magnetization exchange between bound and free protons and helps provide an index of structural tissue integrity.[16,17] In a study by Carrara et al., hyperintensity on T1-MTC was detected along the intracranial CST and splenium of the corpus callosum in 16 of 43 patients with ALS (37.2% sensitivity), but not in the control subjects (100% specificity).[16]
Considering the preferential involvement of the CST, extending from the brain to the spinal cord, and the anatomic distribution along the CST in the anterolateral columns, it can be extrapolated that T1 hyperintensity may be observed in the spinal cord as well. However, to the best of our knowledge, intrinsic spinal cord T1 hyperintensity has been reported sparsely in literature. In a study, performed by Waragai et al. in 1997, T1 hyperintensity was noted in the anterolateral columns of the cervical cord in eight out of 14 patients. These findings were more pronounced from C4–C6 levels as compared to C1–C3 levels. Four out of the eight had similar signal abnormality in the lower cord in the lumbar region, presumably in the terminal cord/conus, as well. Two patients also had T1 and T2 hyperintensity along the expected course of the intracranial CST as well.[18] In this study, anterolateral cord T1 hyperintensity was seen more frequently than other known imaging signs of ALS such as motor cortex hypointensity, T1 and/or T2 hyperintensity of the intracranial CST, and T2 hyperintensity within the spinal cord.[18]
In our index patient, an MRI was performed on the clinical suspicion of ALS. We noted typical findings of ALS which included symmetric, gyriform susceptibility along the precentral gyrus in keeping with the motor band sign as well as symmetric, linear T2 hyperintensity extending across the expected course of the intracranial CSTs. In addition, we also observed corresponding T1 hyperintensity along the intracranial CST. MRI of the cervical spine revealed symmetric T1 hyperintensity in the anterolateral columns throughout the cervical cord. There was no cerebral cortical or cord atrophy.
The cause of the T1 shortening along the CST is not known. Some of the plausible theories include axonal degeneration, myelin breakdown, infiltration of lipid-laden macrophages (sudanophilic lipid), intra-axonal neurofilaments accumulation, and deposition of toxic heavy metals including manganese.[16,18] Waragai et al. suggest that the pathological changes of ALS may cause an increase in intracellular proton density within the spinal cord which contributes to the abnormal T1 hyperintense signal.[18] Further histopathological studies would be needed to understand the pathophysiological basis of this signal alteration and substantiate prior theories.
We recognize the lack of longitudinal imaging follow-up and EMG findings are major limitations of this case report. As discussed, MRI findings are only supportive in the overall diagnosis of ALS; classically described findings like the MBS and CST hyperintensity should not be used for the purpose of labelling a patient with ALS, without appropriate supportive clinical-laboratory findings.[19] However, it would be worthwhile to note that T1 hyperintensity, with or without corresponding T2 hyperintensity, along the expected course of the CSTs is an under-recognized imaging finding in ALS, especially in the spinal cord. While this finding would need prospective trials to justify its role in the diagnosis of ALS, the purpose of our case report was to acquaint the reader with this under-reported finding in the spinal cord.
CONCLUSION
ALS is an uncommon neurodegenerative disease process. Although the diagnosis is predominantly clinical and with EMG, an MRI of the brain and spine may help identify supporting features of ALS and more importantly rule out clinical mimics. Usual findings of ALS include the motor band signs in the precentral gyri and T2 hyperintensity along the CST, however, T1 hyperintensity along the intracranial and spinal course of the CST must alert the radiologist about the potential possibility of ALS and guide further investigations if not available at the time of reporting.
Declaration of patient consent
Patient consent is not required as the patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Amyotrophic lateral sclerosis and its mimics/variants: A comprehensive review. J Clin Imaging Sci. 2018;8:53.
- [CrossRef] [PubMed] [Google Scholar]
- Progress of magnetic resonance imaging in amyotrophic lateral sclerosis. Radiol Infect Dis. 2019;6:1-7.
- [CrossRef] [Google Scholar]
- The present and the future of neuroimaging in amyotrophic lateral sclerosis. Am J Neuroradiol. 2010;31:1769-77.
- [CrossRef] [Google Scholar]
- Diagnostic approach to intrinsic abnormality of spinal cord signal intensity. Radiographics. 2019;39:1824-39.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors for amyotrophic lateral sclerosis. Clinical Epidemiology. 2015;7:181-93.
- [CrossRef] [Google Scholar]
- Amyotrophic lateral sclerosis: A clinical review. Eur J Neurol. 2020;27:1918-29.
- [CrossRef] [PubMed] [Google Scholar]
- Amyotrophic lateral sclerosis: An update for 2013 clinical features, pathophysiology, management and therapeutic trials. Aging Dis. 2013;4:295-310.
- [Google Scholar]
- Electrodiagnosis in persons with amyotrophic lateral sclerosis. PMR. 2013;5:S89-S95.
- [CrossRef] [Google Scholar]
- Neuroimaging in amyotrophic lateral sclerosis. Neurotherapeutics. 2012;6:319-37.
- [CrossRef] [PubMed] [Google Scholar]
- The motor band sign in ALS: Presentations and frequencies in a consecutive series of ALS patients. J Neurol Sci. 2019;406:116440.
- [CrossRef] [PubMed] [Google Scholar]
- The “motor band sign:” susceptibility-weighted imaging in amyotrophic lateral sclerosis. Can J Neurol Sci. 2015;42:260-3.
- [CrossRef] [PubMed] [Google Scholar]
- Dementias and brain degenerations. In: Osborn’s Brain, ed. Imaging, Pathology, and Anatomy (2nd ed.). New York, NA: Elsevier; 2013. p. :1106-8.
- [Google Scholar]
- Hyperintensity of the corticospinal tract on FLAIR: A simple and sensitive objective upper motor neuron degeneration marker in clinically verified amyotrophic lateral sclerosis. J Neurol Sci. 2016;367:177-83.
- [CrossRef] [PubMed] [Google Scholar]
- Amyotrophic Lateral Sclerosis: Hyperintensity of the Corticospinal Tracts on MR Images of the Spinal Cord. AJR Am J Roentgenol. 1993;160:604-6.
- [CrossRef] [PubMed] [Google Scholar]
- A distinct MR imaging phenotype in amyotrophic lateral sclerosis: Correlation between T1 magnetization transfer contrast hyperintensity along the corticospinal tract and diffusion tensor imaging analysis. AJNR Am J Neuroradiol. 2011;33:733-9.
- [CrossRef] [PubMed] [Google Scholar]
- Magnetization transfer imaging and proton MR spectroscopy in the evaluation of axonal injury: Correlation with clinical outcome after traumatic brain injury. AJNR Am J Neuroradiol. 2001;22:143-51.
- [PubMed] [Google Scholar]
- High signal intensity on T1 weighted MRI of the anterolateral column of the spinal cord in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. 1997;62:88-91.
- [CrossRef] [PubMed] [Google Scholar]
- EFNS guidelines on the use of neuroimaging in the management of motor neuron diseases. Eur J Neurol. 2010;17:526-e20.
- [CrossRef] [PubMed] [Google Scholar]







