Translate this page into:
Spectrum of CT Findings Related to Bowel Adhesions Without Bowel Obstruction: A Comprehensive Imaging Review

-
Received: ,
Accepted: ,
How to cite this article: Gopireddy DR, Soule E, Arif-Tiwari H, Sharma S, Kanmaniraja D, Jain K, et al. Spectrum of CT findings related to bowel adhesions without bowel obstruction: A comprehensive imaging review. J Clin Imaging Sci 2020;10:80.
Abstract
Post-operative adhesions can occur following abdominal or pelvic surgery and are a significant cause of morbidity and mortality. Adhesions are an important cause of abdominal complaints, especially chronic abdominal pain, infertility, and bowel obstruction; responsible for an estimated $2.3 billion annual burden in the United States alone. There is a paucity of literature describing CT findings indicative of the presence of adhesions, however, findings suggestive of this entity may be discovered by a discerning eye. Early recognition and diagnosis may be beneficial for reducing consumption of health-care resources related to abdominal complaints. In addition, these findings may impact surgical planning and facilitate recognition of adhesion-related complications, said to be responsible for a significant number of malpractice claims. The following review discusses various CT findings which may indicate the presence of adhesions. These findings may initially be overlooked, but remain valuable as the clinical presentation evolves to become consistent with adhesive disease. Detection of bowel and peritoneal adhesions can be recognized on imaging modalities based on distortion of bowel loops. This pictorial review illustrates the spectrum of findings related to bowel adhesions encountered on computed tomography imaging.
Keywords
Adhesive disease
Computed tomography
Postsurgical adhesions
Radiomics
INTRODUCTION
Peritoneal adhesions may have varying etiologies. Congenital adhesions are adhesive bands present at birth are a rare cause of bowel obstruction. Post-inflammatory adhesions related to prior infectious or chemical peritonitis, prior radiotherapy, or due to chronic irritation (peritoneal dialysis, endometriosis, or pelvic inflammatory disease) are fairly common but less likely to cause obstruction than post-operative adhesions.[1] A post-operative peritoneal adhesion consists of fibrous bands that form after surgery in the lower or upper abdominal area resulting from tissue disturbance. The risk of adhesions is higher following lower abdominal, bowel, and gynecological surgeries. Post-surgical adhesions are the most common cause for small bowel obstruction, accounting for 60–80% of all small bowel obstructions.[2-7] Menzies and Ellis showed that 93% of all intra-abdominal adhesions were post-surgical. Of those, as many as 39% presented with small bowel obstruction, within 1 year of the surgery.[8]
There is significant morbidity and mortality associated with post-operative adhesions due to complications from small bowel obstruction, as well as a range of clinical symptoms from chronic abdominal pain and bloating to female infertility.[8,9] In 1994, the estimated impact for direct patient care related to adhesive disease in the United States was 1.3 billion dollars. This burden increased to 2.3 billion in 2011.[10,11] Development of adhesions and severity of adhesive disease can be correlated with specific types of surgery and the overall area of involvement of peritoneum. Laparoscopy has less propensity for dense adhesive disease compared to laparotomy.[12,13] Obstruction due to adhesions has been estimated at 1–10% after simple surgical procedures like appendectomy.[13,14] This increases to 17–25% following complex procedures including proctocolectomy.[15-18]
Post-operative adhesions developing between small bowel loops are referred to as enteroenteric or interloop adhesions. Adhesions between the bowel and peritoneal surfaces (usually anterior peritoneum) are referred to as enteroparietal adhesions and those between bowel and visceral organs are referred to as enterovisceral adhesions. Although the majority of patients with adhesions are asymptomatic, some patients can present with specific symptoms. Patients with enteroparietal type adhesion often present with chronic pain, while enteroenteric adhesions may be responsible for obstructive symptoms.[19] Recognition of the various types of adhesions can impact clinical and surgical management. For example, a specific site for trocar placement or surgical approach may be altered if there are dense enteroparietal adhesions in the anterior peritoneum.[20,21]
The pathogenesis and development of post-operative adhesions are related to surgical intervention itself. Abdominal surgery inadvertently causes peritoneal injury, initiating an inflammatory process with the deposition of fibrin. Adhesive disease can be compounded by infection, the presence of foreign bodies (sutures), and impaired fibrinolysis. When fibrin is not rapidly degraded (within 5–7 days), it serves as a matrix for proliferation of fibroblasts and capillaries and leads to formation of peritoneal adhesions.[4] These aberrant foci were historically though to be composed of non-functional scar tissue.[22] Subsequent pathologic studies, however, demonstrated them to contain various cellular components; consistent with dynamic regenerating structures.[23] Histologically, adhesions are composed of abnormal fibrous bands incorporating organized vascular, nerve, and smooth muscle tissue.[5,24,25]
The increased prevalence of imaging with multidetector CT technology offering higher resolution and isotropic imaging with multiplanar reconstructions has resulted in better visualization of adhesions. The anterior peritoneum is a common location for enteroparietal adhesions, leading to distortion of bowel loops and visceral organ surfaces with tethering of these structures, which can indicate underlying adhesions. Characteristic CT imaging features of adhesions are often related to the distortion of involved bowel loops with ancillary findings in the involved mesentery and overlying peritoneum.
RESULTS
Abnormal findings indicative of adhesive disease may be observed in the bowel, mesentery, and/or peritoneum [Table 1].
| Bowel findings | Mesenteric findings | Peritoneal findings | Special “signs” |
|---|---|---|---|
| Unchanged location of bowel segments on successive examinations [Figure 5a and b] Focal clustering or crowding of bowel loops [Figure 3] Kinking and angulation of bowel loops [Figure 2] Asymmetric thickness of the bowel wall [Figure 6] Enterovisceral adhesions [Figure 4] |
Focal increased attenuation due to congestion [Figure 7] Vascular crowding [Figure 8] |
Loss of fat planes behind the rectus sheath and anterior peritoneum [Figure 11] Focal peritoneal thickening and enhancement [Figure 10] Peritoneal calcifications and sclerosing peritonitis [Figure 9] |
“Fat notch” sign – insinuation of mesenteric fat at site of a focal bowel caliber change [Figure 12] “Beak” sign – abrupt caliber change of bowel [Figure 13] “Small bowel feces” sign – localized stasis within bowel segments [Figure 13] |
DISCUSSION
Studies have shown that post-operative adhesions range from 67% to 93% in patients with a history of abdominal surgery.[26,27] Roughly 20% of adhesions can occur as early as 1 month of surgery [Figure 1], while 40% of adhesions may occur after 1 year post-surgery.[27] Adhesions can increase in thickness with increased tension overtime, leading to symptoms and complications several years after the initial operation. Understanding and recognition of the imaging signs of adhesive disease are, therefore, crucial when a patient presents with relevant symptoms, especially with a history of abdominal surgery. Furthermore, if adhesive disease is suspected, the patient can be referred for more accurate imaging studies. One such study is functional cine – MRI, with a reported sensitivity of 87.5% and specificity of 92.5%.[28]
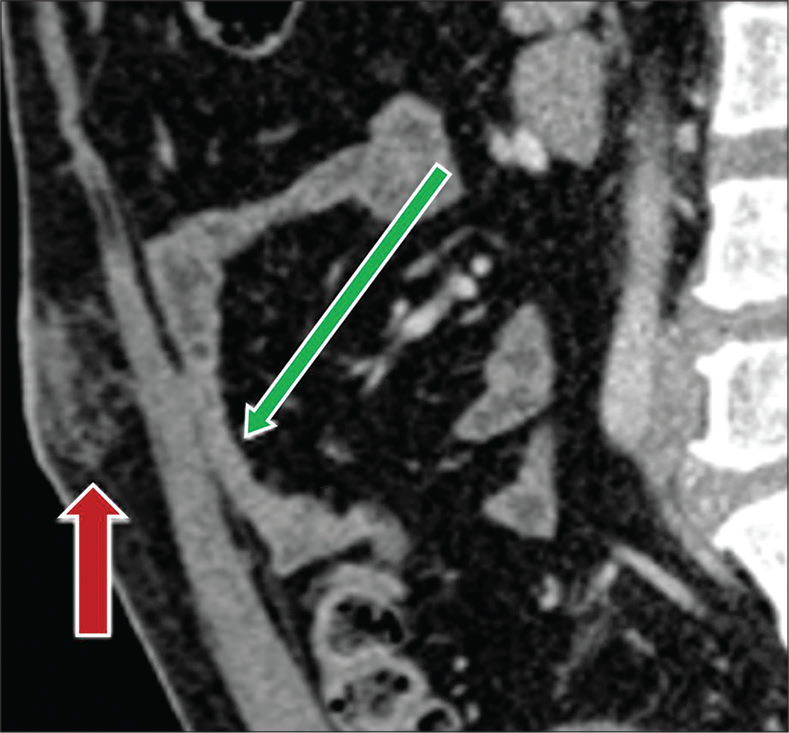
- A 52-year-old man with recent history of laparoscopic cholecystectomy, 2 weeks prior presenting with abdominal pain. Sagittal contrast-enhanced CT image demonstrates formation of early bowel adhesions (green arrow). Note postsurgical changes at the umbilicus from laparoscopic port placement (red arrow).
Bowel findings
One of the most common signs of bowel adhesions is acute angulation of bowel. Normal small bowel being freely mobile normally shows smooth curved loops, evenly distributed in the abdomen and pelvis. With the development of either enteroenteric adhesions or enteroparietal adhesions, the bowel loops become fixed at certain points, leading to acutely angulated segments with kinking and changes in caliber [Figure 2]. Clustered or unusual crowded appearance of bowel segments is typically seen with enteroenteric or interloop adhesions, classically referred to as “matted bowel” [Figure 3]. This finding can also be seen with intra-abdominal infections such as abdominal tuberculosis.[29] Enterovisceral adhesions, or bowel adherent to adjacent organs, may additionally be indicative of adhesive disease [Figures 4 and 5].

- A 52-year-old female status post- hysterectomy, 3 years previously, presenting with chronic lower abdominal pain. Axial contrast-enhanced CT images with IV contrast demonstrate acute angulation of bowel loops (red arrow) consistent with adhesive disease.
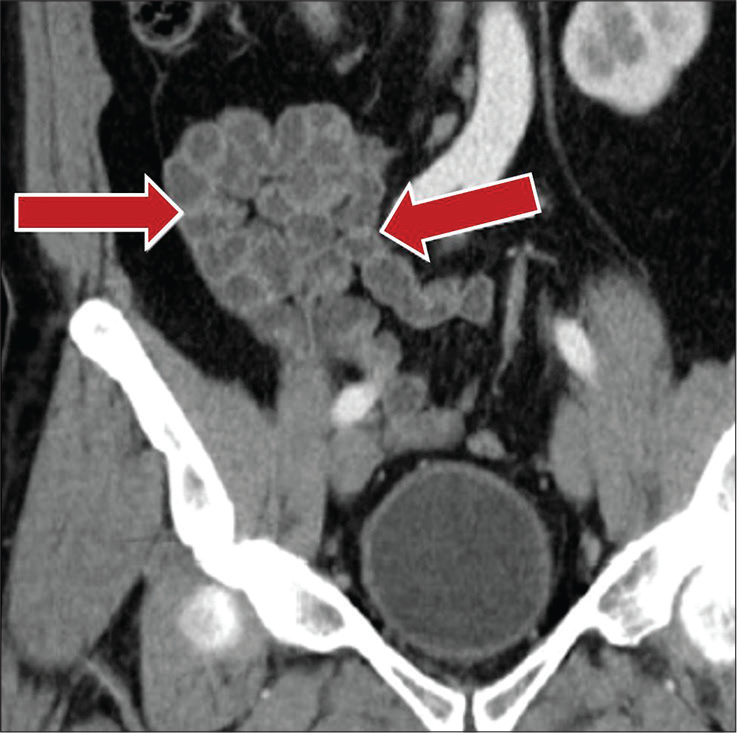
- A 56-year-old female with a history of hysterectomy 6 years prior presenting with intermittent nausea and vomiting. Coronal contrast-enhanced CT image demonstrates clustered small bowel loops in the right lower quadrant consistent with high-grade enteroenteric adhesions (red arrows).
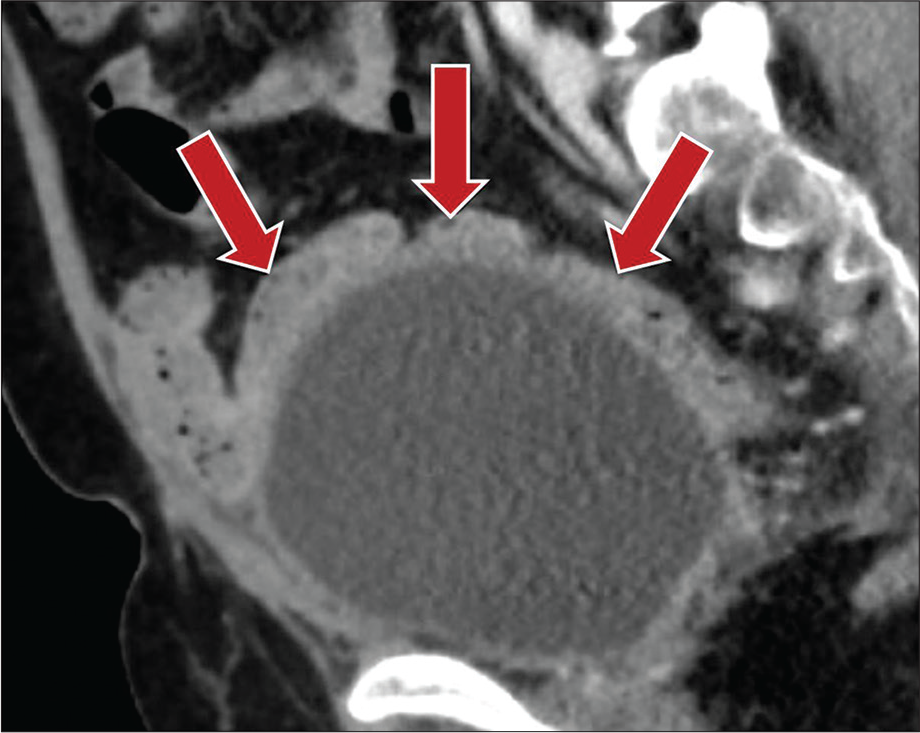
- A 50-year-old male status post-prostatectomy presenting with chronic intermittent abdominal pain. Contrast-enhanced sagittal CT image demonstrates enterovisceral adhesions to the bladder dome with matted appearance of small bowel loops (red arrows).
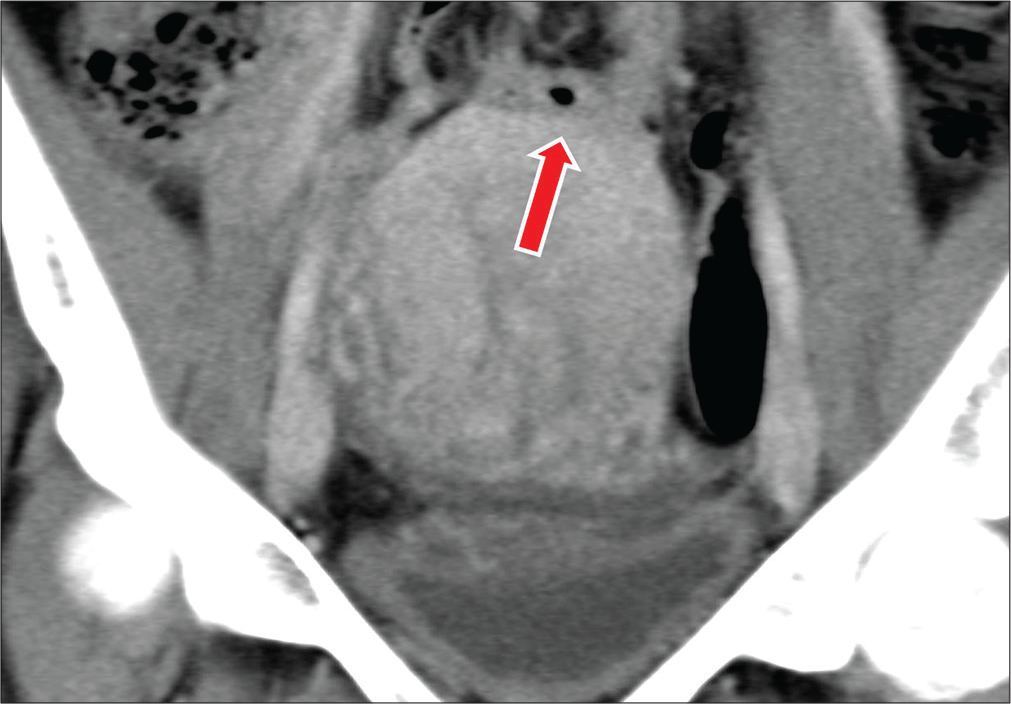
- A 28-year-old female with a history of four cesarean section deliveries presents with abdominal pain. Coronal CT image demonstrate a loop of small bowel adherent to the superior aspect of the uterus (red arrow).
Bowel adhesions may lead to increased transit times through the small bowel.[30] These segments may demonstrate features of stasis including fluid accumulation and fecalized appearance of small bowel loops.[31] The finding of localized stasis in the absence of upstream distension may be an indirect sign of unsuspected bowel adhesions.[32] A static location of bowel segments over multiple studies is a strong predictor of adhesions. Normal bowel is freely mobile in the peritoneal cavity on its mesentery, and presence of adhesions restricts the normal movement of bowel, leading to a “fixed” position, detected on successive CT or MRI examinations [Figure 6a and b]. This finding may be indicative of underlying adhesive bands restricting normal bowel motion/peristalsis. When loops of small bowel and colon appear tethered to each other or the peritoneum, asymmetric thickening of the bowel wall at the site of the adhesion may be observed [Figure 7].
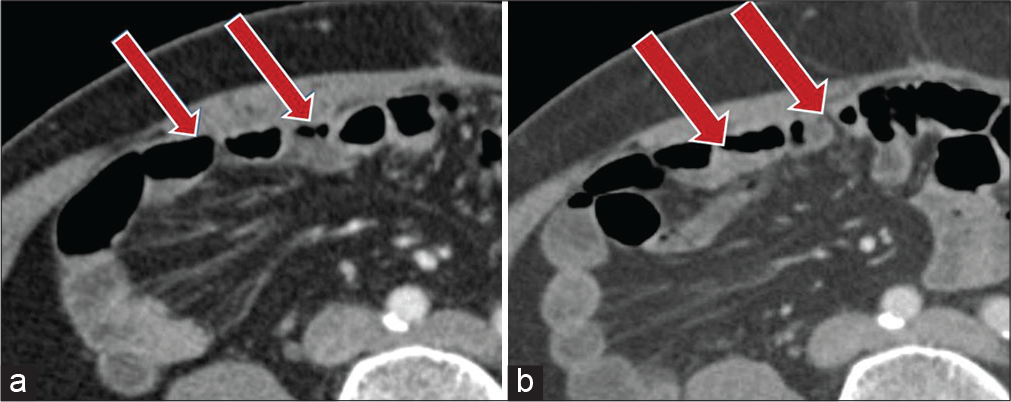
- A 53-year-old male with prior history of laparotomy for gastric gastrointestinal stromal tumor, 3 years ago, presenting with sudden onset abdominal pain. (a) Contrast-enhanced axial CT image demonstrates bowel loops abutting the right anterior abdominal wall with obliteration of the right post-rectus fat plane (red arrows). (b) Two-year follow-up contrast-enhanced axial CT shows small bowel loops unchanged in location (red arrows).
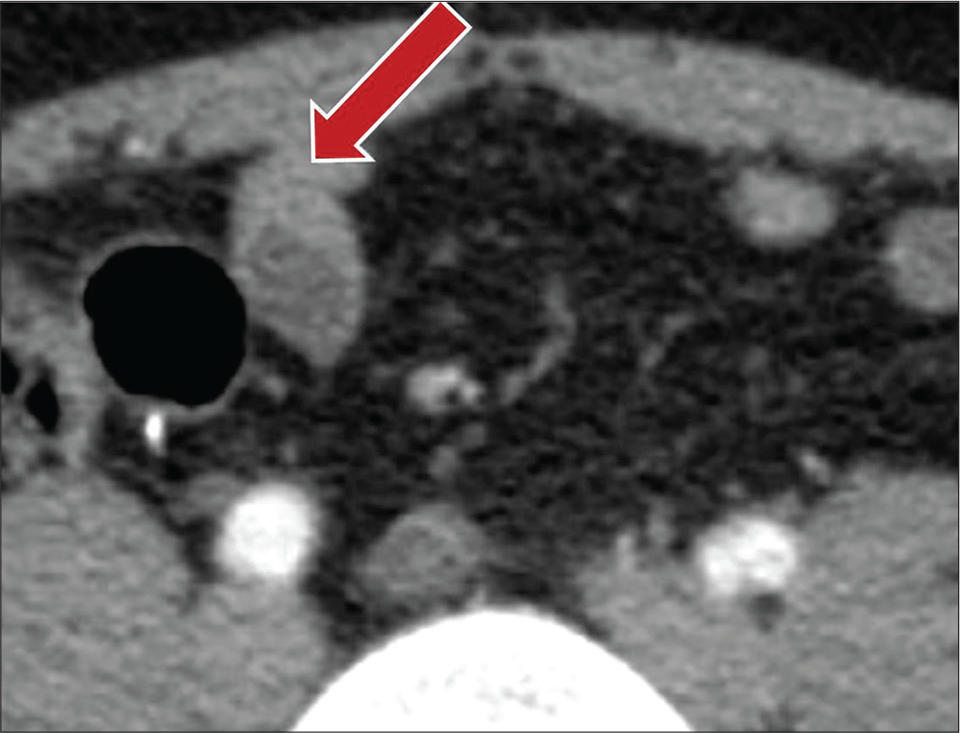
- A 73-year-old male with a history of laparotomy for appendiceal carcinoid presenting with abdominal pain. Contrast-enhanced axial CT images show small bowel loop with asymmetric wall thickening from underlying adhesions (red arrow).
Mesenteric findings
Mesenteric findings associated with adhesions include focal increased attenuation due to congestion, vascular crowding, and focal localized fluid.[29,33,34] Congestion of mesenteric veins is a hemodynamic complication that may be indicative of mesenteric adhesions. This is hypothesized to be due to third space fluid accumulation; secondary to a surgical procedure with vascular congestion of the involved small bowel mesentery. Mesenteric fat on CT is usually easily identified by its attenuation value of around –100 Hounsfield units. When there is mesenteric infiltration by inflammatory cells or fluid, the attenuation of the fat increases [Figure 8]. In the presence of ancillary findings of adhesions and a positive surgical history, adhesive disease could be considered as an etiology of increased mesenteric fat density. Another important mesenteric finding is the asymmetric crowding of mesenteric vessels [Figure 9].
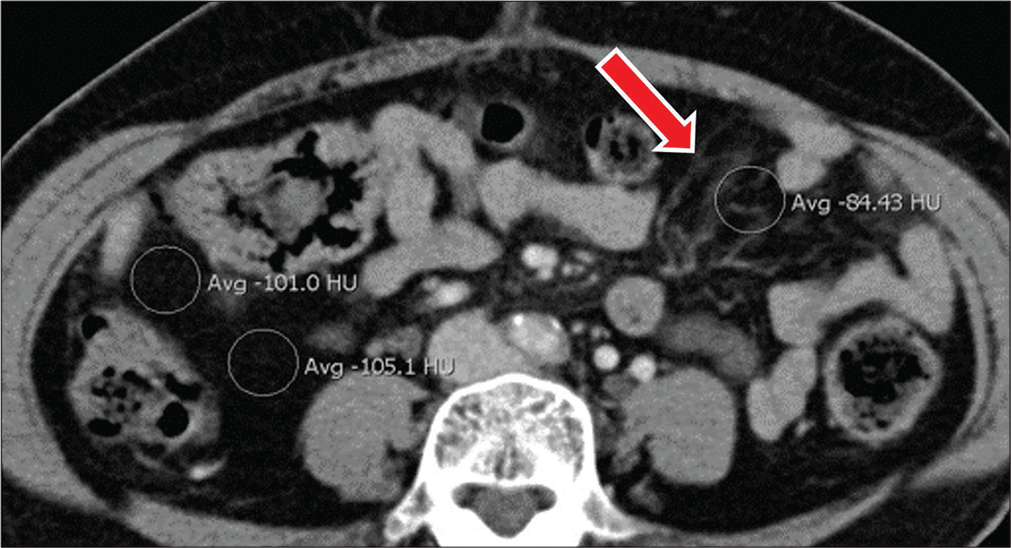
- A 44-year-old female status post-cholecystectomy presents with abdominal pain. Axial contrast-enhanced CT images show localized increase in mesenteric attenuation in the left abdomen when compared to normal mesenteric fat in the right abdomen, related to mesenteric congestion (red arrow) secondary to underlying adhesions.
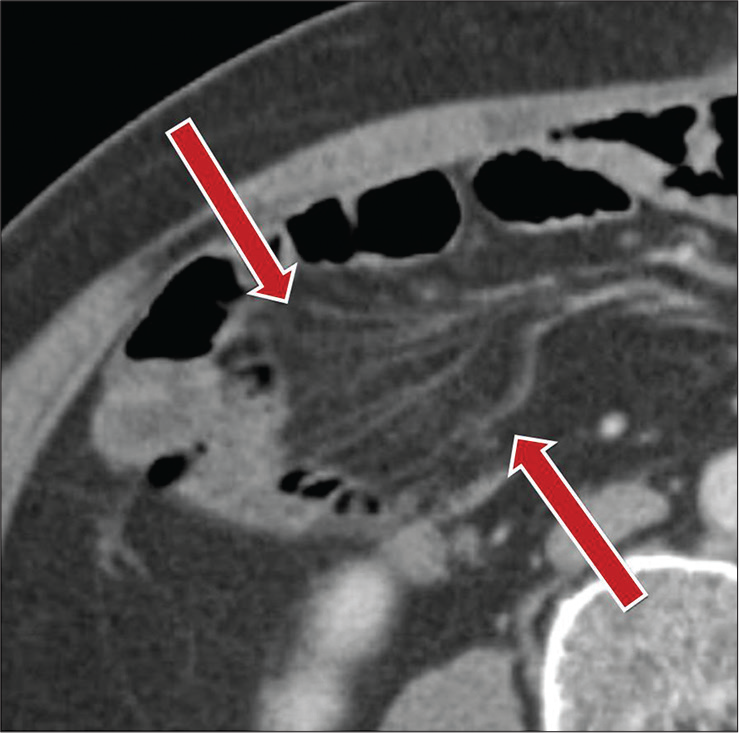
- A 56-year-old male with a history of cholecystectomy presenting with abdominal pain. Axial contrast-enhanced CT image demonstrating localized vascular crowding in the right abdomen related to mesenteric congestion (red arrows) secondary to underlying small bowel adhesions.
Peritoneal findings
Any insult to the peritoneum including abdominal/pelvic surgery, peritoneal dialysis, or peritonitis can lead to peritoneal inflammation and thickening [Figure 10]. The finding of peritoneal thickening may be associated with underlying bowel adhesions.[35-37] Cocoon abdomen refers to closely clustered bowel loops adherent within thickened peritoneum in a sac-like manner [Figure 11]. This finding is classically associated in patients with peritoneal dialysis and underlying enteroparietal and enteroenteric adhesions.
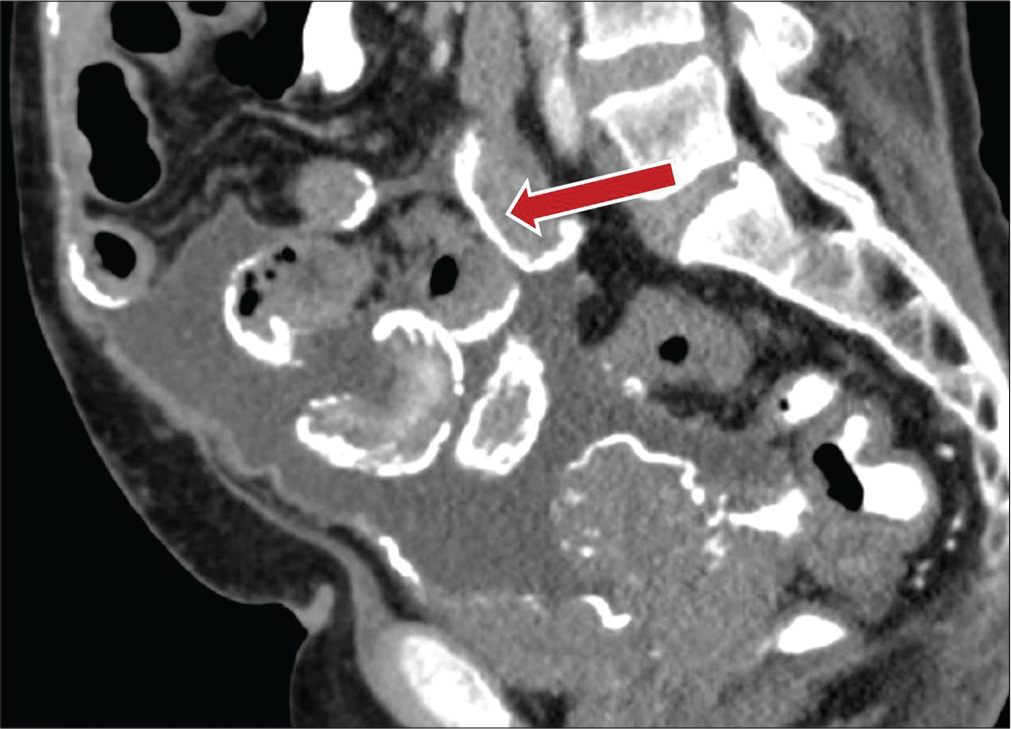
- A 73-year-old male with a history of peritoneal dialysis presenting with abdominal pain. Sagittal contrast-enhanced CT image demonstrates thick peritoneal and mesenteric surface calcifications (red arrow) involving multiple loops of bowel, consistent with encapsulating peritoneal sclerosis.
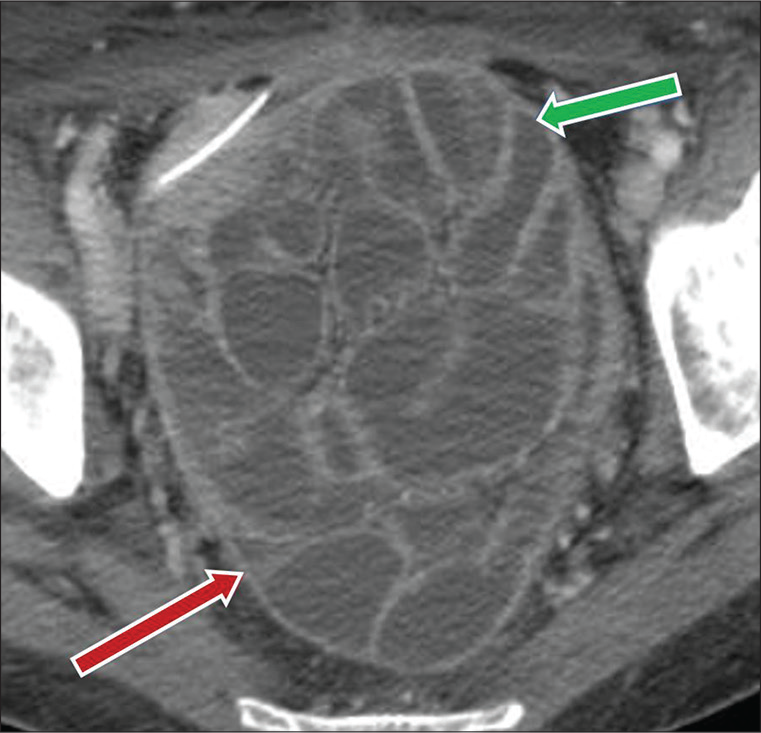
- A 56-year-old male, status post-colonic surgery, presents with abdominal pain and vomiting. Axial CT image demonstrates a cluster of adherent fluid filled mildly dilated small bowel loops in pelvic region (green arrow), enclosed by thickened peritoneal lining (red arrow), consistent with “cocoon abdomen.”
Normal bowel segments are nearly always separated from the posterior aspect of the rectus muscles by mesenteric and omental fat. Following intra-abdominal and pelvic surgeries, the tissue disturbance at the level of the visceral peritoneum results in closely applied adherence of the small bowel with resultant loss of the retrorectus fat planes. This may represent a secondary finding of enteroparietal adhesions [Figure 12].

- A 50-year-old female patient status post-cholecystectomy presenting with abdominal pain. Contrast-enhanced axial CT image demonstrates loss of the posterior rectus sheath fat plane (red arrows).
Special “signs”
Radiographic “signs” of adhesive disease on CT include the fat notch sign, the beak sign, and the small bowel feces sign. The fat notch sign may develop due to insinuation of mesenteric fat at an area of focal caliber change or angulation of the bowel [Figure 13]. Mesentery, tethered by an adhesive band, may be dragged around the segment of bowel in question by subsequent peristalsis.[38] This has been described in cases of small bowel obstruction related to adhesions.[39] The beak sign is demonstrated when there is an abrupt change in the caliber of the bowel lumen [Figure 14]. If associated with upstream dilated loops of bowel, this may represent obstruction. Adhesive disease, however, commonly presents with intermittent obstruction. As such, obstructive symptoms exist on a continuum. The presence of a beak sign in the absence of frank symptomatic obstruction, therefore, could represent subclinical or unsuspected adhesive disease. Finally, the small bowel feces sign is caused by fecal stasis in the small bowel, which could be due to adhesions [Figure 15]. The small bowel feces sign is likely to represent obstruction if associated with significant bowel distension. In the absence of upstream dilation and downstream collapse of small bowel, one study found that 68% of patients with a positive small bowel feces sign were non-obstructed.[32]
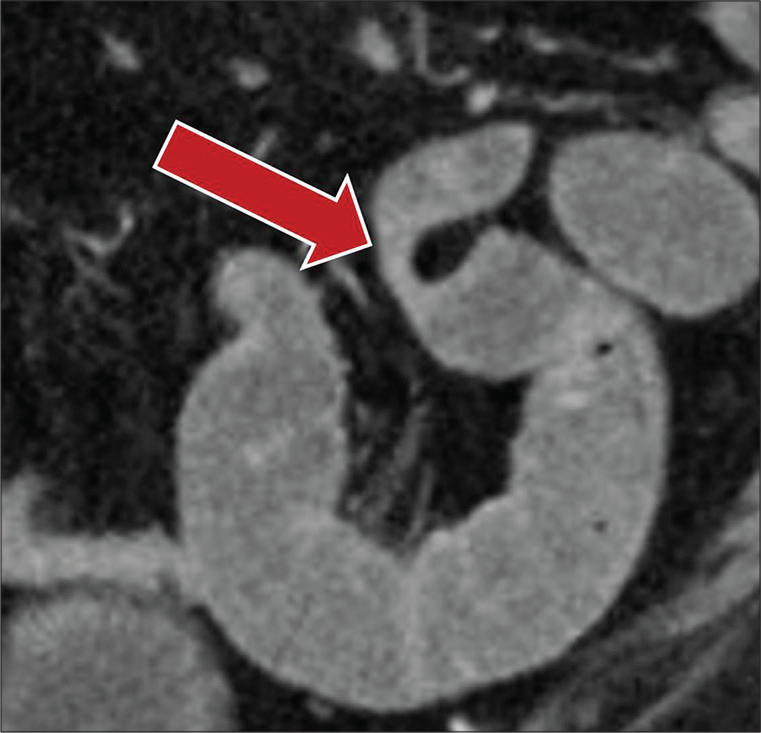
- A 44-year-old female with a surgical history of hysterectomy presenting with abdominal pain, nausea, and vomiting. Coronal CT image demonstrates the fat notch sign (red arrow), secondary to mesenteric fat insinuating between bowel loops secondary to underlying adhesions.
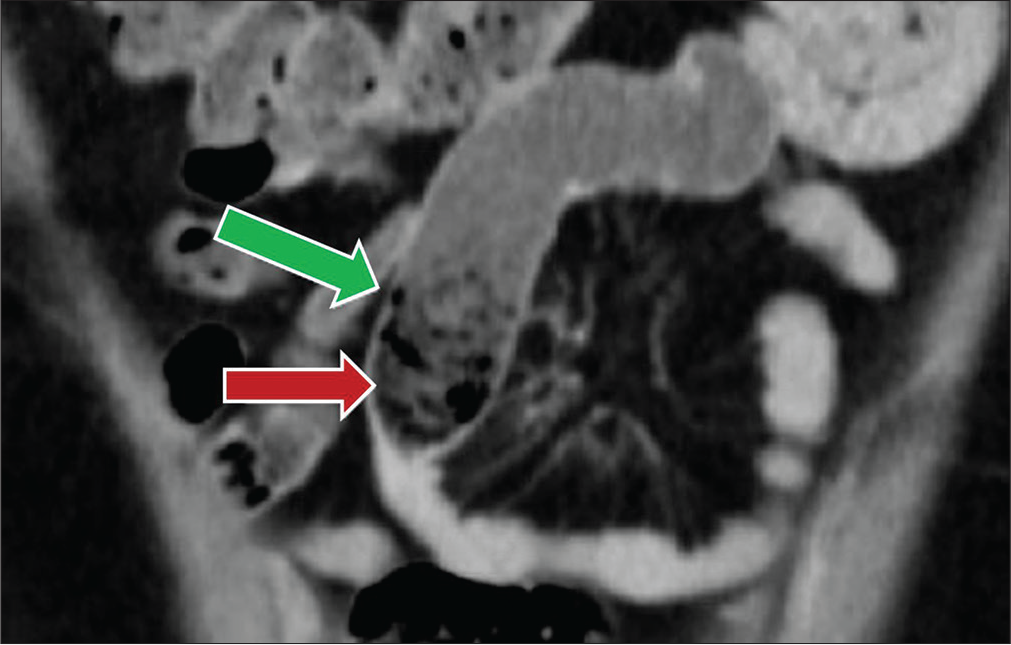
- A 47-year-old female status post-hysterectomy presenting with nausea. Coronal contrast-enhanced CT image demonstrates the “beak” sign or focal caliber change of a small bowel loop in the mid abdomen (red arrow) with fecalized material “small bowel feces sign” present proximal to the caliber change (green arrow).
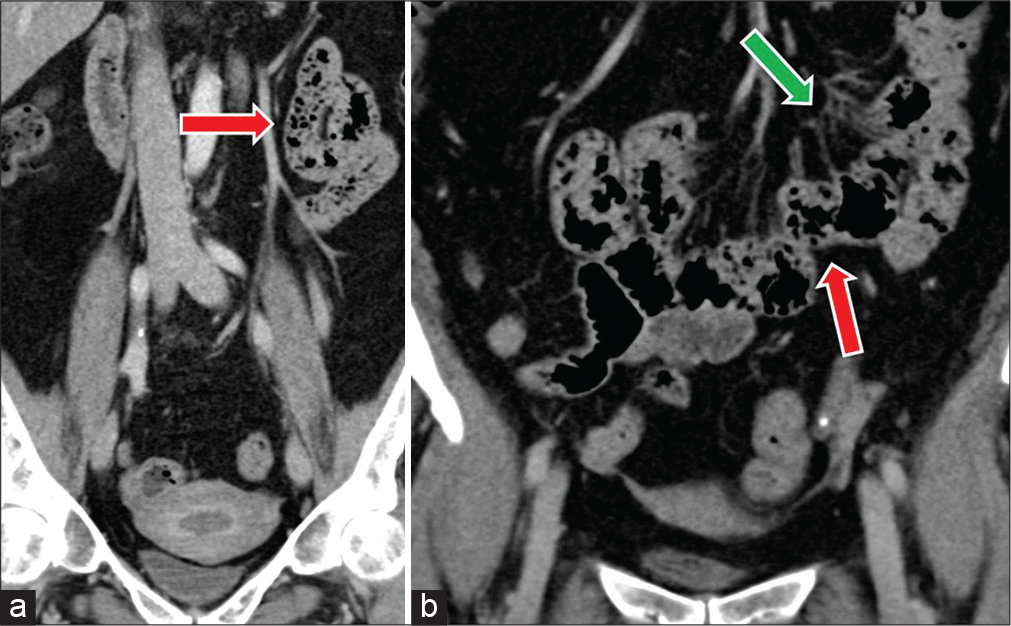
- (a and b) A 69-year-old female with a history of tubal ligation presents with abdominal pain. Coronal CT images demonstrate non-distended small bowel loops clustered in the mid-abdomen containing fecalized material consistent with small bowel feces sign (red arrows). Note is made of regional mesenteric congestion (green arrow).
CONCLUSION
Adhesive disease may be encountered on cross-sectional imaging exams performed routinely in clinical practice. They are the most common cause of small bowel obstruction and can have varied clinical presentation ranging from chronic abdominal pain to infertility. Recognition of the CT findings associated with adhesive disease has important clinical implications for both patients and the referring physicians. Precise pre-operative planning in patients with adhesions is crucial to minimize the morbidity and mortality associated with bowel injury.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Peritoneal healing and adhesion formation/ reformation. Hum Reprod Update. 2001;7:556-66.
- [CrossRef] [PubMed] [Google Scholar]
- The clinical significance of adhesions: Focus on intestinal obstruction. Eur J Surg Suppl. 1997;577:5-9.
- [Google Scholar]
- Clinical implications of postsurgical adhesions. Hum Reprod Update. 2001;7:567-76.
- [CrossRef] [PubMed] [Google Scholar]
- Pathophysiology and prevention of postoperative peritoneal adhesions. World J Gastroenterol. 2011;17:4545-53.
- [CrossRef] [PubMed] [Google Scholar]
- Presence and distribution of sensory nerve fibers in human peritoneal adhesions. Ann Surg. 2001;234:256-61.
- [CrossRef] [PubMed] [Google Scholar]
- Adhesive small bowel obstruction: Epidemiology, biology and prevention. Can J Surg. 2007;50:291-300.
- [Google Scholar]
- Peritoneal adhesions: Etiology, pathophysiology, and clinical significance, Recent advances in prevention and management. Dig Surg. 2001;18:260-73.
- [CrossRef] [PubMed] [Google Scholar]
- Intestinal obstruction from adhesions-how big is the problem? Ann R Coll Surg Engl. 1990;72:60-3.
- [Google Scholar]
- Adhesions after extensive gynecologic surgery: Clinical significance, etiology, and prevention. Am J Obstet Gynecol. 1994;170:1396-403.
- [CrossRef] [Google Scholar]
- Abdominal adhesiolysis: Inpatient care and expenditures in the United States in 1994. J Am Coll Surg. 1998;186:1-9.
- [CrossRef] [Google Scholar]
- The inpatient burden of abdominal and gynecological adhesiolysis in the US. BMC Surg. 2011;11:13.
- [CrossRef] [PubMed] [Google Scholar]
- Fewer adhesions induced by laparoscopic surgery? Surg Endosc. 2004;18:898-906.
- [CrossRef] [PubMed] [Google Scholar]
- The postoperative incidence of small bowel obstruction following standard, open appendectomy and cholecystectomy: A six-year retrospective cohort study at Yale-New Haven Hospital. Conn Med. 1993;57:123-7.
- [Google Scholar]
- Mechanical small-bowel obstruction after conventional appendectomy in children. Eur J Pediatr Surg. 1997;7:13-5.
- [CrossRef] [PubMed] [Google Scholar]
- Risk of small bowel obstruction after the ileal pouch-anal anastomosis. Ann Surg. 2002;235:200-6.
- [CrossRef] [PubMed] [Google Scholar]
- Ileal pouch-anal anastomoses complications and function in 1005 patients. Ann Surg. 1995;222:120-7.
- [CrossRef] [PubMed] [Google Scholar]
- Obstruction after ileal pouch-anal anastomosis: A preventable complication? Dis Colon Rectum. 1993;36:1105-11.
- [CrossRef] [PubMed] [Google Scholar]
- Small intestinal obstruction complicating ileal pouch-anal anastomosis. Ann Surg. 1989;209:46-50.
- [CrossRef] [PubMed] [Google Scholar]
- Direct visual or blind insertion of the primary trocar. Surg Endosc. 2002;16:626-9.
- [CrossRef] [PubMed] [Google Scholar]
- Safe laparoscopy: Is it possible? Curr Opin Obstet Gynecol. 2004;16:283-8.
- [CrossRef] [PubMed] [Google Scholar]
- Computed tomography and magnetic resonance imaging in the evaluation of pelvic peritoneal adhesions: What radiologists need to know? Indian J Radiol Imaging. 2014;24:149-55.
- [CrossRef] [PubMed] [Google Scholar]
- Human peritoneal adhesions are highly cellular, innervated, and vascularized. J Pathol. 2000;192:67-72.
- [CrossRef] [Google Scholar]
- Immunohistochemical demonstration of nerve fibers in pelvic adhesions. Obstet Gynecol. 1993;82:566-8.
- [CrossRef] [PubMed] [Google Scholar]
- A study of nerve fibers and histopathology of postsurgical, postinfectious, and endometriosis-related adhesions. Obstet Gynecol. 1998;92:766-8.
- [CrossRef] [Google Scholar]
- The biology of adhesion formation in the peritoneal cavity. Semin Pediatr Surg. 2014;23:336-43.
- [CrossRef] [PubMed] [Google Scholar]
- Post-operative adhesions after digestive surgery: Their incidence and prevention: Review of the literature. J Visc Surg. 2012;149:e104-14.
- [CrossRef] [PubMed] [Google Scholar]
- Detection and mapping of intraabdominal adhesions by using functional cine MR imaging: Preliminary results. Radiology. 2000;217:421-5.
- [CrossRef] [PubMed] [Google Scholar]
- Abdominal cocoon secondary to tuberculosis. Saudi J Gastroenterol. 2008;14:139-41.
- [CrossRef] [PubMed] [Google Scholar]
- Small bowel transit time in patients with intra-abdominal adhesions. Br J Surg. 1992;79:1076.
- [CrossRef] [PubMed] [Google Scholar]
- The CT small bowel faeces sign: Description and clinical significance. Clin Radiol. 1995;50:765-7.
- [CrossRef] [Google Scholar]
- Small bowel faeces sign in patients without small bowel obstruction. Clin Radiol. 2007;62:353-7.
- [CrossRef] [PubMed] [Google Scholar]
- The misty mesentery on CT: Differential diagnosis. AJR Am J Roentgenol. 1996;167:61-5.
- [CrossRef] [PubMed] [Google Scholar]
- Segmental misty mesentery: Analysis of CT features and primary causes. Radiology. 2003;226:86-94.
- [CrossRef] [PubMed] [Google Scholar]
- Computed tomographic findings characteristic for encapsulating peritoneal sclerosis: A case-control study. Perit Dial Int. 2009;29:517-22.
- [CrossRef] [PubMed] [Google Scholar]
- Encapsulating peritoneal sclerosis-a rare but devastating peritoneal disease. Front Physiol. 2014;5:470.
- [CrossRef] [PubMed] [Google Scholar]
- Small bowel obstruction from adhesions: Which CT severity criteria to research? J Radiol. 2003;84:27-31.
- [Google Scholar]
- Multidetector CT appearance of adhesion-induced small bowel obstructions: Matted adhesions versus single adhesive bands. Jpn J Radiol. 2012;30:706-12.
- [CrossRef] [PubMed] [Google Scholar]
- Small-bowel obstruction from adhesive bands and matted adhesions: CT differentiation. AJR Am J Roentgenol. 2009;192:693-7.
- [CrossRef] [PubMed] [Google Scholar]






