Translate this page into:
Role of Chest CT in COVID-19

-
Received: ,
Accepted: ,
How to cite this article: Malguria N, Yen L, Lin T, Hussein A, Fishman E.K. Role of chest CT in COVID-19. J Clin Imaging Sci 2021;11:30.
Abstract
In December 2019, a disease attributed to a new severe acute respiratory syndrome coronavirus 2, and named coronavirus disease 2019 (COVID-19), broke out in Wuhan, China and has spread rapidly throughout the world. CT has been advocated in selected indications as a tool toward rapid and early diagnosis. The CT patterns of COVID-19 include ground glass opacities GGO, consolidation, and crazy paving. Additional signs include a “rounded morphology” of lesions, vascular enlargement sign, nodules, and fibrous stripe. Signs of healing and organization include subpleural bands, a reticular pattern, reversed halo sign and traction bronchiectasis. Cavitation and tree in bud signs are absent and pleural effusions are rare. There is a high incidence of pulmonary embolism associated with COVID-19. CT findings in COVID-19 appear to follow a predictable timeline with maximal involvement approximately 6–11 days after symptom onset. The stages of evolution include early stage (days 0–4) with GGO being the predominant abnormality, progressive stage (days 5–8) with increasing crazy paving; and peak stage (days 9–13) with predominance of consolidation and absorption phase (after day 14) with gradual absorption of consolidation with residual GGO and subpleural bands. CT findings in COVID-19 have a high sensitivity and low specificity, determined to be 98% and 25% in a retrospective study of 1014 patients. The low specificity of CT for the diagnosis of COVID-19 pneumonia is due to the overlap of CT findings with other viral pneumonias and other infections, lung involvement in connective tissue disorders, drug reaction, pulmonary edema, and hemorrhage.
Keywords
Consolidation
Crazy paving
Ground glass opacities
Rounded consolidation
Vascular enlargement sign
INTRODUCTION
In December 2019, a cluster of cases of unexplained pneumonia broke out in Wuhan, China.[1,2] This disease was attributed to a new virus severe acute respiratory syndrome coronavirus 2,[3] and named coronavirus disease 2019 (COVID-19). It has spread rapidly throughout the world and there are currently over 18 million people infected and 689,245 deaths worldwide.[4]
At present, treatment is predominantly supportive and/or experimental[5-8] with the focus of management being rapid diagnosis, isolation of those infected and of asymptomatic contacts. Diagnosis is primarily using real-time polymerase chain reaction (RT-PCR) testing of nasopharyngeal swabs, which have a positive detection rate of 32–63%.[9-11] Recent large-scale studies demonstrate RT-PCR tests turning positive in high risk patients after as many as two initial false negative results.[9,11] CT was used as a means of rapid diagnosis and disease containment in Hubei, China, with the lower mortality than other parts of the world, prompting some authors to advocate widespread use of chest CT as a tool for rapid and early diagnosis and as a measure toward pandemic control;[12,13] while others including the Fleischner society, recommend the use of CT in selected settings.[14,15] In this review, we will discuss the imaging appearances of COVID-19 on CT, review the literature on the diagnostic performance of CT with respect to RT-PCR and the current recommendations for the role CT in COVID-19 pneumonia.
CT IMAGING IN SYMPTOMATIC AND ASYMPTOMATIC PATIENTS
Ground glass opacities GGO and consolidations are the most common CT presentation, with a bilateral middle and lower lobe predominance[16-19] and a peripheral distribution (76– 85%).[16,18,20] The disease is usually multifocal, although single lesions may be seen on early imaging, Zhou et al. found single lesions on the initial CT scan in 16.1% of their 62 patients when the scans were performed 2.2 ± 1.8 days after symptom onset and multiple lesions on CT scans performed 6.6 ± 4.4 days after symptom onset.[17]
GGO are areas of hazy increased opacity of the lung parenchyma, with vascular and bronchial margins visible through the opacification [Figure 1], the underlying mechanism being partial displacement of air in the affected alveoli.[21] They are the most common and earliest presentation and have been reported in 34–98% of patients depending on patient population and timing of the CT scan.[12,22-33] Autopsy and pathologic studies have showed the presence of diffuse alveolar damage, perivascular T-cell infiltration and/or hyaline membrane formation,[34-36] features which have been speculated to be the pathological basis of the GGO appearance in COVID-19 pneumonia.[37]
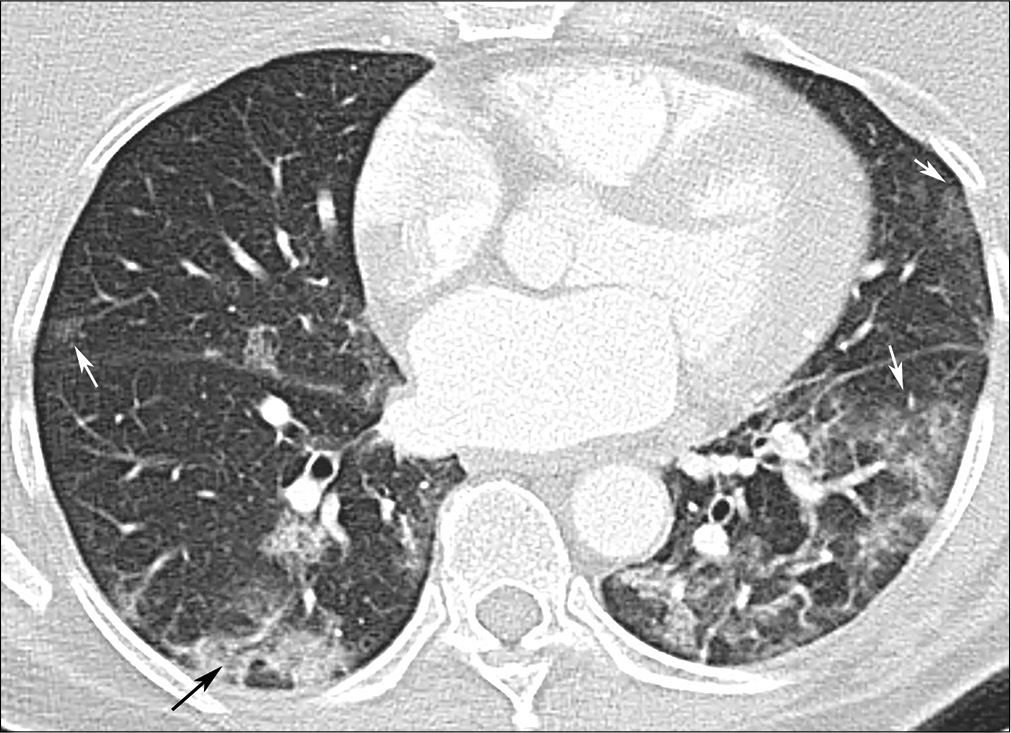
- A 61-year-old female with coronavirus disease 2019 pneumonia. Ground glass opacities GGO (white arrows), predominantly in the left lower lobe, but also in the right middle lobe and the lingula. Right lower lobe consolidation with air bronchogram (black arrow).
Consolidation [Figure 1] is the second most common pattern seen in patients with COVID-19 and may present in isolation or in combination with GGO.[16,20,25,27,35-40] Consolidation and GGO together represented 83–85% of abnormalities seen by Wang et al. in their series of 90 patients.[41] Consolidation represents more severe disease, compared to GGO. A prospective analysis of 41 patients from Wuhan, China, showed patients admitted to the intensive care unit were more likely to have consolidations that were bilateral and larger (lobular pattern), compared to patients who were not admitted to the intensive care unit who had bilateral GGO and/or smaller (segmental) areas of consolidation.[42] Similarly, Song et al. in their series found consolidations tend to occur more often in older patients compared to younger patients, who have more GGO.[18] Crazypaving refers to the presence of GGO with superimposed interlacing network of lines corresponding to thickening of inter and intralobular septa, similar to the appearance of irregularly shaped paving stones.[41-43] This sign has been reported in CT scans of 5–36% COVID-19 patients [Figures 2 and 3].[24,27,28,44,45] These imaging patterns of peripheral and lower predominant GGO, consolidations, and crazy paving have previously described in diffuse alveolar damage in response to acute lung injury.[46,47]
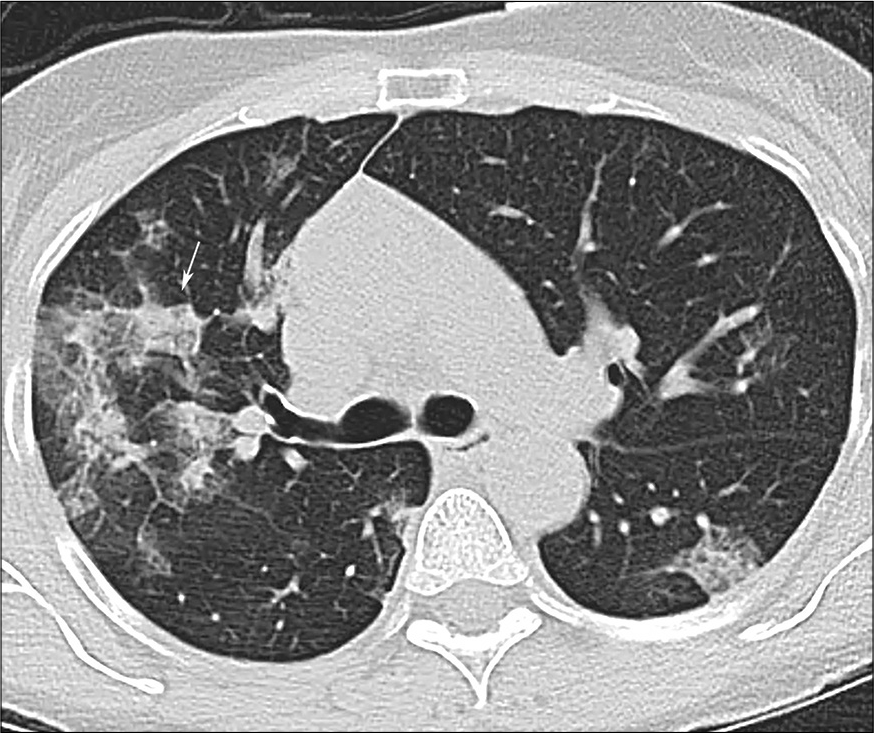
- A 58-year-old male with COVID-19 pneumonia. Crazy paving sign in the right upper lobe (arrow).
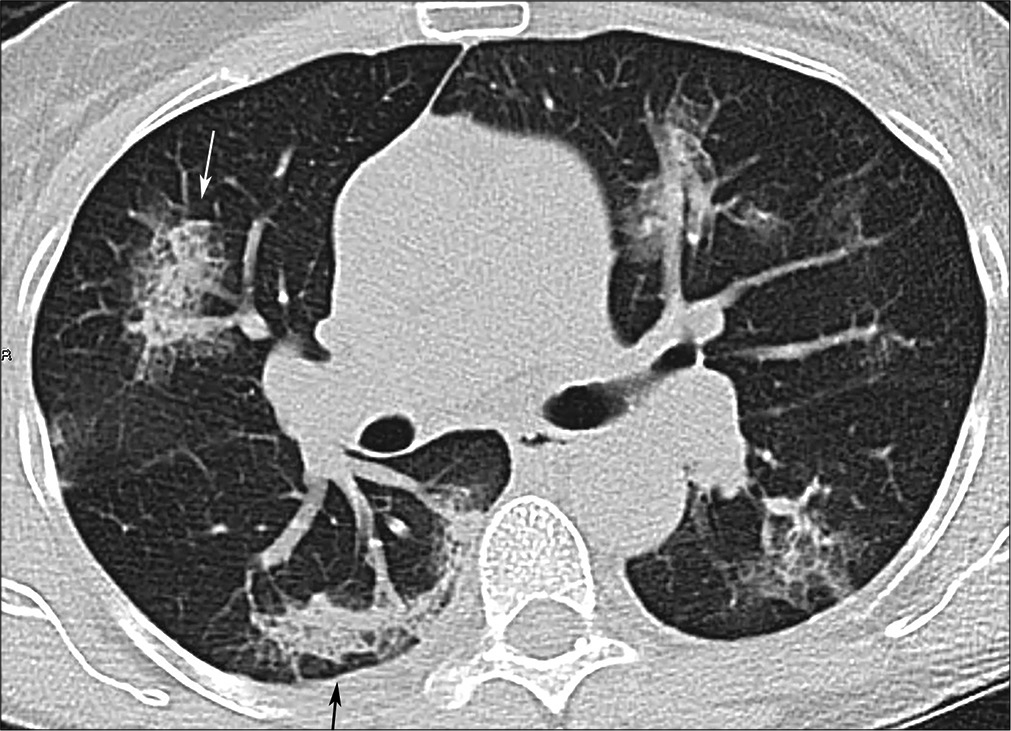
- A 58-year-old male with coronavirus disease 2019 pneumonia, same patient as Figure 2, more inferior slice. “Rounded” consolidation and crazy paving in the right upper lobe (white arrow); developing reticulation in the right lower lobe (black arrow).
In addition to the patterns of lung involvement described above, few additional signs have been described. A “rounded morphology” of the consolidation and GGO has been described in 18–33% of patients [Figure 3].[25,39] Vascular thickening, or microvascular enlargement sign[48] consists of enlarged small vessels within the lesions,[49] has been described in 45–80% of patients with COVID-19 pneumonia [Figure 4].[17,41,51] The sign is presumably related to an inflammatory response and release of pro-inflammatory factors.[37,41] Nodules are seen in 2–13% of CT scans in patient with COVID-19 infection,[12,23,49] are usually solid, irregular multifocal, may enlarge or coalesce on follow-up studies or may heal by reduction in density to ground-glass (the melting sugar appearance).[23,52] Fibrous stripes, described by Pan et al. in 17% of patients in their study[23] are linear densities that may appear early and evolve into consolidative areas or appear late in the disease as a sign of healing and organization [Figure 5].[23,37] Other signs of organization and healing by fibrosis[46] include subpleural bands [Figure 6], a reticular pattern [Figure 3], that is, fine linear opacities that by summation produce a net like pattern,[18,45] reversed halo sign [Figure 7] or a ring of peripheral consolidation with central lucency,[27,37,51] and traction bronchiectasis [Figure 8].[50]
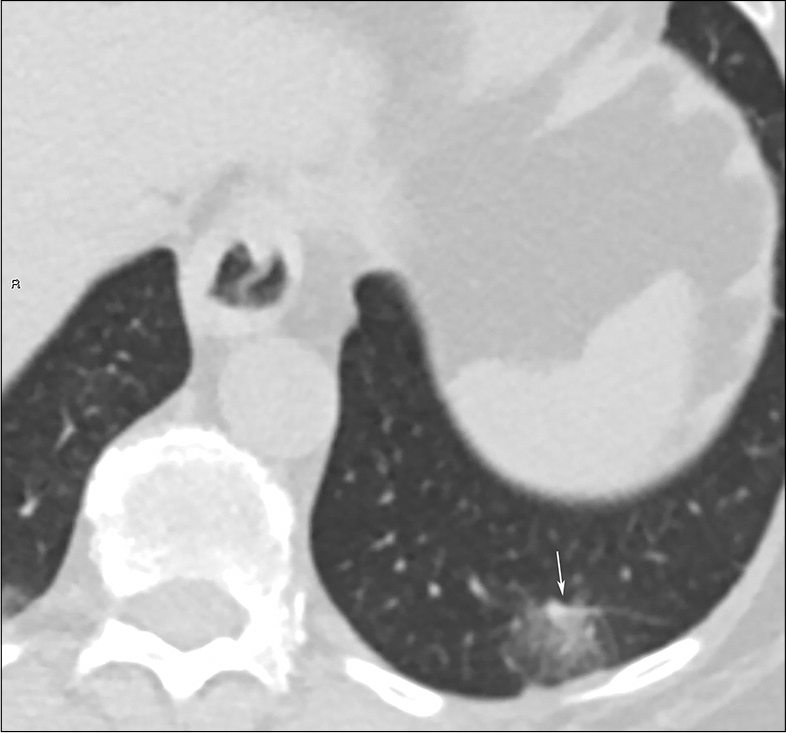
- A 69-year-old female with coronavirus disease 2019 pneumonia. GGO with vascular enlargement sign in the left lower lobe (white arrow).
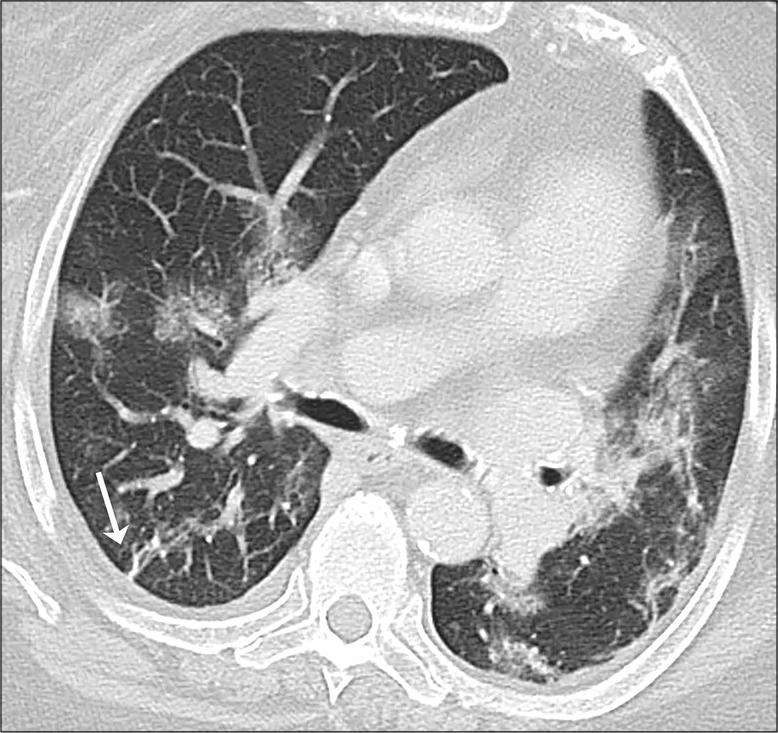
- A 71-year-old female with coronavirus disease 2019 pneumonia. “Fibrous stripe” (arrow) in the right lower lobe. Other bilateral GGO and consolidations.
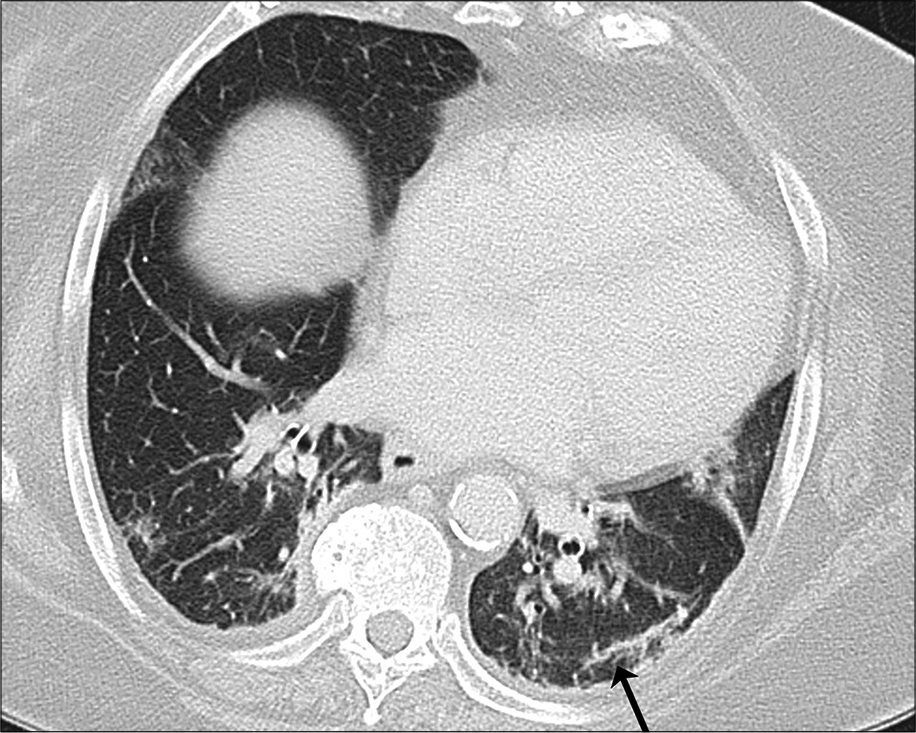
- A 71-year-old with coronavirus disease 2019 pneumonia, same patient as in Figure 6, lower slice. Subpleural band (black arrow) in the right lower lobe.
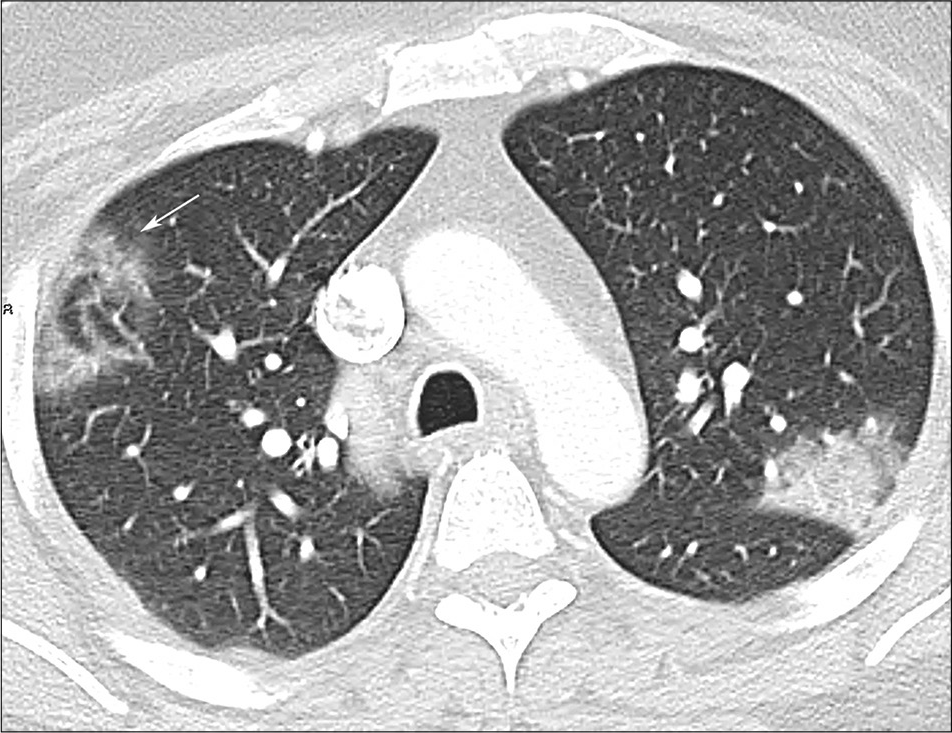
- A 43-year-old male with coronavirus disease 2019 pneumonia. Reversed halo sign in the right upper lobe (arrow).
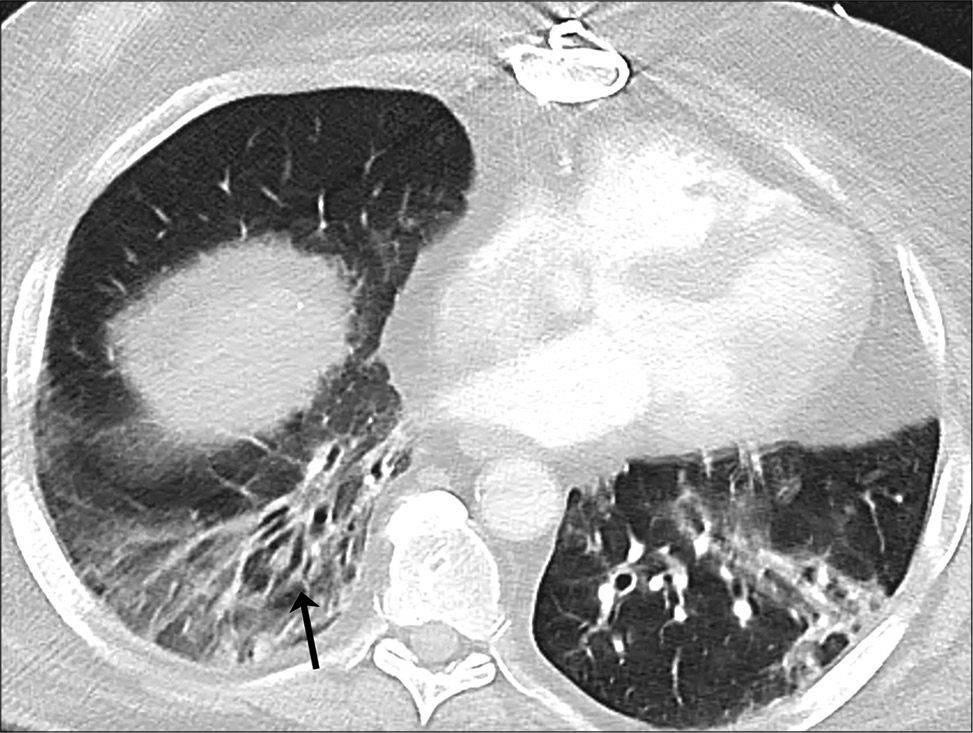
- A 48-year-old female with coronavirus disease 2019 pneumonia. Traction bronchiectasis in the right lower lobe (arrow) with adjacent GGO.
Significantly, cavitation and tree in bud signs are absent.[24,42] Pleural effusion appears to be an unusual finding, seen in only one of 121 patients by Bernheim et al.[27] It is relatively rare in early phase disease (<7 days) and appears to be a marker of either advanced[17,24] or severe[50] disease. Other CT markers of disease severity and high viral load described by Zhao et al. include architectural distortion and traction bronchiectasis. Intrathoracic lymph node enlargement is also a rare finding,[27,50] for example, seen by Zhao et al. in only one patient in a series of 101, in a patient with severe clinical disease.
There is a high incidence of acute pulmonary embolism associated with COVID-19 infection,[53,54] reported to be between 22 and 30% among patients who received contrast enhanced CT angiography,[55-57] this risk appears to be higher than that seen in other critically ill patients (1.3%)[58] and emergency room populations (3– 10%).[55,59] On autopsy examination, the lungs of patients who died of COVID-19 demonstrated widespread vascular thrombosis and microangiopathy.[32] Poyiadi et al. observed a reduced risk of pulmonary thromboembolism in COVID-19 patients who had previously been on statin therapy.[57]
CT findings are observed in asymptomatic individuals with positive RT-PCR.[60] GGO appears to be the predominant presentation in asymptomatic patients, seen in 95% of patients, with consolidation seen in the remaining 5%.[60] The distribution is predominantly peripheral, subpleural and lower lobe with a tendency to be confined to one or two lobes (65%). Other signs of COVID-19 pneumonia including reticulation, subpleural curvilinear sign, air bronchogram, and vascular enlargement may be present.[60]
In view of the vast spectrum of findings of CT findings, described in the literature; CT findings maybe categorized as typical, indeterminate, and atypical for COVID-19, with typical features being bilateral peripheral ground glass opacities, with or without consolidation [Figure 1] and crazy paving [Figure 2]; “rounded” morphology lesions [Figure 3] and later in the disease, signs of organizing pneumonia such as reverse halo sign [Figure 7]. Atypical signs on CT, where alternative or additional diseases should be considered include tree in bud nodularity or lung cavitation.[61]
At the time of writing this article, there is limited data to predict outcomes and prognosis based on appearance on imaging. Shi et al. have described imaging appearances in three patients who died of COVID-19 pneumonia.[24] The first patient had underlying chronic lung disease (tuberculosis), and complete opacification of lung parenchyma (white out lung) at presentation. The other two patients demonstrated rapid imaging progression on serial CT scans and one developed a pleural effusion.[24] Colombi et al. quantified the percentage of lung parenchyma not involved by disease on CT (well aerated lung) visually, using segmentation software and also determined the absolute volume of uninvolved lung and they found all three models were superior to clinical parameters alone in predicting intensive care unit admission or death.[62]
TIME COURSE EVOLUTION OF CT FINDINGS
Pan et al.[26] have described the time course of lung changes in 21 patients with mild to moderate COVID-19 pneumonia, and a mean hospitalization period of 17 days, with serial CT scans obtained approximately 4 days apart. They identified four distinct stages of lung involvement (a) early stage; days 0–4 with GGO (75%) being the predominant abnormality, (b) progressive stage; days 5–8 increasing crazy paving (53%); (c) peak stage; days 9–13 with predominance of consolidation (91% of patients); and (d) absorption phase; after day 14 with gradual absorption of consolidation and residual ground glass opacities and subpleural bands. The extent of lung involvement was maximum on day 10 after onset of symptoms. Wang et al. in their prospective longitudinal series of 90 hospitalized patients analyzed for evolution of CT findings similarly showed peak lung involvement on days 6–11.[41] In their series which included severe cases, the extent of lung involvement showed a slow decline after peak involvement. From days 12 to 17 onward they observed a mixed pattern, with architectural distortion and perilobular distribution, suggestive of development of organizing pneumonia.[41]
PERFORMANCE OF CT AS A DIAGNOSTIC MODALITY COMPARED TO RT-PCR
Ai et al. in their retrospective analysis of 1014 patients evaluated during peak endemicity in Wuhan, China (January 6 to February 6 2020) found an initial positive detection rate of 88% (888/1014) for CT and 59% (601/1014) for RT-PCR.[12] Similarly, Fang et al. in their analysis of 51 high risk patients, studied by serial RT-PCR and chest CTs, found an initial detection rate of 98% by CT (95% CI 90–100%), and compared to 71% by RT-PCR (95% CI 56– 83%) (P < 0.001).[63] These low rates of positive detection by RT-PCR using nasopharyngeal swabs are similar to those reported by Wang et al.[12] and may be related to variability between manufacturers, initial low viral load or improper clinical sampling techniques.[52,61-67] On imaging, patients with clinical suspicion of COVID-19 pneumonia who have initial negative RT-PCR that subsequently turns positive on repeat testing tend to have more ground glass and less of consolidation pattern, suggestive of early stage disease and presumably, low viral load.[65,68] In contrast in areas of low COVID-19 prevalence (<10%), Kim et al. in their meta-analysis found that the positive predictive value of RT-PCR (range: 47.3%, –84.3%) was more than 10 times higher than that of CT scans (range: 1.5–8.3%).[69]
The sensitivity and specificity of CT for the detection of COVID-19 pneumonia are hard to estimate precisely, in view of absence of a robust gold standard. Using RT-PCR as a reference standard Ai et al. estimated the sensitivity, specificity, and accuracy of CT in their series of 1014 patients to be 97%, 25%, and 68%, respectively.[12] However, the authors of this study acknowledged that the relatively low positive detection rate of the reference standard likely led to overestimation of sensitivity and underestimation of specificity.[12] Other studies and meta-analysis have similarly demonstrated low specificity for CT in the diagnosis of COVID-19.[69,70] The low specificity of CT for the diagnosis of COVID-19 pneumonia is due to the overlap of CT findings with other viral pneumonias and with other diffuse diseases of the chest such as other infections, lung involvement in connective tissue disorders, drug reaction, pulmonary edema, and hemorrhage.[24,31,71]
Literature describing the performance of radiologists in correctly distinguishing COVID-19 pneumonia from other viral pneumonias or infections is limited. In the study by Bai et al.,[16] six out of seven radiologists reached a specificity of 93–100% in correctly distinguishing COVID-19 infection from other viral pneumonias.[16,72] In their study, the most useful discriminating features for COVID-19 pneumonia from other pneumonias were a peripheral distribution (80% vs. 57%, P < 0.001), GGO (91% vs. 68%, P < 0.001), and vascular thickening (58% vs. 22%, P < 0.001).[16] There was been some interest in the role of artificial intelligence (AI) in diagnosing COVID-19 pneumonia with variable results.[73,74] Ito R published a review of 17 studies that used AI with four studies reaching very high sensitivity, specificity, and AUC, in the range of 0.9–0.98, 0.91–0.96, and 0.96–0.99, respectively.[73]
RECOMMENDATIONS FOR THE ROLE OF CT IN COVID-19 PNEUMONIA
Fleischner Society has provided a statement for the role of imaging (chest X-ray and CT scan) in directing patient management in various clinical scenarios.[15] When patients presenting with mild respiratory features of COVID-19 and either suspected or confirmed laboratory diagnosis, imaging is recommended only in the setting of risk factors for disease progression, with common risk factors being age >65 years, cardiovascular disease, diabetes, chronic respiratory disease, hypertension, and immune-compromised status.[15,19,75,76] In patients with clinical presentation of moderate to severe respiratory features of COVID-19 disease (e.g., hypoxemia, moderate to severe dyspnea) imaging is recommended irrespective of laboratory test results. In patients with moderate to severe respiratory features of COVID-19 disease, within an environment of high disease burden, when resources are limited such as medical beds, ventilators, PFE, and no rapid COVID-19 tests, CT can be used to make triage decisions. The society did emphasize that although CT is more sensitive, depending on local expertise and availability, if access to CT is limited, chest X-ray may be the preferred modality; and CT being warranted in circumstances of respiratory worsening.[15] In addition, the Fleischner Society maintains an online repository of characteristic images of proven COVID-19 cases for teaching purposes.[72]
CONCLUSION
CT scan can help in diagnosis of COVID-19 in the appropriate setting, set a baseline for patients who are at risk for progression, may help to suggest alternate diagnosis and co-morbidities in the high risk patient with COVID-19. In resource limited situations and high disease burden, it is a tool for rapid triage. As the disease and pandemic evolves, there may be further updates in the role of CT in disease management and containment.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Pneumonia of Unknown Cause -China. 2020. Available from: https://www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-cause-china/en [Last accessed on 2021 May 19]
- [Google Scholar]
- A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727-33.
- [CrossRef] [PubMed] [Google Scholar]
- Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382:1564-7.
- [CrossRef] [PubMed] [Google Scholar]
- Center for Systems Science and Engineering. 2020. Available from: https://www.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6 [Last accessed on 2020 Mar 08]
- [Google Scholar]
- Remdesivir in adults with severe COVID-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395:1569-78.
- [CrossRef] [Google Scholar]
- Remdesivir for the treatment of Covid-19-final report. N Engl J Med. 2020;383:1813-26.
- [CrossRef] [PubMed] [Google Scholar]
- A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382:1787-99.
- [CrossRef] [PubMed] [Google Scholar]
- Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;382:2411-8.
- [CrossRef] [PubMed] [Google Scholar]
- Stability issues of RTPCR testing of SARS-CoV-2 for hospitalized patients clinically diagnosed with COVID-19. J Med Virol. 2020;92:903-8.
- [CrossRef] [PubMed] [Google Scholar]
- Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843-4.
- [CrossRef] [PubMed] [Google Scholar]
- Dynamic profile of RT-PCR findings from 301 COVID-19 patients in Wuhan, China: A descriptive study. J Clin Virol. 2020;127:104346.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation of chest CT and RT-PCR testing for Coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology. 2020;296:E32-40.
- [CrossRef] [PubMed] [Google Scholar]
- Targeted early chest CT in COVID-19 outbreaks as diagnostic tool for containment of the pandemic-A multinational opinion. Diagn Interv Radiol. 2020;26:292-5.
- [CrossRef] [PubMed] [Google Scholar]
- An update on COVID-19 for the radiologist-A British Society of thoracic imaging statement. Clin Radiol. 2020;75:323-5.
- [CrossRef] [PubMed] [Google Scholar]
- The role of chest imaging in patient management during the COVID-19 pandemic: A multinational consensus statement from the Fleischner society. Chest. 2020;158:106-16.
- [CrossRef] [PubMed] [Google Scholar]
- Performance of radiologists in differentiating COVID-19 from non-COVID-19 viral pneumonia at chest CT. Radiology. 2020;296:E46-54.
- [CrossRef] [PubMed] [Google Scholar]
- CT features of coronavirus disease 2019 (COVID-19) pneumonia in 62 patients in Wuhan, China. AJR Am J Roentgenol. 2020;214:1287-94.
- [CrossRef] [PubMed] [Google Scholar]
- Emerging 2019 Novel Coronavirus (2019-nCoV) Pneumonia. Radiology. 2020;295:210-7.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061-9.
- [CrossRef] [PubMed] [Google Scholar]
- Coronavirus disease 2019 (COVID-19): A systematic review of imaging findings in 919 patients. AJR Am J Roentgenol. 2020;215:87-93.
- [CrossRef] [PubMed] [Google Scholar]
- Fleischner Society: Glossary of terms for thoracic imaging. Radiology. 2008;246:697-722.
- [CrossRef] [PubMed] [Google Scholar]
- Chest CT findings in patients with coronavirus disease 2019 and its relationship with clinical features. Invest Radiol. 2020;55:257-61.
- [CrossRef] [PubMed] [Google Scholar]
- Initial CT findings and temporal changes in patients with the novel coronavirus pneumonia (2019-nCoV): A study of 63 patients in Wuhan, China. Eur Radiol. 2020;30:3306-9.
- [CrossRef] [PubMed] [Google Scholar]
- Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: A descriptive study. Lancet Infect Dis. 2020;20:425-34.
- [CrossRef] [Google Scholar]
- CT imaging features of 2019 novel coronavirus (2019-nCoV) Radiology. 2020;295:202-7.
- [CrossRef] [PubMed] [Google Scholar]
- Time course of lung changes at chest CT during recovery from coronavirus disease 2019 (COVID-19) Radiology. 2020;295:715-21.
- [CrossRef] [PubMed] [Google Scholar]
- Chest CT findings in coronavirus disease-19 (COVID-19): Relationship to duration of infection. Radiology. 2020;295:200463.
- [CrossRef] [PubMed] [Google Scholar]
- The clinical and chest CT features associated with severe and critical COVID-19 pneumonia. Invest Radiol. 2020;55:327-31.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet. 2020;395:507-13.
- [CrossRef] [Google Scholar]
- Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-20.
- [CrossRef] [PubMed] [Google Scholar]
- Coronavirus disease 2019 (COVID-19): Role of chest CT in diagnosis and management. AJR Am J Roentgenol. 2020;214:1280-6.
- [CrossRef] [PubMed] [Google Scholar]
- Role of computed tomography in COVID-19. J Cardiovasc Comput Tomogr. 2021;15:27-36.
- [CrossRef] [PubMed] [Google Scholar]
- Chest CT in COVID-19: What the radiologist needs to know. Radiographics. 2020;40:1848-65.
- [CrossRef] [PubMed] [Google Scholar]
- Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020;383:120-8.
- [CrossRef] [PubMed] [Google Scholar]
- Pulmonary pathology of early-phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J Thorac Oncol. 2020;15:700-4.
- [CrossRef] [PubMed] [Google Scholar]
- Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-2.
- [CrossRef] [Google Scholar]
- Chest CT manifestations of new coronavirus disease 2019 (COVID-19): A pictorial review. Eur Radiol. 2020;30:4381-9.
- [CrossRef] [PubMed] [Google Scholar]
- Chest CT features of COVID-19 in Rome, Italy. Radiology. 2020;296:E79-85.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical features and chest CT manifestations of coronavirus disease 2019 (COVID-19) in a single-center study in Shanghai, China. AJR Am J Roentgenol. 2020;215:121-6.
- [CrossRef] [PubMed] [Google Scholar]
- CT findings of coronavirus disease (COVID-19) severe pneumonia. AJR Am J Roentgenol. 2020;214:W85-6.
- [CrossRef] [PubMed] [Google Scholar]
- Temporal changes of CT findings in 90 patients with COVID-19 pneumonia: A longitudinal study. Radiology. 2020;296:E55-64.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506.
- [CrossRef] [Google Scholar]
- "Crazy paving appearance" on high resolution CT in various diseases. J Comput Assist Tomogr. 1999;23:749-52.
- [CrossRef] [PubMed] [Google Scholar]
- "Crazy-paving" pattern at thin-section CT of the lungs: Radiologic-pathologic overview. Radiographics. 2003;23:1509-19.
- [CrossRef] [PubMed] [Google Scholar]
- Coronavirus disease 2019: Initial chest CT findings. Eur Radiol. 2020;30:4398-406.
- [CrossRef] [PubMed] [Google Scholar]
- From the radiologic pathology archives: Organization and fibrosis as a response to lung injury in diffuse alveolar damage, organizing pneumonia, and acute fibrinous and organizing pneumonia. Radiographics. 2013;33:1951-75.
- [CrossRef] [PubMed] [Google Scholar]
- Crazy-paving appearance at thin-section CT: Spectrum of disease and pathologic findings. Radiology. 1999;211:155-60.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular changes detected with thoracic CT in coronavirus disease (COVID-19) might be significant determinants for accurate diagnosis and optimal patient management. AJR Am J Roentgenol. 2020;215:W15.
- [CrossRef] [PubMed] [Google Scholar]
- COVID-19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: Single-center study and comprehensive radiologic literature review. Eur J Radiol Open. 2020;7:100231.
- [CrossRef] [PubMed] [Google Scholar]
- Relation between chest CT findings and clinical conditions of coronavirus disease (COVID-19) pneumonia: A multicenter study. AJR Am J Roentgenol. 2020;214:1072-7.
- [CrossRef] [PubMed] [Google Scholar]
- Early clinical and CT manifestations of coronavirus disease 2019 (COVID-19) pneumonia. AJR Am J Roentgenol. 2020;215:338-43.
- [CrossRef] [PubMed] [Google Scholar]
- Use of chest CT in combination with negative RT-PCR assay for the 2019 novel coronavirus but high clinical suspicion. Radiology. 2020;295:22-3.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnosis, prevention, and treatment of thromboembolic complications in COVID-19: Report of the national institute for public health of the Netherlands. Radiology. 2020;297:E216-222.
- [CrossRef] [PubMed] [Google Scholar]
- COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135:2033-40.
- [CrossRef] [PubMed] [Google Scholar]
- Acute pulmonary embolism in patients with COVID-19 at CT angiography and relationship to d-dimer levels. Radiology. 2020;296:E189-91.
- [CrossRef] [PubMed] [Google Scholar]
- Acute pulmonary embolism associated with COVID-19 pneumonia detected with pulmonary CT angiography. Radiology. 2020;296:E186-8.
- [CrossRef] [PubMed] [Google Scholar]
- Acute pulmonary embolism and COVID-19. Radiology. 2020;297:E335-6.
- [CrossRef] [PubMed] [Google Scholar]
- Failure of anticoagulant thromboprophylaxis: Risk factors in medical-surgical critically ill patients*. Crit Care Med. 2015;43:401-10.
- [CrossRef] [PubMed] [Google Scholar]
- Pulmonary embolism: The diagnosis, risk-stratification, treatment and disposition of emergency department patients. Clin Exp Emerg Med. 2016;3:117-25.
- [CrossRef] [PubMed] [Google Scholar]
- CT imaging and clinical course of asymptomatic cases with COVID-19 pneumonia at admission in Wuhan, China. J Infect. 2020;81:e33-9.
- [CrossRef] [PubMed] [Google Scholar]
- Radiological society of North America expert consensus document on reporting chest CT findings related to COVID-19: Endorsed by the society of thoracic radiology, the American College of Radiology, and RSNA. J Thorac Imaging. 2020;35:219-27.
- [CrossRef] [PubMed] [Google Scholar]
- Well-aerated lung on admitting chest CT to predict adverse outcome in COVID-19 pneumonia. Radiology. 2020;296:E86-96.
- [CrossRef] [PubMed] [Google Scholar]
- Sensitivity of chest CT for COVID-19: Comparison to RT-PCR. Radiology. 2020;296:E115-7.
- [CrossRef] [PubMed] [Google Scholar]
- A meta-analysis to evaluate the effectiveness of real-time PCR for diagnosing novel coronavirus infections. Genet Mol Res. 2015;14:15634-41.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical diagnostic value of CT imaging in COVID-19 with multiple negative RT-PCR testing. Travel Med Infect Dis. 2020;34:101627.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnostic performance between CT and initial real-time RT-PCR for clinically suspected 2019 coronavirus disease (COVID-19) patients outside Wuhan, China. Respir Med. 2020;168:105980.
- [CrossRef] [PubMed] [Google Scholar]
- Potential preanalytical and analytical vulnerabilities in the laboratory diagnosis of coronavirus disease 2019 (COVID-19) Clin Chem Lab Med. 2020;58:1070-6.
- [CrossRef] [PubMed] [Google Scholar]
- Can chest CT features distinguish patients with negative from those with positive initial RT-PCR results for coronavirus disease (COVID-19)? AJR Am J Roentgenol. 2020;216:66-70.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnostic performance of CT and reverse transcriptase polymerase chain reaction for coronavirus disease 2019: A meta-analysis. Radiology. 2020;296:E145-55.
- [CrossRef] [PubMed] [Google Scholar]
- Systematic review and meta-analysis on the value of chest CT in the diagnosis of coronavirus disease (COVID-19): Sol Scientiae, Illustra Nos. AJR Am J Roentgenol. 2020;215:1342-50.
- [CrossRef] [PubMed] [Google Scholar]
- Chest CT and coronavirus disease (COVID-19): A critical review of the literature to date. AJR Am J Roentgenol. 2020;215:839-42.
- [CrossRef] [Google Scholar]
- Teaching Cases. 2020. Available from: https://www.fleischner-covid19.org/# [Last accessed on 2020 May 28]
- [Google Scholar]
- A review on the use of artificial intelligence for medical imaging of the lungs of patients with coronavirus disease 2019. Diagn Interv Radiol. 2020;26:443-8.
- [CrossRef] [PubMed] [Google Scholar]
- Clinically applicable AI system for accurate diagnosis, quantitative measurements, and prognosis of COVID-19 pneumonia using computed tomography. Cell. 2020;181:1423-33.
- [CrossRef] [PubMed] [Google Scholar]
- Multi-detector spiral CT study of the relationships between pulmonary ground-glass nodules and blood vessels. Eur Radiol. 2013;23:3271-7.
- [CrossRef] [PubMed] [Google Scholar]
- Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239-42.
- [CrossRef] [PubMed] [Google Scholar]






