Translate this page into:
Gastric mucormycosis with splenic invasion a rare abdominal complication of COVID-19 pneumonia

*Corresponding author: Pavankumar M. Mathapati, Department of Radio-diagnosis, Saveetha Medical College and Hospital, Chennai, Tamil Nadu, India. pavankumarmathapati@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Yuvaraj M, Mathapati PM, Seena CR, Ramaswami S. Gastric mucormycosis with splenic invasion a rare abdominal complication of COVID-19 pneumonia. J Clin Imaging Sci 2021;11:62.
Abstract
Mucormycosis (previously called zygomycosis or phycomycosis) is an uncommon opportunistic infection with the gastrointestinal form being the rarest occurrence and one of the most lethal infections known to humanity. The most common risk factors predisposing to develop gastric mucormycosis are uncontrolled diabetes mellitus, stem cell transplantation, or underlying any hematologic malignancy and major trauma. Pain abdomen, hematemesis, and melena are common symptoms which the patient presents. The exact diagnosis of gastric mucormycosis can be missed due rarity of the disease. A high level of suspicion is required in the early diagnosis and management of disease, particularly in immunocompromised patients. The radiological imaging modalities such as CT scan or MRI of the abdomen initially usually reveal non-specific findings such as mucosal wall thickening, mass, and reactive lymphadenopathy and prompt additional investigation with endoscopic or surgical biopsy of the lesions. The disease outcome and mortality are very high with gastrointestinal mucormycosis.
Keywords
Gastric mucormycosis
COVID-19 pneumonia
Zygomycosis
Phycomycosis
Rhizopus
INTRODUCTION
Gastric mucormycosis occurs due to the invasion of filamentous fungus (Mucorales) into the mucosa of the stomach resulting in high mortality (up to 54%) predominantly in immunocompromised patients.[1] Approximately 75% of mucormycosis infection is caused by fungi class known as zygomycetes and particularly Rhizopus, Rhizomucorspecies, or Mucor.[1-3] The most common sites being the paranasal sinuses, upper respiratory tract, orbit, brain, and skin; out of these sites, the gastrointestinal tract infection is infrequent.[4] Serologic biomarkers are unremarkable; yet, a biopsy from the infected region forms the gold standard in histological diagnosis of mucormycosis. Polymerase chain reaction or positive culture of the biopsied specimen is rarely required for confirmation of diagnosis. It can be managed medically in the early stage with antifungal medications known as lipid formulation of posaconazole, amphotericin B, and a few of the newer agents such as triazole or isavuconazole and these remain the primary choice in treating the patients who are affected with gastric mucormycosis.[5,6] Further in the progression of diseases that results in the necrosis of the tissue and delayed presentation of mucormycosis, can be managed medically with early surgical resection of the affected site which results in a better outcome of the condition. Here, we present a patient who was diagnosed with COVID-19 after using immunosuppressant (steroid) for treating COVID-19 with a known case of DM and developed gastric mucormycosis.
CASE REPORT
A 53-year-old male with a medical history of diabetes mellitus (DM) and alcoholic abuse presented to the emergency room with acute abdominal pain, associated with Hematemesis – 3 episodes and Malena – 2 episodes, since 3 days. The abdominal pain was predominantly in the left hypochondrium and epigastrium which were non-radiating, without precipitating or relieving factors. On physical examination, vitals were as follows: BP – 110/70 mmHg, PR – 63/min, RR – 20/min, and Spo2 – 99 % at room air. Epigastric tenderness was noted on abdominal examination. Laboratory workup showed mild leukocytosis (14700 Cells/cumm) and reduced HB −8.6 g/ DL. Then, patient was further advised computed tomography (CT) of the abdomen and pelvis, initially, plain CT was advised and it showed an exophytic ulcerative lesion along the greater curvature of the stomach associate with perigastric and perisplenic fat stranding [Figure 1] and further on contrast-enhanced CT and it demonstrated focal asymmetrical wall thickening with exophytic ulcerative component involving fundus and the proximal body along the greater curvature of the stomach extending up to the splenic hilum causing splenic infarction (vascular invasion) and associated perigastric inflammation and reactive adenopathy [Figure 2]. Then, the patient was advised for esophagogastroduodenoscopy (EGD) and it revealed proliferative growth in the region of the gastric fundus with surrounding fresh or altered blood and exudative material [Figure 6], then further, on worsening of pain follow-up contrast-enhanced computed tomography (CECT) was taken and it revealed the formation of the gastrosplenic fistulous communication and liquefaction of the splenic infracted parenchyma [Figure 3], Corresponding USG findings during follow-up revealed presence of few echogenic foci in the splenic parenchyma (air pockets) - [Figure 4] and further on biopsy of the mass revealed as lamina propria showing lymphoplasmacytic and minimal neutrophilic infiltrate, necro-hemorrhagic fragment, infiltration by broad aseptate branched fungal hyphae, with no evidence of intestinal metaplasia [Figure 5]. After confirmation of gastric mucormycosis, the patient was started with amphotericin-B 300 mg in 500 ml 5% dextrose IV for initial hours and later was planned for gastrectomy + splenectomy; however, the outcome of the patient was grave before the surgery, the gastric mucormycosis was considered the possible cause of death.
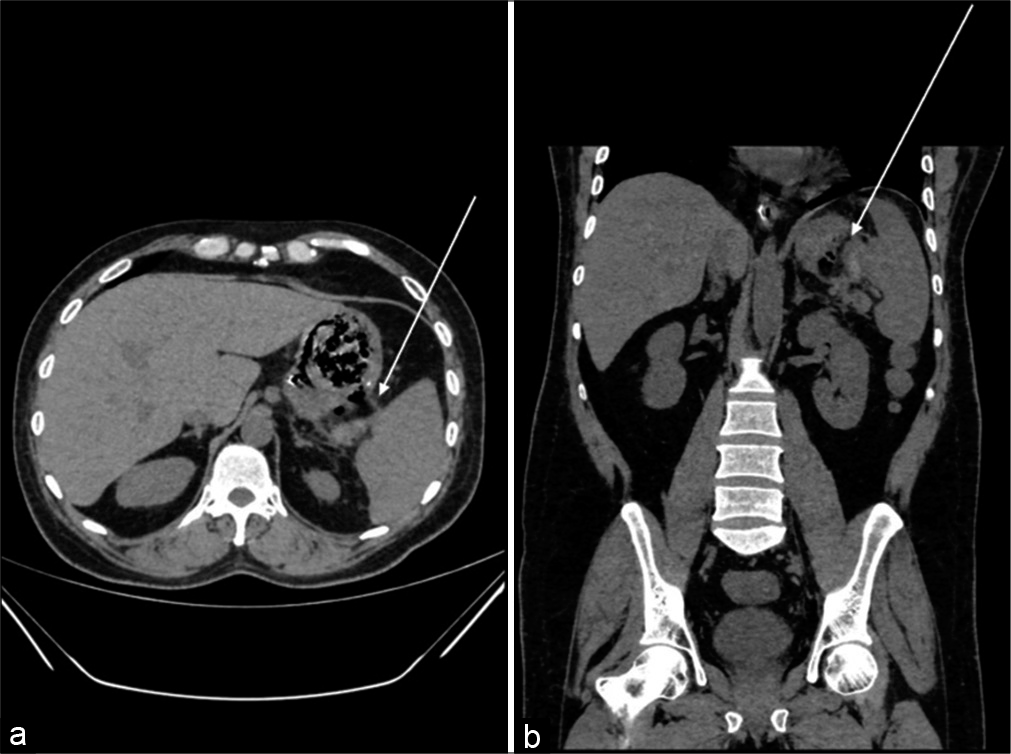
- A 53-year-old male presented to the emergency room with acute abdominal pain, Hematemesis, Malena non-contrast-enhanced computed tomography (axial and coronal reformatted sections) (a and b) – Abdomen shows an exophytic ulcerative lesion seen along the greater curvature of stomach associate with perigastric and peri splenic fat stranding (white arrow).

- A 53-year-old male presented to the emergency room with acute abdominal pain, Hematemesis, Malena contrast-enhanced computed tomography (axial section and coronal) (a and b) – Abdomen shows an exophytic enhancing ulcerative component seen arising from the proximal body of the stomach and extending up to the splenic hilum with loss of fat plane (white arrow) and non-enhancing wedge-shaped area with the apex toward the splenic hilum - splenic infarction (yellow arrow).

- A 53-year-old male presented to the emergency room with acute abdominal pain, Hematemesis, Malena contrast enhanced computed tomography – Abdomen on follow-up imaging shows an exophytic enhancing ulcerative component from the proximal body of the stomach and extending up to the splenic hilum with fistulous communication between the spleen and the stomach (white arrow) and few air pockets noted within (yellow arrow).

- A 53-year-old male presented to the emergency room with acute abdominal pain, Hematemesis, Malena on follow-up scan - USG image shows – There is the presence of few echogenic foci (air pockets) in the splenic parenchyma (white arrow).

- Multiple serial sections studied show (a) H and E stain, scanner view shows fragments of gastric mucosa with the necrotic area on the luminal side. (b) H and E ×40, high power view shows enterohemorrhagic fragments with broad aseptate branched fungal hyphae (indicated by arrow) (c) GMS Giemsa stain shows fungal hyphae stained blue (indicated by arrow) (d) GMS stain showing fungal hyphae stained black; however, there is no intestinal metaplasia or dysplasia.

- Multiple serial images of esophagogastroduodenoscopy at different angulation show (a) exudative material (indicated by the blue arrow) (b) proliferative growth in the region of the gastric fundus (indicated by white arrows) (c) and (d) fresh or altered blood (indicated by black arrows).
DISCUSSION
An estimated prevalence of mucormycosis is 0.16 (0.12–0.20)/10,000 patients.[7] The prevalence of patients with uncontrolled diabetes who are diagnosed with mucormycosis is 36%.[8] Here, we presented a patient with acute gastric mucormycosis complicated by a splenic infarct in a patient with H/o COVID-19 after treating with immunosuppressant (steroids) and known diabetes, uncontrolled diabetes, H/o COVID-19, and steroid treatment were major risk factors predisposing to the development of gastric mucormycosis. The patient was initially under symptomatic treatment and was not responsive to proton pump inhibitors which prompted further analysis including abdominal radiological imaging and upper gastrointestinal endoscopy. Initially, radiological imaging plain CT shows an exophytic ulcerative lesion along the greater curvature of the stomach [Figure 1] and (CECT) revealed focal asymmetrical wall thickening with exophytic ulcerative component involving fundus and proximal body along the greater curvature of the stomach extending up to the splenic hilum causing splenic infarction, with associated perigastric inflammation and reactive adenopathy [Figure 2] and further he was advised (EGD) and it revealed proliferative growth in the region of the gastric fundus with surrounding fresh and altered blood [Figure 6]. These mucosal lesions were covered with a large greyish exudate, biopsy from the region of interest was taken and diagnosis of gastric mucormycosis was further confirmed with histological examination. Further, he was immediately started with amphotericin-B 300 mg in 500 ml 5% dextrose IV for initial hours, follow-up CECT took which shows the formation of the gastro-splenic fistulous communication and liquefaction of the splenic infracted parenchyma and later was planned for gastrectomy + splenectomy, due to lethal nature of disease the patient did not survive.
Gastrointestinal mucosal involvement could be the unique manifestation of mucormycosis that accounts for approximately 7% of all reported cases.[1] Among the gastrointestinal mucormycosis, the stomach is the most commonly affected organ (67%), then followed by the large bowel (21%), small bowel (4%), and esophagus being the least (2%).[1,4,8,9] Inhalation, inoculation, or ingestion of the spores being the primary mode of transmission in humans.[8] The primary mode of transmission of Mucorales to the gastrointestinal tract is by ingestion of the spores possibly from which are present in fermented milk, porridges, dried bread products, and alcoholic beverages which are predominantly derived from infected corn, and even from unsterilized/infected tongue depressors at clinics.[2] An incident of iatrogenic gastric mucormycosis was reported in the literature from using a wooden tongue depressor and wooden applicators for crushing and mixing medication for a critically ill patient on tube feeding.[10] While immunocompetent hosts can fight off and resist the invasion of Mucorales once ingestion of the spores into the gastrointestinal tract, and patients who are immunocompromised will not be able to resist mucosal invasion because of poor defense mechanisms and further are liable to develop severe other infection. The etiopathogenesis of mucormycosis infection in known diabetic patients remains to be elusive at the present time. The proposed mechanism of gastric mucormycosis infection is due to impaired chemotaxis, phagocytic dysfunction, and defective intracellular destruction of Mucorales in the presence of an acidic environment of the stomach.[11] The uncontrolled diabetes may result in a slightly acidic environment in the body and predispose patients to mucormycosis and the propensity to form fistula is due to the gastric perforation and spread of Mucorales infection from gastric ulcers to the adjacent organs.
The predisposing risk factors to develop gastric mucormycosis could be prolonged uncontrolled DM, solid organ or stem cell transplantation, underlying hematologic malignancy, major trauma, the utility of steroids, disseminated chronic infections, and iron overload states. The gastric body is more prone to be affected location of gastric mucormycosis. The invasion of blood vessels may result in serious life-threatening gastrointestinal hemorrhage and is a poor prognostic factor of disease.[4]
The radiological imaging modalities such as CT scan or MRI of the abdomen initially usually reveal nonspecific findings such as mucosal wall thickening, mass, and reactive lymphadenopathy and prompt additional investigation with endoscopic or surgical biopsy of the lesions. EGD finding of ulcerated patchy mucosal lesions with overlying greenish or greyish exudate is a characteristic feature of gastric mucormycosis; however, a biopsy of lesions is essential to differentiate it from gastric malignancy.[4,8] EGD alone does not help to make a definitive diagnosis and additional testing with biopsy specimens is required. Histopathological and culture testing is the most definite in establishing the diagnosis.
CONCLUSION
Mucormycosis is a lethal opportunistic fungal infection that may result in high mortality in untreated patients. A high index of suspicion and awareness of physicians is required for early diagnosis and management of disease particularly in immunocompromised patients. A typical presentation such as worsening abdominal pain should be investigated with a CECT scan and EGD in those with inconclusive findings on radiological imaging. Biopsy of the suspected mucosal lesions is the diagnostic of gastric mucormycosis and the success of the treatment depends on early diagnosis, control of risk factors and quick antifungal therapy as well as timely surgical resection is prime features to manage the disease.
Acknowledgment
The authors acknowledge the patient as a great source of learning.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Epidemiology and outcome of zygomycosis: A review of 929 reported cases. Clin Infect Dis. 2005;41:634-53.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and clinical manifestations of mucormycosis. Clin Infect Dis. 2012;54(Suppl 1):S23-34.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: Analysis of multicenter prospective antifungal therapy (PATH) alliance registry. Clin Infect Dis. 2009;48:265-73.
- [CrossRef] [PubMed] [Google Scholar]
- Gastric mucormycosis complicated by a gastropleural fistula: A case report and review of the literature. Medicine (Baltimore). 2019;98:e18142.
- [CrossRef] [PubMed] [Google Scholar]
- Isavuconazole treatment for mucormycosis: A single-arm open-label trial and case-control analysis. Lancet Infect Dis. 2016;16:828-37.
- [CrossRef] [Google Scholar]
- Prevalence, clinical and economic burden of mucormycosis-related hospitalizations in the United States: A retrospective study. BMC Infect Dis. 2016;16:730.
- [CrossRef] [PubMed] [Google Scholar]
- Invasive gastrointestinal zygomycosis in a liver transplant recipient: Case report. Transplantation. 2002;73:145-7.
- [CrossRef] [PubMed] [Google Scholar]
- Total gastric necrosis due to mucormycosis: A rare case of gastric perforation. Am J Case Rep. 2018;19:527-33.
- [CrossRef] [PubMed] [Google Scholar]
- Outbreak of gastric mucormycosis associated with the use of wooden tongue depressors in critically ill patients. Intensive Care Med. 2004;30:724-8.
- [CrossRef] [PubMed] [Google Scholar]
- Update on mucormycosis pathogenesis. Curr Opin Infect Dis. 2013;26:508-15.
- [CrossRef] [PubMed] [Google Scholar]






