Translate this page into:
Renal Collision Tumor in Association with Xanthogranulomatous Pyelonephritis
Address for correspondence: Dr. Jennifer Rothschild, Department of Urology, University of Rochester School of Medicine and Dentistry, 601 Elmwood Avenue, Box 648 Rochester, NY 14642, USA. jennifer_rothschild@urmc.rochester.edu
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Collision tumor is a rare condition in which two neoplasms (usually benign and malignant), both growing in the same general area, collide with each other and become intermingled. We present histopathology and imaging correlation of xanthogranulomatous pyelonephritis coexistent with squamous cell carcinoma and osteogenic sarcoma of the kidney.
Keywords
Computed tomography
kidney
MAG3
neoplasms
osteogenic sarcoma
squamous cell carcinoma
xanthogranulomatous pyelonephritis
INTRODUCTION

Collision tumors, in which neoplasms with different histogenesis arise in close proximity and intermingle, are exceedingly uncommon. Primary renal osteogenic sarcoma is also very infrequent, with only 20 cases previously reported.[1] We report the first case of a synchronous, colocalized squamous cell carcinoma (SCC) and osteogenic sarcoma of the kidney in the setting of xanthogranulomatous pyelonephritis (XGP), and describe the radiological and pathological features of this rare entity.
Our patient was a 74-year-old Hispanic female with a long history of recurrent urinary tract infections, flank pain, and staghorn calculi. The patient was first evaluated with ultrasound that revealed an enlarged left kidney with multiple renal stones and presence of multiple cystic areas within the kidney. The exact location and morphology of these cysts could not be discerned appropriately on the ultrasound study. Subsequent computed tomography (CT) with intravenous contrast in nephrographic and excretory phases of evaluation demonstrated a large staghorn calculus in the renal pelvis along with multiple stones in the enlarged left kidney [Figure 1]. The entire renal parenchyma appeared to be replaced with cystic areas, some of which appeared complex with septations and internal debris/nodularity. Some of these cystic areas were thought to be due to hydronephrosis secondary to the obstructing calculi. Excretory phase of the study demonstrated absence of opacification of the collecting system with contrast. CT findings were thought to represent XGP, although malignancy was not entirely excluded. Subsequent mercapto acetyl tri glycine (MAG3) nuclear scan of the kidneys revealed complete absence of blood flow and function in the left kidney [Figure 2], following which a radical left nephrectomy was performed. The patient's surgery and postoperative course were uneventful.
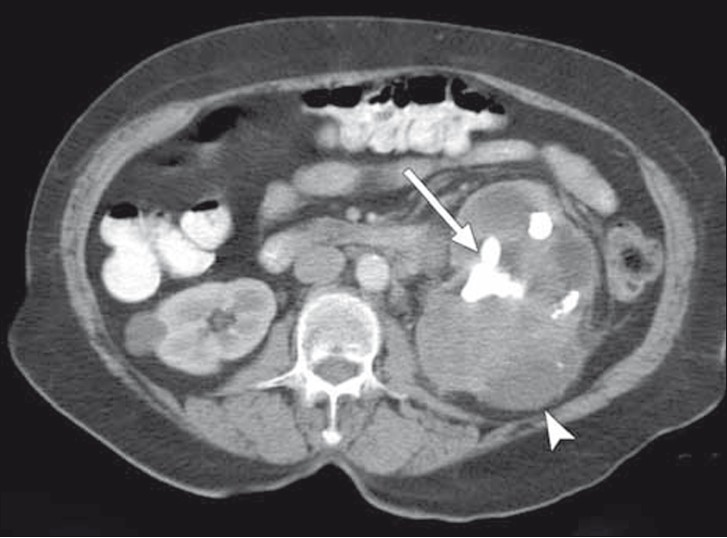
- A 74-year-old female with collision tumor of the kidney. Nephrographic phase image of the contrast enhanced axial CT demonstrates an enlarged left kidney with a staghorn calculus (arrow) and multiple other calcifications/calculi. Renal parenchyma is replaced with multiple cystic low attenuating lesions (arrowhead). There was no local lymphadenopathy.
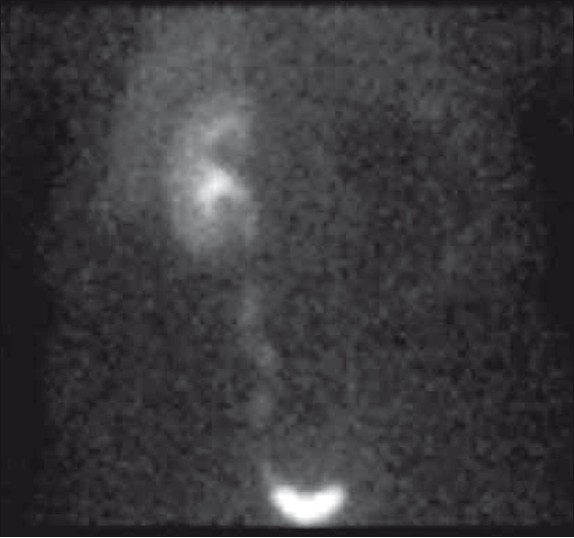
- Image from a mercapto acetyl tri glycine (MAG3) scan shows absence of blood flow and function in the left kidney. The right kidney is normal.
RADIOLOGICAL FEATURES
XGP is an uncommon form of chronic pyelonephritis that occurs as a result of chronic obstruction and subsequent infection. Almost all cases of XGP (90%) are associated with renal calculi.[23] CT is the imaging modality of choice for XGP as it provides an accurate estimate of the (extrarenal) extent of the disease, thus helping in surgical planning. Diagnosis is usually made by the presence of an enlarged non-functioning kidney with staghorn calculus, caliceal dilatation, low attenuation areas replacing the renal parenchyma secondary to inflammatory infiltrate, and perinephric stranding.[2] All the above mentioned features were present on the CT images of our patient, and therefore XGP was the leading consideration.
SCC of the renal pelvis is the second most common malignancy after adenocarcinoma, and usually is secondary to chronic irritation caused by long standing renal calculi. The kidney is usually non-functional due to chronic obstruction. SCC presents as a renal pelvic infiltrative lesion without evidence of a distinct mass.[4] SCC cannot be distinguished from XGP on imaging due to common radiological features including an enlarged non-functional kidney with calculus and renal pelvic infiltrative process. Histopathology is useful in making this distinction.
Primary renal sarcomas are extremely rare (about 1% of renal mesenchymal tumors).[4] Extraosseous primary osteogenic sarcoma of the kidney is a very unusual tumor, and must be differentiated from osteoid formation in a sarcomatoid tumor of the kidney. Imaging features of a renal osteogenic sarcoma have been rarely described in literature. In one of the case reports, it presented as a partially calcified mass in the renal parenchyma.[5]
Coexistence of renal osteogenic sarcoma along with XGP and SCC in our patient made it extremely difficult to distinctly identify each entity separately, and the final diagnosis was based on the histopathological evaluation of the kidney.
PATHOLOGICAL FEATURES
Grossly, the entire kidney was occupied by a friable, yellow-white lobulated mass that was 12.0 cm in its greatest dimension and extended through the renal parenchyma and the renal pelvis [Figure 3]. The periphery and upper pole of the kidney contained multiple cystic spaces that may have been true renal cysts versus dilated renal calyces. Contained within the mass were a 7.0 cm staghorn calculus, numerous smaller calculi, and a separate 1.9 cm granular white-pink nodule.
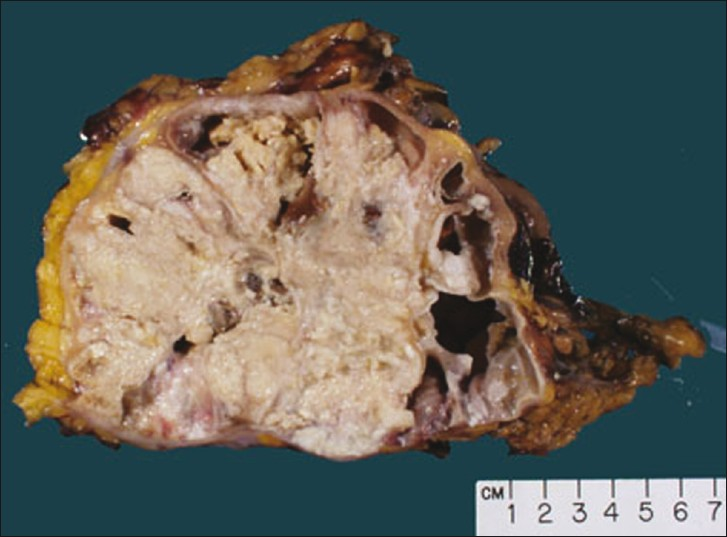
- Gross photograph of a sagittal section of the kidney demonstrates a friable, yellow-white mass that extends through the renal parenchyma and pelvis. The periphery and upper pole of the kidney contains multiple cystic spaces.
On histopathological examination, the larger mass consisted of a moderately differentiated keratinizing SCC with extensive necrosis and angiolymphatic invasion. In areas this tumor appeared to collide with, but not arise from a separate tumor [Figure 4a]. These tumor cells were spindle to polyhedral in shape and arranged in cords and interweaving fascicles. They displayed amphophilic cytoplasm and large pleomorphic nuclei with vesicular chromatin and prominent nucleoli. Mitotic figures were easily identifiable. Ramifying throughout the cords of tumor cells were wisps of pale pink, homogenous unmineralized osteoid matrix, consistent with a diagnosis of high-grade osteosarcoma [Figure 4b]. There was associated abscess formation and necrosis. Sarcomatous cells stained positively with CD10 and negatively with pancytokeratin, epithelial membrane antigen, smooth muscle actin, S-100 protein and Alk-1 by immunohistochemistry. The positive staining with CD10 and lack of staining with pancytokeratin are consistent with a diagnosis of a sarcoma, as opposed to a neoplasm of epithelial origin. Although the vast majority of the specimen comprised of SCC, evidence of XGP was also seen microscopically [Figure 4c]. Six perinephric lymph nodes were negative for malignancy.
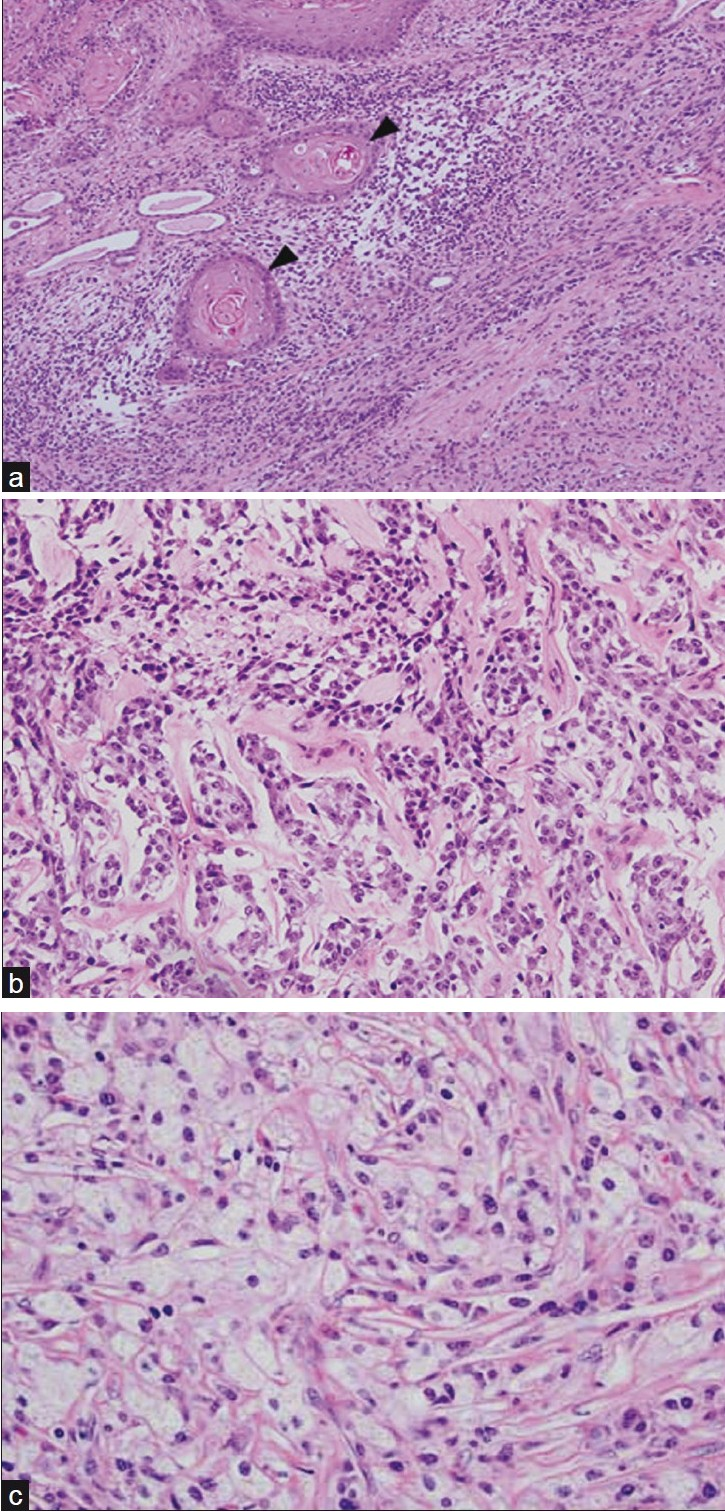
- (a) Photomicrograph (10×, hematoxylin and eosin) illustrates several squamous pearls (arrowheads) of a moderately differentiated, keratinizing squamous cell carcinoma “colliding” with spindled osteosarcoma cells. (b) Photomicrograph (20×, hematoxylin and eosin) shows spindled to polyhedral tumor cells arranged in cords and interweaving fascicles. Ramifying throughout the cords of tumor cells are wisps of pale pink, homogenous chondro-osseous matrix. (c) Photomicrograph (40×, hematoxylin and eosin) demonstrates characteristic lipid-laden foamy macrophages or “xanthoma cells”, the hallmark of xanthogranulomatous pyelonephritis.
XGP has rarely been associated with the development of malignancies; namely, renal cell carcinoma.[67] Chronic irritation with subsequent or concomitant infection is known to cause SCC. SCC in the urinary tract usually results secondary to long standing urinary calculi or pyelonephritis, as a result of a series of reactive changes including metaplasia, dysplasias, and finally neoplasia.[8] This process explains the occurrence of SCC in the urotheium, which has no squamous cell structures.
To our knowledge, there has been no reported association of XGP with renal SCC, although squamous metaplasia has been described in this setting.[9] The immunohistochemical staining pattern of the osteosarcoma (pancytokeratin negative) makes it less likely to have arisen from sarcomatoid differentiation of the SCC, and seemingly more likely that this second malignancy arose de novo.
In conclusion, it is important to consider coexistence of malignancy in patients with radiographic features of XGP.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2011/1/9/75263
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Primary osteosarcoma of the kidney with retroperitoneal hemorrhage: Case report and review of the literature. Tumori. 2007;93:213-6.
- [Google Scholar]
- Pyelonephritis: Radiologic-pathologic review. Radiographics. 2008;28:255-77. quiz 327-8
- [Google Scholar]
- Xanthogranulomatous pyelonephritis: Clinical experience with 41 cases. Urology. 2008;71:178-80.
- [Google Scholar]
- From the archives of the AFIP.Infiltrative renal lesions: Radiologic-pathologic correlation. Radiographics. 2000;20:215-43. Armed Forces Institute of Pathology
- [Google Scholar]
- Xanthogranulomatous pyelonephritis associated with renal cell carcinoma: Report on two cases and review of the literature. Eur Urol. 1990;18:74-6.
- [Google Scholar]
- Xanthogranulomatous pyelonephritis and renal malignancy: Unusual fellows in the renal bed. BJU Int. 2000;86:558-9.
- [Google Scholar]
- Primary adenosquamous and squamous cell carcinoma of the upper urinary tract: Report on three cases. Urol Int. 2000;64:47-51.
- [Google Scholar]
- Rare coexistence of keratinizing squamous metaplasia with xanthogranulomatous pyelonephritis: Report of a case with the role of immunocytochemistry in the differential diagnosis. Acta Cytol. 2007;51:92-4.
- [Google Scholar]






