Translate this page into:
Radiography of Cardiac Conduction Devices: A Pictorial Review of Pacemakers and Implantable Cardioverter Defibrillators
Address for correspondence: MS. Stephanie C Torres-Ayala, Department of Diagnostic Radiology, School of Medicine, University of Puerto Rico, HC 05 BOX 50873, Mayaguez - 00680, Puerto Rico, United States. E-mail: Stephanie.torres19@upr.edu
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Cardiac conduction devices (CCDs) depend on correct anatomic positioning to function properly. Chest radiography is the preferred imaging modality to evaluate CCD's anatomic location, lead wire integrity, and help in identifying several complications. In this pictorial review, our goal is to familiarize radiologists with CCD implantation techniques, appropriate positioning of the device, common causes of malfunction, methods to improve report accuracy, and assure maximal therapeutic benefit.
Keywords
Cardiac conduction device
implantable cardioverter defibrillator
pacemaker
radiography
INTRODUCTION

Cardiac conduction devices (CCDs) were created in the 1950s to prevent asystole. The Swedish implanted the first permanent pacemaker, increasing the average life expectancy to over 40 years.[1] By 2002, 3 million people worldwide had pacemakers. Each year 600,000 devices are implanted.[2] Numbers continue to increase, with 1 million people in the United States with a permanent CCD in the year 2011.[3]
CCDs have three main parts: A pulse generator, an electrode, and lead wires. The electrode line is proximally introduced into the generator via a terminal connector pin, which should be engaged. The distal end of the electrode is composed of lead wires with different tips for attachment to the heart [Figure 1]. Helical lead tips are screwed onto the myocardium, this is known as active fixation. The tips can have other shapes that anchor onto heart trabeculae by development of fibrosis, this is known as passive fixation.[4]
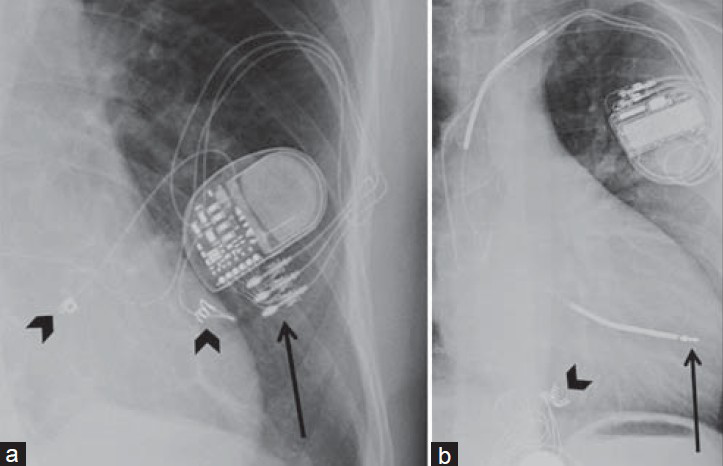
- Lead components and fixation mechanisms. X-ray (a) PA view shows helical epicardial lead tips actively fixed and sutured to the epimyocardium (black arrowheads). Image also shows a Pulse generator (G) with lead terminal connector pins engaged on the generator connector block (black arrow). (b) LAT view shows ICD and pacemaker combination with ICD lead tip passively fixed on the heart trabeculae (black arrow). Note the epicardial pacemaker with the helical lead tips actively fixed (black arrowhead).
Chest radiography is used for evaluation of lead location and integrity after device implantation and for diagnosis of complications and malfunction. At least two well-penetrated chest radiographic views are required for thorough examination.[3] A posterio-anterior (PA) radiograph provides a two-dimensional view for physical integrity evaluation of the lead. To evaluate lead tip location, both PA and lateral (LAT) chest views are necessary. PA views provide a presumed lead location; therefore, a misplaced lead cannot be ruled out without a LAT view, which provides a third chest dimension.[5] LAT views, important for ventricular lead evaluation, allow distinction to be made between an anteriorly coursed right ventricle (RV) lead and a posteriorly coursed left ventricle lead.[3] It is important to be familiar with conventional chest PA and LAT radiographic cardiac anatomy [Figure 2] to distinguish correct lead locations.
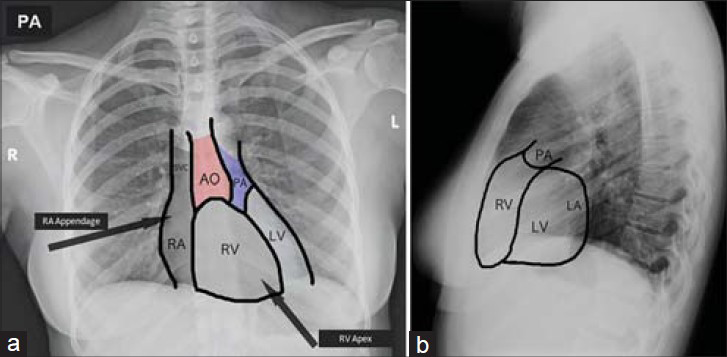
- Normal chest PA and LAT radiographic views. Chest X-ray (a) posterio-anterior (PA) view and (b) lateral (LAT) view of normal cardiac anatomy shows the aorta (AO), pulmonary artery (PA), right ventricle (RV), left ventricle (LV), and right atrium (RA).
PLACEMENT TECHNIQUES
Different anatomical approaches and techniques are used for CCD implantation. The percutaneous transvenous technique is used almost exclusively as the first-line intracardiac approach for lead placement. Fluoroscopically guided leads are inserted via the subclavian vein (preferred), cephalic, or jugular vein for RV or atrial access. The generator is placed in a subcutaneous pocket in the left infraclavicular fossa, right infraclavicular fossa, axilla, submammary, or under the pectoralis major muscle.[6] A video-assisted thoracoscopy is an alternative approach for cases where endocardial lead placement is not possible. Currently, sternotomy for direct placement of leads on the epicardium is rarely necessary.
TYPES OF CARDIAC CONDUCTION DEVICES
Pacemaker
The sinoatrial (SA) node, located at the junction of the superior vena cava and the right atrium, is the heart's natural pacemaker which generates electrical impulses to stimulate myocardial contraction. When disrupted by myocardial infarction or disease, cardiac electrical activity is altered causing dysrhythmia or bradycardia. An artificial pacemaker is then programmed and implanted to provide electrical impulses and maintain a stable heart rate and cardiac output.
Pacemakers vary according to lead amount, paced heart chambers, and placement approach. The simplest, a single-chamber pacemaker, contains one lead. Through the transvenous approach, the lead can pace the right atrium in patients with sinus node disease or the RV in patients with atrial fibrillation with slow ventricular response.[6] In right atrial pacing, PA chest radiograph shows an inferiorly directed lead into the right atrium, which then curves anteriorly, known as the “J loop,” with the tip at the right atrial appendage. When pacing the RV, the lead is directed inferiorly through the right atrium, curves through the tricuspid valve, and ends in an anterior–inferior position at the RV apex, and is seen left of the spine on PA X-ray. The LAT view showing an anteriorly oriented lead confirms the lead tip is in the RV. Note that in some cases, the right ventricular outflow tract (RVOT) may be used as an alternate site for pacing if the RV apex is not electrophysiologically suitable. Dual-chamber pacemakers contain two lead wires allowing pacing of the right atrium and RV simultaneously [Figure 3]. These CCDs are used to coordinate electrical signals and contraction between the atrium and ventricle in heart block patients.[3] RV leads are commonly misplaced when the lead takes a “snake-like” course and goes through the coronary sinus, ending in a posterior or lateral epicardial vein instead of the RV apex [Figure 4]. Misplacement of either lead can cause device malfunction and recurrence of symptom [Figures 5 and 6]. Radiography can demonstrate the inadvertent placement of leads in unintended sites, including: (1) Through a patent foramen ovale or atrial septal defect and then coursing through the mitral valve into the left ventricle; (2) leads that inadvertently puncture the subclavian artery and then course through the aortic valve into the left ventricle; (3) leads that go through a ventricular septal defect or perforate the interventricular septum; or (4) leads that go beyond the heart by perforating the cardiac chamber walls [Figure 7]. All of the above have distinct radiological features.
- Dual-chamber pacemaker. 89-year-old male with a dual-chamber pacemaker placed via the left subclavian vein with the generator in the subcutaneous left infraclavicular fossa. Chest X-ray (a) PA view and (b) LAT view show the right atrial lead is inferiorly directed and takes a “J loop” curve with tip pacing the right atrial appendage (white arrow). Right ventricle lead directed inferiorly through the right atrium and tricuspid valve, then courses anteriorly and inferiorly with the tip at the right ventricular apex (black arrowhead). In single-chamber pacemakers, only the right atrial or ventricular lead is present.
- Single-chamber pacemaker with abnormal position of the lead. 73-year-old male with a single-chamber pacemaker placed using the subclavian transvenous approach with an abnormally positioned lead. Chest X-ray (a) PA view shows the lead is directed inferiorly through the right atrium taking a serpentine course (black arrow) through the coronary sinus with lead tip misplaced in an epicardial vein (black arrowhead) instead of the right ventricular apex (white arrow). Lead tip is passively fixed and pulse generator is placed in a subcutaneous pocket in the infraclavicular fossa. (b) Lateral view confirms lead misplacement with lead curving in a loop and then coursing anteriorly (black arrow) instead of descending and coursing anteriorly as would be seen in with a correctly placed right ventricular lead.
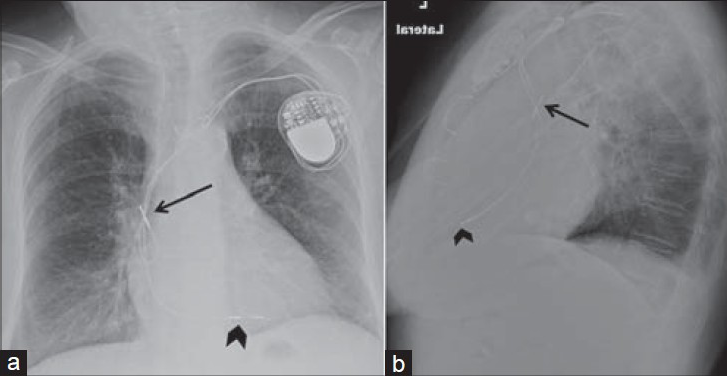
- Unsatisfactory right atrium lead position. 80-year-old female with a dual-chamber pacemaker placed via the subclavian vein approach. Chest X-ray (a) PA view shows the right atrial lead curls upward and crosses itself, with the tip unsatisfactorily positioned in the superior vena cava (black arrow) instead of taking a “J loop” course toward the right atrial appendage. Right ventricular lead correctly placed (black arrowhead), directed inferiorly through the right atrium and tricuspid valve, and courses anteriorly and inferiorly to the left of the spine, with the tip at the right ventricular apex. (b) LAT view shows right ventricular lead courses anteriorly ending at the right ventricular apex (black arrowhead). Right atrial lead curls upward with the tip misplaced in the superior vena cava (black arrow).
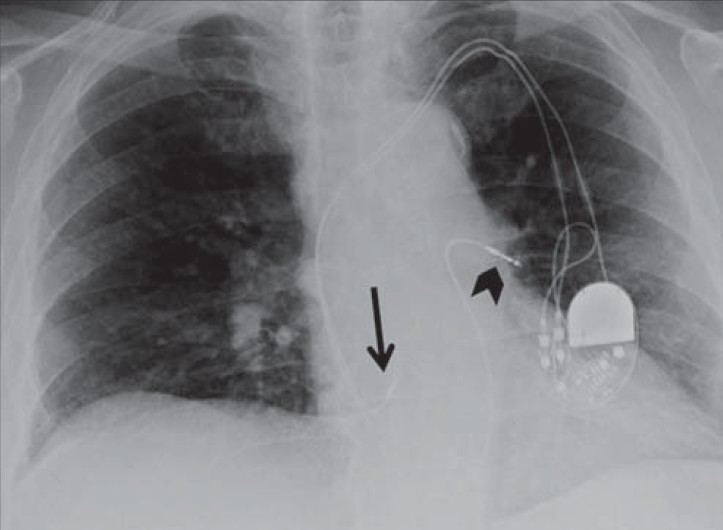
- Right ventricle lead in left main pulmonary artery. 73-year-old male with a dual-chamber pacemaker. Chest X-ray PA view shows the right ventricle lead is misplaced (black arrowhead), taking a “swan ganz”-like course and ending in the left main pulmonary artery instead of the right ventricle apex. Right atrium lead is correctly placed at the right atrial appendage (black arrow).
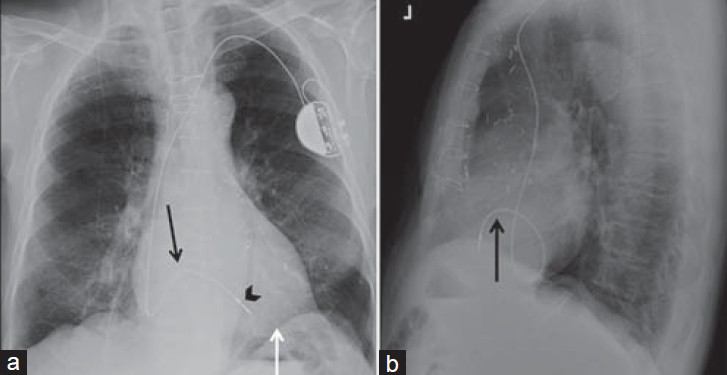
Biventricular pacemakers, also called cardiac resynchronization therapy (CRT) devices, treat refractory heart failure associated with ventricular dyssynchrony.[3] These devices have one lead pacing each ventricle; some have a right atrial lead. In the transvenous approach, the left ventricular lead enters the right atrium through the coronary sinus orifice, taking an endovenous “snake-like” course until the tip reaches a left ventricular wall epicardial vein [Figure 8]. This is confirmed using a lateral view showing a posteriorly oriented lead. Biventricular pacing can also be achieved using epicardial leads placed through a thoracotomy [Figure 9]. Even though more invasive, epicardial pacing is used in pediatric populations due to small heart size or congenital anatomical defects restricting access to the desired area.[6] It is also an alternative for patients with aberrant cardiovascular anatomy or unsuitable transvenous access.
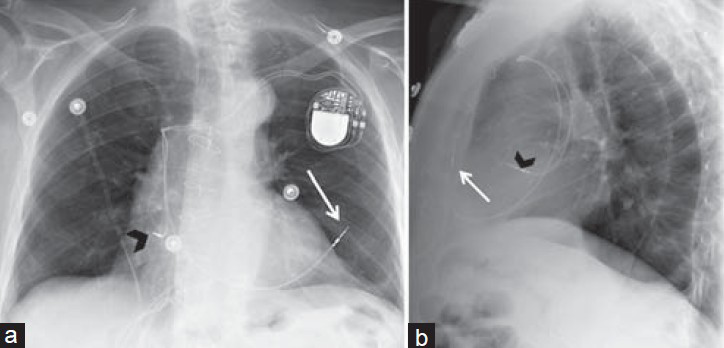
- Inadvertent lead placement. Chest X-ray (a) PA and (b) LAT views of a dual-chamber pacemaker show where the right ventricular lead has inadvertently perforated the right ventricular wall, causing the lead tip to lie outside the cardiac chambers, within the anterior thoracic cavity (white arrow) and right atrial lead with the tip correctly placed in the right atrial appendage (black arrowhead).
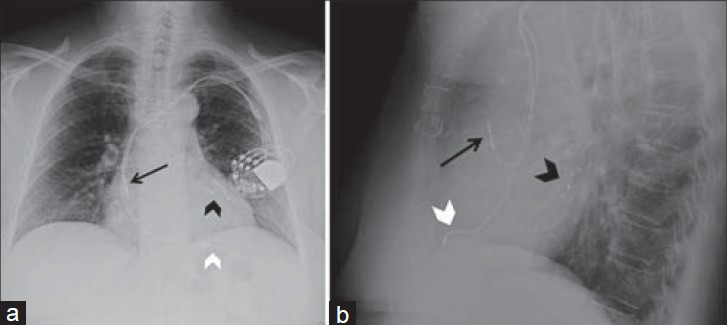
- Biventricular pacemaker. 71-year-old female with a biventricular pacemaker with leads placed via transvenous approach with the generator in a subcutaneous pocket in the infraclavicular fossa. Chest X-ray (a) PA view shows the right atrial lead correctly placed at the right atrial appendage (black arrow) with “J loop” appearance. Right ventricular lead correctly placed at the apex (white arrowhead), seen left of the spine. Left ventricle lead, directed inferiorly through the right atrium, takes a serpentine course via the coronary sinus (black arrowhead) with the tip in a posterolateral epicardial vein of the left ventricular wall. (b) LAT view shows anteriorly and superiorly oriented right atrial lead (black arrow) and an anteriorly and inferiorly directed right ventricular lead (white arrowhead). Left ventricular lead correct position is confirmed with the posterior course of lead (black arrowhead) on LAT view.
- Biventricular pacemaker with epicardial leads. 66-year-old female with a biventricular pacemaker with epicardial leads placed using a transthoracic approach with the generator lodged in the subcutaneous tissues of the left anterior chest wall. Chest X-ray (a) PA view and (b) LAT view show epicardial leads are sutured to the epimyocardium on the right ventricle (black arrow) and left ventricle (black arrowhead). A right atrial lead is also present (white arrow).
In efforts to eliminate pacemaker wires, leadless cardiac pacemakers (LCPs) were recently introduced. LCPs are similar to an AAA battery in size and shape. Device implantation involves a minimally invasive procedure where the device is delivered via a steerable catheter through the femoral vein into the RV where it is attached.[7] As of the date of completion of this manuscript, LCPs have not yet received US Food and Drug Administration (FDA) approval.
Implantable cardioverter defibrillator
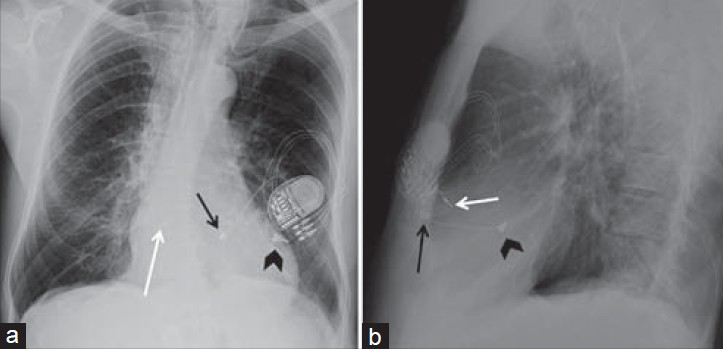
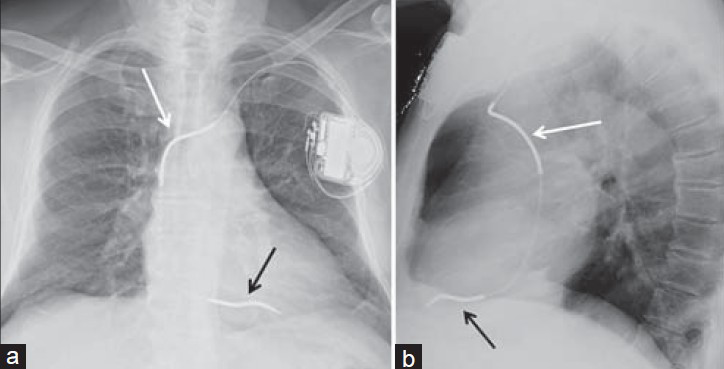
Implantable cardioverter defibrillator (ICD) is an electronic device with a defibrillator unit capable of generating and delivering an electrical impulse and shock to abort dysrhythmias and return the heart to sinus rhythm. ICDs can be either transvenous (T-ICD) or subcutaneous (S-ICD). T-ICDs contain a lead with either one or two metal shock coils placed at the brachiocephalic vein/superior vena cava junction and the RV [Figures 10–12]. T-ICDs are often combined with biventricular pacemakers to provide CRT to high-risk heart failure patients [Figure 13].[3] Combined biventricular devices have three leads; pacemaker leads are placed in the right atrial appendage and left ventricle and the ICD lead in the RV. T-ICDs can also be combined with single-lead pacemakers or epicardial leads [Figures 14–16]. An S-ICD is a novel technology that does not require transvenous lead insertion for defibrillation. Instead, a pulse generator placed at the left lateral chest delivers a transthoracic shock via a left parasternal subcutaneous lead containing sensing electrodes and shock coils [Figure 17]. These devices can be implanted in patients requiring an ICD as long as pacing for tachycardia or bradycardia is not required.[8]
- Correct ICD position. 73-year-old female with an ICD, chest X-ray (a) PA view shows proximal shock coil located at the left brachiocephalic– superior vena cava junction (white arrow), while distant shock coil is at the right ventricular apex (black arrow). (b) Lateral view shows proximal shock coil (white arrow), and since the lead courses anteriorly and inferiorly, distal shock coils confirmed at the right ventricle apex (black arrow).
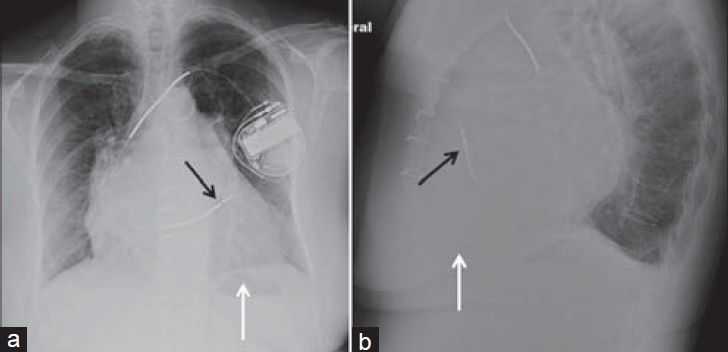
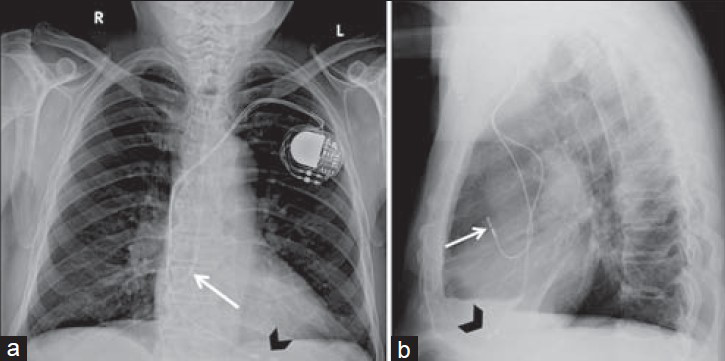
- Right ventricular outflow tract (RVOT) ICD position. 69-year-old female with ICD chest X-ray (a) PA and (b) LAT views show distal shock coil located at the right ventricular outflow tract (black arrow) instead of the right ventricular apex (white arrow). Lateral view confirms that the lead, by taking an anterior and superior course, is placed in the RVOT (black arrow) and not in the right ventricle apex (white arrow).
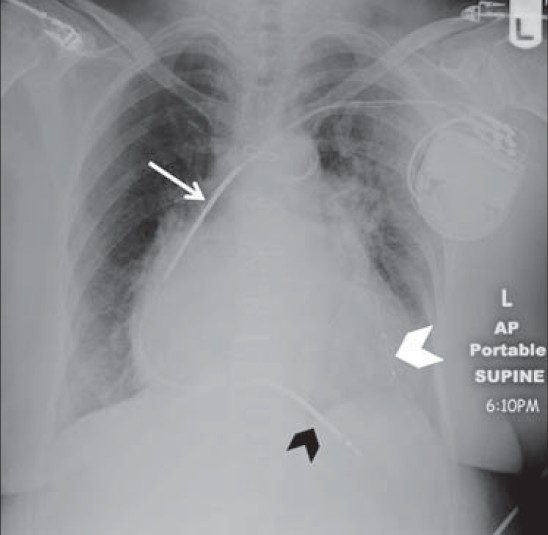
- Corrected ICD in 69-year-old female seen in Figure 11. Chest X-ray AP view shows distal shock coil originally placed in the right ventricular outflow tract, but was later repositioned to achieve better defibrillation threshold. Shock coils are correctly placed at the superior vena cava (white arrow) and the right ventricle apex (black arrowhead). A left ventricle lead via the coronary sinus has been placed in the interval (white arrowhead).
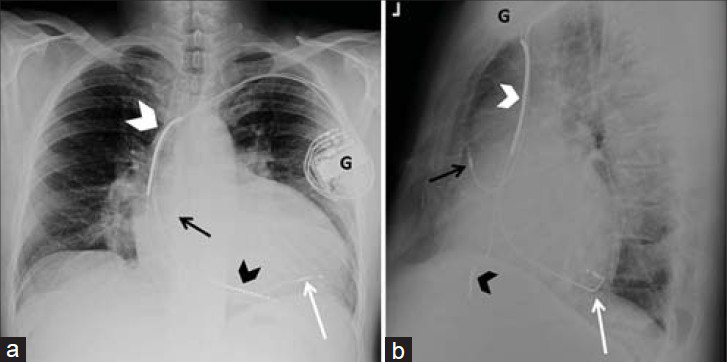
- ICD and CRT combination. 52-year-old male with an implantable cardioverter defibrillator combined with cardiac resynchronization therapy. Chest X-ray (a) PA and (b) LAT views show the leads are placed via the subclavian vein with the generator (G) in a subcutaneous pocket in the infraclavicular fossa. The image shows ICD lead with shock coils at the brachiocephalic–superior vena cava junction (white arrowhead) and the right ventricle (black arrowhead). Pacemaker leads are correctly placed in the right atrial appendage (black arrow) and left ventricle (white arrow), the latter through the coronary sinus and the epicardiac vein.
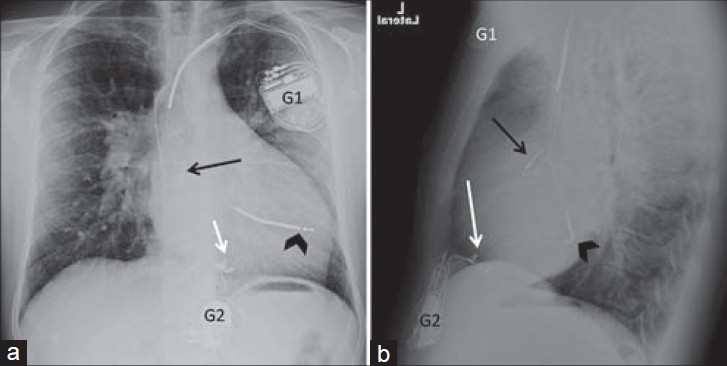
- Biventricular pacing with ICD and two generators. 35-year-old male with complex congenital heart disease. Chest X-ray (a) PA and (b) LAT views show an ICD (black arrowhead) in the left ventricle through an atrial baffle and a right atrial lead (black arrow), both via the left subclavian venous approach with generator (G1) in the left infraclavicular fossa. A second generator in the epigastrium (G2) has epicardal leads (white arrow) to the right ventricle through the subxiphoid approach.
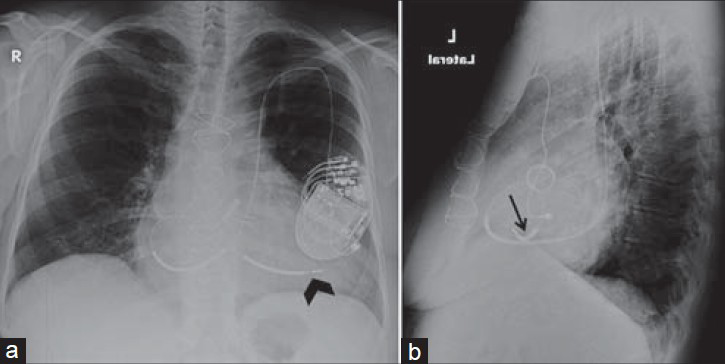
- ICD via left subclavian vein and epicardial lead. 44-year-old female with an ICD. (a) Chest X-ray PA view shows an ICD inserted via the left superior vena cava directed to the right ventricle (black arrowhead) through the coronary sinus and the right atrium. (b) LAT view shows a transthoracic epicardial lead to the left ventricle (black arrow).
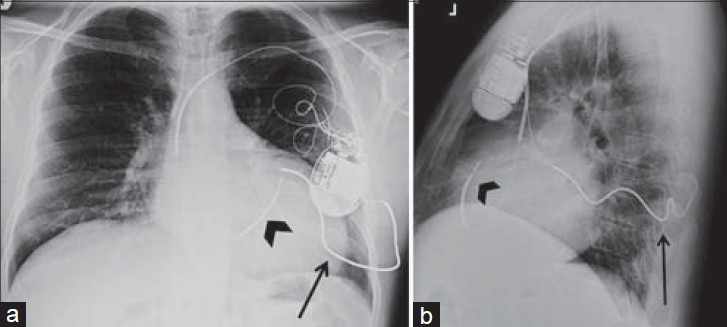
- Subcutaneous array and unsatisfactory ICD position. 51-year-old male with an ICD. Chest X-ray (a) PA and (b) LAT views show the ICD with distal shock coil in the right ventricle outflow tract (black arrowhead). This patient also has a subcutaneous array in the left chest wall (black arrow), which incorporates more of the ventricular wall into the shock focus, improving the defibrillation threshold.
![Subcutaneous ICD. 48-year-old female with a subcutaneous ICD. Chest X-ray (a) PA and (b) LAT views of an S-ICD[8] show the pulse generator (G) is implanted close to the left mid-axillary line. The subcutaneous lead, containing sensing electrodes and shock coils, is placed on average 1.5 cm to the left of the mid-sternal line (white arrow). Images courtesy of Juan C. Sotomonte, MD.](/content/12/2014/4/1/img/JCIS-4-74-g018.png)
- Subcutaneous ICD. 48-year-old female with a subcutaneous ICD. Chest X-ray (a) PA and (b) LAT views of an S-ICD[8] show the pulse generator (G) is implanted close to the left mid-axillary line. The subcutaneous lead, containing sensing electrodes and shock coils, is placed on average 1.5 cm to the left of the mid-sternal line (white arrow). Images courtesy of Juan C. Sotomonte, MD.
MALFUNCTIONING CCD
CCD may malfunction due to physical damage to the lead such as kinked, pinched, or fractured leads [Figures 18–20]. A kinked lead results from bending of the lead during implantation, causing impaired conduction. Conduction is also impaired when leads are pinched by anatomical structures, commonly seen in subclavian crush syndrome where the wire is hindered proximal to the generator before it enters the vein through the clavicle.[4] Subclavian crush can also cause lead fracture: Incomplete fracture, seen as lead thinning, or complete fracture, seen as lead discontinuation with distal wire migration.[4] To be able to detect subtle damages, radiologists must cautiously evaluate the radiograph and follow the leads from the generator origin to the tip on the heart.
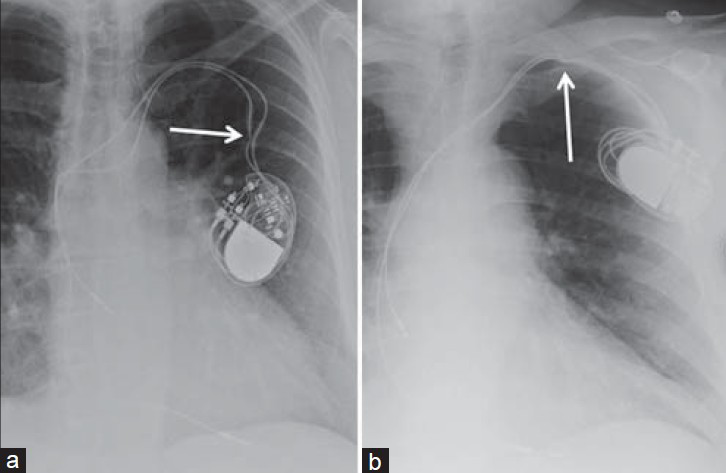
- Malfunctioning leads. (a) 56-year-old female with a dual-chamber malfunctioning pacemaker. Chest X-ray PA view shows the pacemaker malfunction is due to a kinked lead (white arrow). (b) 86-year-old male with a malfunctioning dual-chamber pacemaker. Chest X-ray AP view shows malfunction is due to pinching of the lead (white arrow) before entering the subclavian vein by the clavicle.
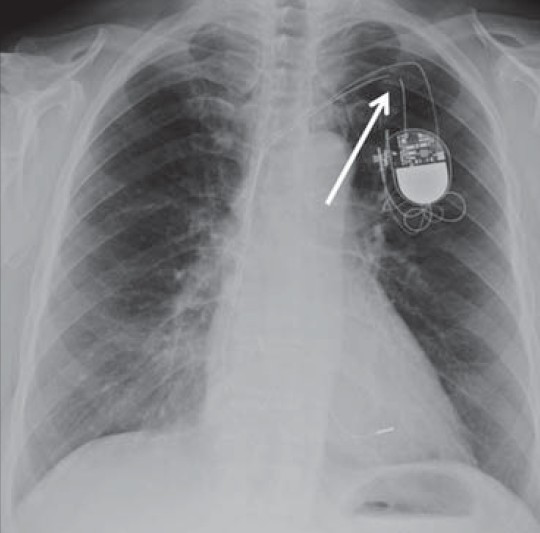
- Complete lead fracture. PA chest X-ray of a malfunctioning pacemaker due to lead complete fracture shows discontinuation of the lead wire (white arrow) proximal to the generator with wide gaps among the lead segments representing dislodgement and migration.
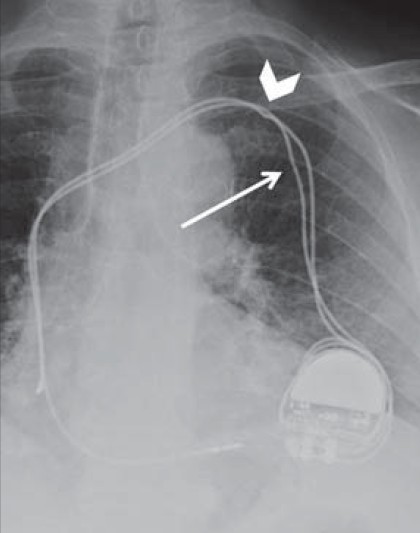
- Pinched lead and “incomplete” lead fracture. 84-year-old female with a malfunctioning dual-chamber pacemaker. Chest X-ray PA view shows pacemaker malfunction is due to pinching of lead wire by the clavicle (white arrowhead). The relative thinning of caliber of the other lead (white arrow) simulates an incomplete fracture, but is in fact caused by the tie-down, which affixes the lead to the chest wall.
Malfunctioning CCD can also occur due to patient device manipulation. Twiddler syndrome occurs when patients rotate the CCD generator and reel the wire [Figure 21]. Leads dislodge from their position and end around the generator, eliminating therapeutic benefit of the CCD. Twitching of the arm or diaphragm can occur if electrodes contact the brachial plexus or phrenic nerve. More serious complications including ventricular perforation can also occur as the leads are pulled back.
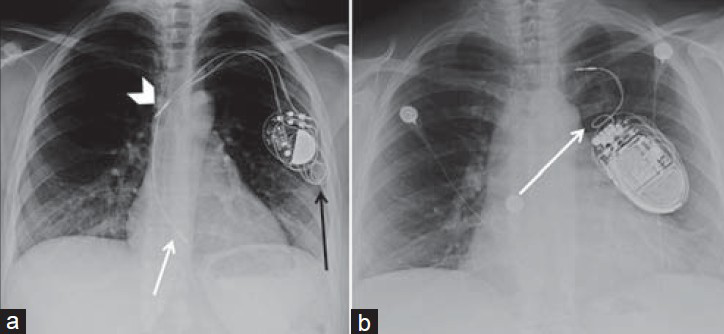
- Twiddler syndrome. (a) 29-year-old female with a dual-chamber pacemaker with twiddler syndrome. Chest X-ray PA view shows the generator has been rotated by the patient causing the lead wires to dislodge and wind around the generator (black arrow). The right atrial lead (white arrowhead) has been removed from the right atrial appendage and lies in the superior vena cava and the right ventricular lead (white arrow) now lies within the right atrium. (b) 54-year-old female with single-chamber pacemaker and a severe case of twiddler syndrome. Chest X-ray PA view shows the lead wire has been completely reeled around the generator (white arrow) causing the patient to receive no cardiac electrical therapy.
RADIOLOGIST CHECKLIST
In order to assure a thorough evaluation and complete report, when interpreting chest radiographs containing CCDs, radiologists should go through the following checklist and make sure all points are addressed:
-
Device type (pacemaker, T-ICD/cardiac resynchronization therapy device, S-ICD, LCP)
-
Connector pin insertion
-
Number and position of leads
-
Lead integrity (fractured/kinked/pinched lead)
-
Complications (e.g. lead dislodgement, pneumothorax, heart chamber perforation).
CONCLUSION
In conclusion, it is crucial for radiologists to be familiar with the radiographic appearance of different types of CCD and correct lead locations to be able to detect complications and abnormal lead position because suboptimal cardiac electrical therapy can be life threatening for the patient.
ACKNOWLEDGMENT
The authors would like to thank Dr. Juan C. Sotomonte (Centro Cardiovascular de Puerto Rico, Ave. Americo Miranda, Esq Centro Medico 1, suite 10, San Juan, PR 00936) for his valuable collaboration.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/74/148269
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- History of electrical therapy for the heart. Eur Heart J Suppl. 2007;9(Suppl I):I3-10.
- [Google Scholar]
- Cardiology patient pages. Cardiac pacemakers from the patient's perspective. Circulation. 2012;105:2136-8.
- [Google Scholar]
- Radiography of cardiac conduction devices: A comprehensive review. Radiographics. 2011;31:1669-82.
- [Google Scholar]
- Radiography of pacemakers and implantable cardioverter defibrillators. AJR Am J Roentgenol. 2012;199:1252-8.
- [Google Scholar]
- Cardiac pacemaker lead placement: Do you need a lateral chest radiograph? Radiography. 2010;16:248-52.
- [Google Scholar]
- Cardiac pacing systems and implantable cardiac defibrillators (ICDs): A radiological perspective of equipment, anatomy and complications. Clin Radiol. 2004;59:699-708.
- [Google Scholar]
- Permanent leadless cardiac pacing: Results of the LEADLESS trial. Circulation. 2014;129:1466-71.
- [Google Scholar]
- The subcutaneous implantable cardioverter-defibrillator: Should patients want one? J Am Coll Cardiol. 2013;61:20-2.
- [Google Scholar]






