Translate this page into:
Primary Sarcoid of the Breast with Incidental Malignancy
Address for correspondence: Dr. Laura M. Isley, Department of Radiology, MSC 323, Medical University of South Carolina, 96 Jonathan Lucas Street, Rm. 210 CSB, Charleston, SC 29425-3230, USA. E-mail: laura.isley1@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Breast sarcoidosis is rare and usually presents in patients with known sarcoid involving other organ systems. In the breast, sarcoidosis may mimic malignancy which must be excluded by core biopsy. We report a very unusual case of primary breast sarcoidosis with incidentally discovered breast carcinoma. The roles of mammography, ultrasound, and MRI in the diagnosis as well as other potential differential diagnosis are discussed.
Keywords
Breast sarcoid
sarcoid
sarcoid and malignancy
INTRODUCTION

Sarcoidosis is a granulomatous disease the exact cause of which is unknown. It is a systemic disease and can affect almost any tissue in the body. Sarcoid is most common between the ages of 20-40 years and is more common in African Americans. Involvement of the breast is rare, but when it occurs, it can be confused with malignancy.[1] The diagnosis of sarcoid does not exclude malignancy. Because malignancy is far more common than sarcoid of the breast, malignancy must always be considered.
CASE REPORT
A 64-year-old African American female was referred to our center with a complaint of new bilateral palpable breast masses and a reported history of diffuse increasing bilateral breast densities on the mammogram taken at another center. She denied skin changes, nipple discharge, or other breast problems. The patient denied family history of breast or ovarian cancer and had no history of trauma to her breasts. Her past medical history included hypothyroidism and hypertension. Physical exam was remarkable for multiple palpable masses throughout both breasts with no overlying skin or nipple changes. No axillary, supraclavicular, or cervical lymphadenopathy was noted bilaterally.
The patient presented to our Breast Imaging Department with printed copies of her digital bilateral mammogram images which were taken at an outside facility on 11/11/2011. Bilateral mediolateral oblique (MLO) and craniocaudal (CC) views were reviewed and demonstrated multiple new irregular spiculated masses within all quadrants of both breasts most prominent in the right upper outer quadrant and the left lower inner quadrant [Figure 1]. There were triangle skin markers denoting the areas of palpable abnormality in the patient and these corresponded to the areas of greatest increase in density. These mammograms were compared to her prior images which were dated 06/01/2010, also from an outside facility [Figure 2]. Additional compression views of the irregular masses in the right lower outer quadrant and the left lower central breast demonstrated that the irregular masses persisted. Further evaluation with bilateral breast ultrasound was recommended to evaluate the new masses.
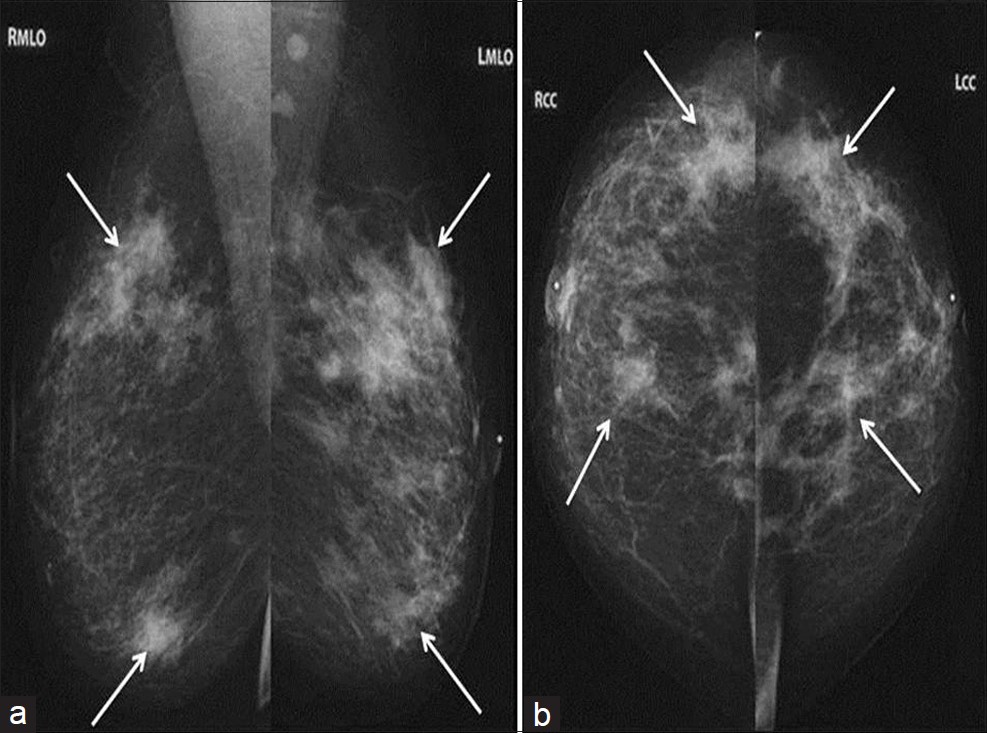
- Bilateral diagnostic mammogram with (a) MLO and (b) CC views demonstrates new spiculated masses throughout both breasts (arrows) which were all initially suspicious for carcinoma.
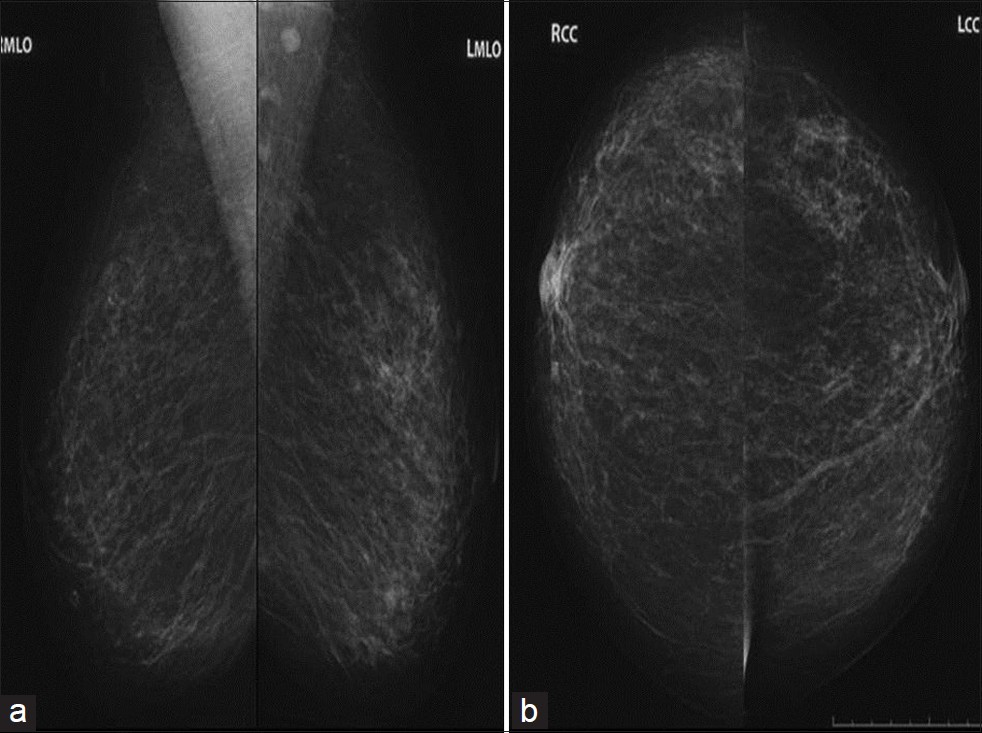
- Comparison mammogram (a) MLO and (b) CC views 1 year prior shows only normal scattered fibroglandular breast tissue with no abnormal masses present.
Ultrasound examination of both breasts was performed utilizing a 12 MHz transducer (Toshiba Alplio MX, Japan). Ultrasound of the bilateral breasts demonstrated multiple hypoechoic, taller than wide irregular masses with angular margins throughout both breasts at nearly every clock position [Figure 3]. These masses correlated with the mammographic findings. Normal axillary lymph nodes were present bilaterally.
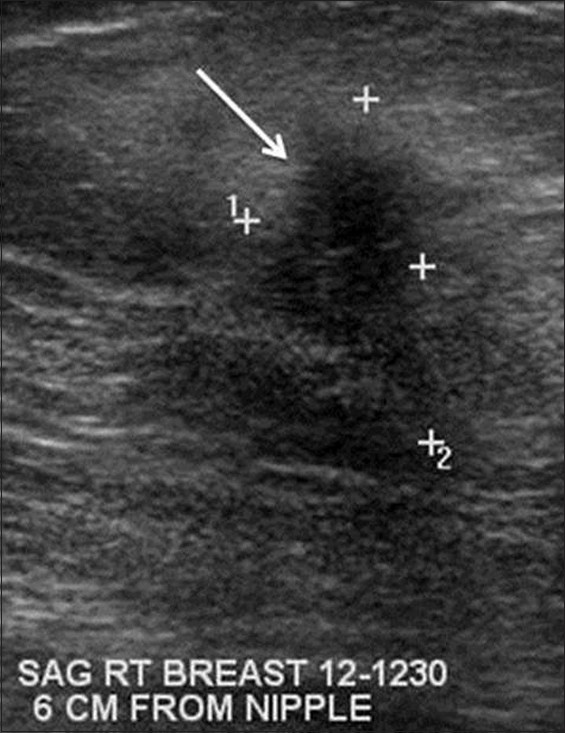
- Gray-scale sonograhic image of the right breast demonstrates an irregular hypoechoic mass with angular margins (arrow). Similar masses were present in the bilateral breasts at nearly every clock position by ultrasound. This mass was biopsy proven to be sarcoidosis.
The mammographic and ultrasound findings were suggestive of malignancy, and the patient subsequently underwent bilateral ultrasound guided core biopsy using a 14 gauge Achieve biopsy device (Cardinal Health, McGraw Park, IL) of the most suspicious appearing masses bilaterally at 6:00 and 11:00 o’clock position in the left breast and at 6:00 and 12:00 o’clock positions in the right breast. Pathology results of the left breast lesion at the 6:00 o’clock position showed invasive lobular carcinoma Nottingham combined histologic Grade II/III moderately differentiated [Figure 4]. Estrogen and progesterone receptors were positive, and HER2 was unamplified. There was an additional finding of granulomatous inflammation consistent with sarcoid. Pathology results of the left breast at the 11:00 o’clock and right breast at 6:00 and 12:00 o’clock positions all showed granulomatous inflammation consistent with sarcoid. Auramine rhodamine and Grocott's methenamine silver stains were negative for acid-fast bacteria and fungus at all four biopsy sites.
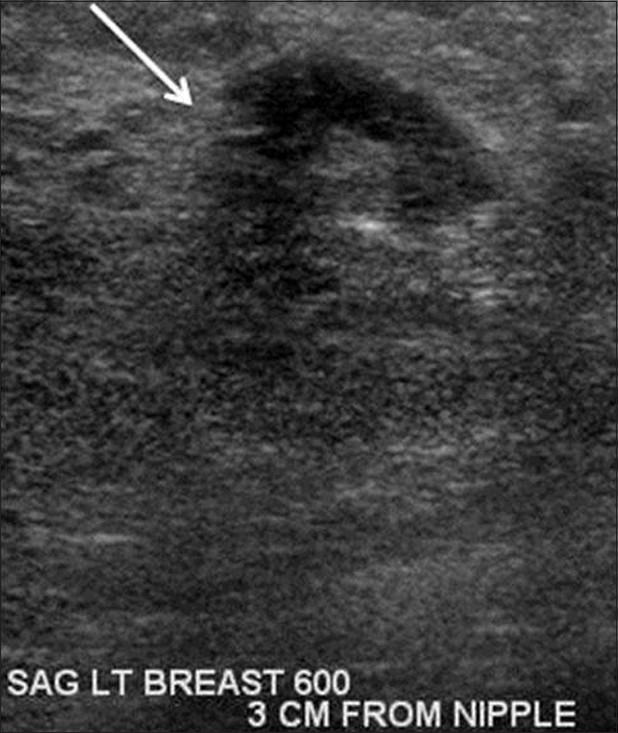
- Gray-scale sonographic image of the left breast at the 6:00 o’clock position at the site of known lobular carcinoma also demonstrates an irregular hypoechoic mass (arrow).
Breast MRI (Magnetom Verio 3T, In vivo Breast Coil, Siemens, Germany) was performed with intravenous contrast to evaluate the extent of disease. The MRI demonstrated patchy areas of non-mass enhancement throughout all quadrants of both breasts [Figure 5a]. The patient's known invasive lobular carcinoma in the left breast at the 6:00 o’clock position was indistinguishable from the known sarcoid lesions [Figure 5b]. No abnormal lymph nodes were identified by MRI.
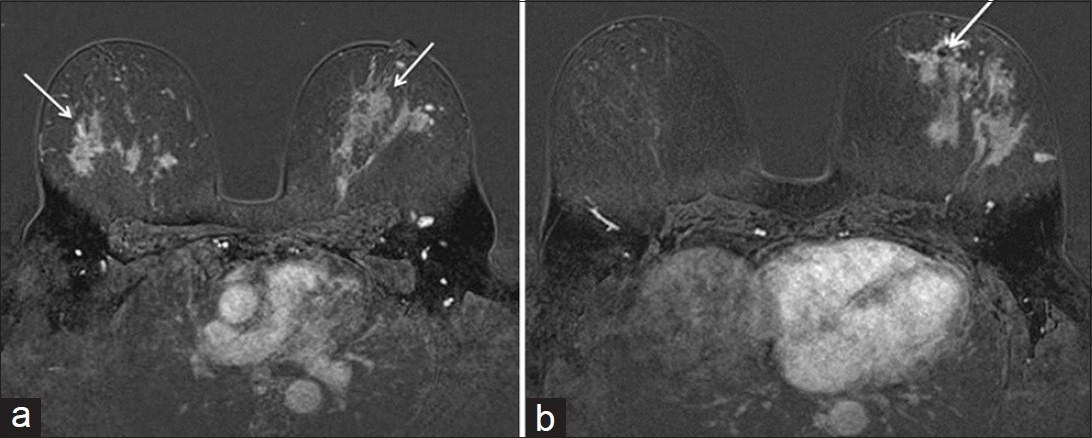
- Axial MRI T1WI post contrast fat saturation subtracted images of the bilateral breasts (a) Breast sarcoidosis presents as multiple patchy bilateral areas of non-mass enhancement (arrows) (b) Known left breast lobular carcinoma at 6:00 o’clock position (arrow) with susceptibility artifact from biopsy clip is indistinguishable from the surrounding sarcoid lesions.
The patient underwent CT (Lightspeed, GE Healthcare, UK) of the chest to evaluate for sequela of sarcoid which revealed upper lobe predominant perihilar ground glass opacities with mild bronchiectasis consistent with sarcoidosis. On CT of the abdomen and pelvis, multiple soft tissue nodules of the omentum, left flank, and gluteal regions were noted which were all subsequently biopsy-proven to be granulomas consistent with sarcoid.
The patient initially desired breast conserving therapy and was treated with left partial mastectomy and sentinel lymph node biopsy. Surgical pathology revealed the presence of a 4.5 cm invasive lobular carcinoma of the left breast with ductal carcinoma in situ nuclear Grade II/III (intermediate) and lobular carcinoma in situ. The margins were involved by invasive lobular carcinoma. The sentinel lymph node biopsy revealed metastatic carcinoma. There was also extensive involvement of the breast tissue by sarcoid granulomas.
Given the margin positivity, extent of the cancer, the complexity of the mammographic, sonographic, and MRI imaging, and the need for treatment with steroids as well as adjuvant chemotherapy, the patient ultimately underwent a completion mastectomy with complete axillary lymph node dissection on the left side. The patient also underwent a right prophylactic mastectomy. Final staging was T2 N2 (5/13 lymph nodes positive). The patient's treatment plan for adjuvant therapy included cytotoxic chemotherapy, post mastectomy radiation, and endocrine therapy.
DISCUSSION
Sarcoidosis primarily involves the lungs, lymph nodes, spleen, liver, skin, eyes, muscles, myocardium, bones, CNS, and salivary glands.[1] Sarcoid involvement of the breasts is rare, and occurs in less than 1% of sarcoidosis patients.[2] It is also very rare for breast involvement to be the primary manifestation of sarcoid. Most cases of breast sarcoid occur in patients who already have a known sarcoid diagnosis elsewhere in the body. In an article by Ojeda et al., 35 cases of sarcoidosis of the breast published between 1921 and 1997 were reviewed. Only 20% (seven cases) had a breast mass as the initial manifestation, and only one patient (3%) had bilateral breast masses.[3]
Breast sarcoidosis most often presents mammographically as an irregular and/or spiculated mass.[4] The most common finding of breast sarcoidosis on ultrasound is an irregular hypoechoic mass.[4] Other findings may include enlarged axillary or intramammary lymph nodes; however, in our case there was no abnormal lymphadenopathy present. These imaging appearances are all suspicious for malignancy and should be evaluated by core biopsy. The differential diagnosis of new irregular spiculated masses include malignancy, fibrosis, radial scar, prior surgery, granulomatous mastitis, diabetic mastopathy, and fungal or mycobacterial infections such as tuberculosis.
Particular note should be made that even MRI is unhelpful for excluding malignancy in cases of breast sarcoidosis. On MRI, sarcoid of the breast often presents as irregular enhancing masses making it difficult to distinguish from carcinoma as in the other imaging modalities. Ishimaru et al., reported a case of breast sarcoid presenting as an irregular mass which demonstrated slow progressive enhancement on serial post contrast MRI imaging.[5] However, other authors have reported breast sarcoid lesions demonstrating rapid enhancement with wash-out phenomenon which is commonly seen in breast carcinoma, making sarcoid lesions very difficult to distinguish from cancer.[1]
In our case, both the bilateral sarcoid lesions and the invasive lobular carcinoma of the left breast at the 6:00 o’clock position demonstrated progressive enhancement on post contrast MRI images. There was also a large area of sarcoidosis surrounding the area of known invasive lobular carcinoma seen at surgical pathology, making the extent of the cancer difficult to determine on any imaging modality.
The possible association of sarcoidosis with a higher rate of malignancy remains controversial and unproven with conflicting results among various studies.[6] Many authors theorize that patients with sarcoidosis are predisposed to malignancy, although others believe that malignancy may lead to sarcodosis.[7] In very rare cases such as ours, both pathologies may coexist in the breast.
CONCLUSION
As in this case, breast sarcoidosis often has imaging features mimicking malignancy on all imaging modalities and carcinoma must always be excluded by core biopsy. One possible management strategy is a baseline mammogram at the time of diagnosis of sarcoid or age 40. All suspicious changes in the breast or axilla require core biopsy to exclude malignancy. Our case was exceptional and presented a particular diagnostic and management challenge with both malignancy and primary sarcoidosis involving the breasts.
Source of Support: Nil
Conflict of Interest: None declared.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2012/2/1/46/99180
REFERENCES
- Sarcoidosis of the breast: Mammographic, ultrasound, and MR findings. J Comput Assist Tomogr. 1997;21:439-41.
- [Google Scholar]
- Sarcoidosis of the breast: Implications for the general surgeon. Am Surg. 2000;66:1144-8.
- [Google Scholar]
- Radiologic evaluation of uncommon inflammatory and reactive breast disorders. Radiographics. 2005;25:411-24.
- [Google Scholar]
- Darier-Roussy sarcoidosis mimicking metastatic breast cancer. Case Rep Oncol. 2009;2:251-4.
- [Google Scholar]






