Translate this page into:
Prenatal and Postnatal Schizencephaly Findings by 2D and 3D Ultrasound: Pictorial Essay
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Schizencephaly is a brain-destructive lesion relating to clefts in the fetal brain that usually communicate with the ventricular system, subarachnoid space, or both. It presents as two types: Type I (closed) and Type II (open). During pregnancy, it may be suspected in the course of ultrasonography, which highlights ventricular dilatation and brain abnormalities. Usually, the diagnosis is confirmed postnatally using trans-fontanel ultrasonography. The etiology of schizencephaly is still unknown, but it may be derived from a primary disorder of brain development or from bilateral middle cerebral artery occlusion. Many causative agents including vascular insult, infections, toxins, and medications have been associated with this malformation. We present a pictorial essay of six cases of schizencephaly that were suspected on prenatal ultrasound scans and confirmed postnatally by trans-fontanel, two-dimensional and three-dimensional ultrasonography.
Keywords
Prenatal diagnosis
postnatal finding
schizencephaly
two-dimensional ultrasonography
three-dimensional ultrasonography
INTRODUCTION

Schizencephaly is a congenital malformation of the brain characterized by clefts that go from the brain hemispheres to the ventricles and the subarachnoid space. The cleft tissue may be in contact with the ventricles (Type I; open lip) or completely closed (Type II; closed lip). This term was first used by Yakovlev and Wadsworth in 1941. They proposed that both types came from primary failure of the cerebral mantle development at the end of the second month of fetal life.[1] These clefts can be located in any part of the hemispheres, but they are more frequent in the perisylvian region.[2]
Schizencephaly is part of a group of neuronal migration disorders, and is caused by an early event that interrupts the migration of neuroblasts from the germinal matrix to the cerebral cortex.[3] Familial cases of schizencephaly have been registered, and are generally associated with heterozygote mutation on the EMX2 gene.[4]
The diagnosis may be suspected prenatally if open or closed clefts are viewed within the cerebral hemispheres by two-dimensional ultrasonography (2DUS).[5] Trans-fontanel three-dimensional ultrasonography (3DUS) performed postnatally enables diagnostic confirmation, and provides greater accuracy in assessing the degree of hydrocephalus.[6]
We present a pictorial essay of six schizencephaly cases that were suspected prenatally and were confirmed postnatally by trans-fontanel ultrasonography.
Ultrasound Imaging
Ultrasonography examinations were conducted on six patients who were attended to at the Prenatal Service of the Mother-Child Unit, University Hospital, Federal University of Maranhão (UFMA), between 2005 and 2010. All of the images were produced by a single examiner (Lívia Teresa Moreira Rios - LTMR) with 10 years of experience in obstetric and neonatal ultrasonography. The device used in all examinations was a Voluson 730 Pro machine (General Electric Medical Systems, Zipf, Austria) equipped with two multifrequency volumetric transducers: RAB4-8 and RIC5-9. The obstetric 2D and 3D scans were done through the mother's abdomen, while the postnatal images were produced through the infant's fontanel. The 3D analyses were done offline by the same examiner (LTRM), using the 4D View software, version 9.0 (General Electric Medical Systems, Zipf, Austria). The sample consisted of five cases of schizencephaly that had been suspected prenatally and were all confirmed postnatally, and one case that had been suspected prenatally to be holoprosencephaly, was confirmed to be schizencephaly postnatally.
Case Series
Figure 1 shows the presence of a large cleft in the cerebral left parietotemporal region, extending to the lateral ventricle (Type II), on prenatal 2DUS. 3DUS in multiplanar mode clearly shows the spatial relationship between the cleft and the ventricular system. 3DUS in the tomographic ultrasound imaging (TUI) mode shows the sequential axis planes of the fetal encephalon, thus showing the lesion level [Figure 2]. Figure 3 exhibits the trans-fontanel 2DUS, which shows the location of the cleft precisely in the cerebral hemisphere and also its measurements [Figure 3]. Figure 4 presents a case of bilateral schizencephaly, in which prenatal 2DUS clearly shows a cleft that goes up to the subarachnoid space, thus confirming that this case was Type II. Trans-fontanel 2DUS makes the cleft location and extension obvious, while 3DUS trans-fontanel in the TUI mode demonstrates bilateral extension of the cleft to the subarachnoid space, thus showing that this case was Type II [Figure 5]. Figure 6 shows the pre- and postnatal findings from 2DUS in a case of extensive bilateral schizencephaly, in which the subarachnoid space was reached, with widespread destruction of the brain parenchyma. In this case, 3DUS was not necessary for confirming this type of schizencephaly. Figure 7 shows a case of extensive bilateral schizencephaly, in the temporal-occipital region. The prenatal 2DUS demonstrates a large posterior cystic cavity, thus suggesting a diagnosis of holoprosencephaly. A 3DUS in TUI mode shows that this case is schizencephaly Type II, caused by non-fusion of the thalami.
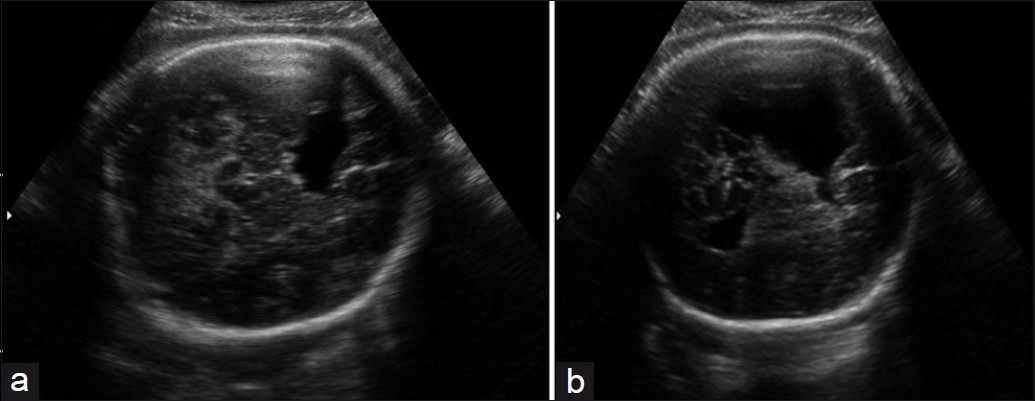
- (a) and (b) Prenatal 2DUS images show the presence of a large cleft in the left parietotemporal region of the brain, extending to the lateral ventricle. 2DUS: two-dimensional ultrasound.
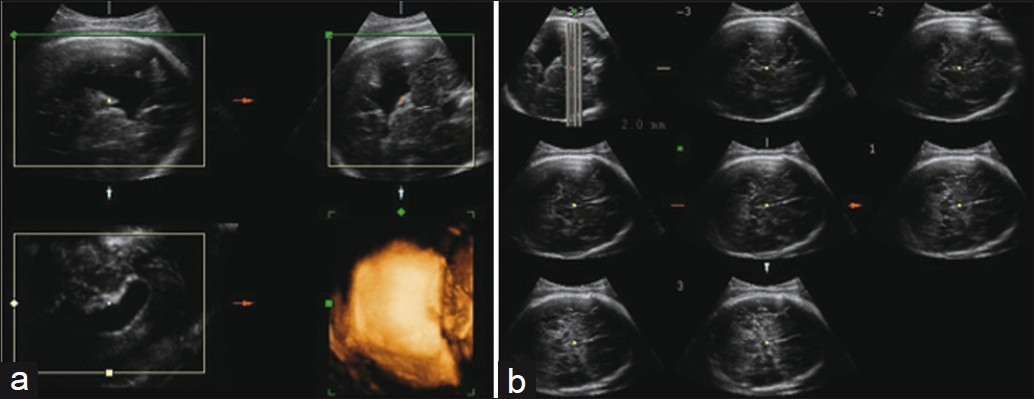
- (a) Prenatal 3DUS images in multiplanar mode (top right — axial plane; top left — sagittal plane; lower right — coronal plane; lower left — rendering mode) clearly show the spatial relationship between the cleft (white arrow) and the ventricular system. (b) 3DUS in TUI mode (eight axial sequential planes, with 2.0 mm intervals) shows the lesion level. 3DUS: threedimensional ultrasound; TUI: tomographic ultrasound imaging.
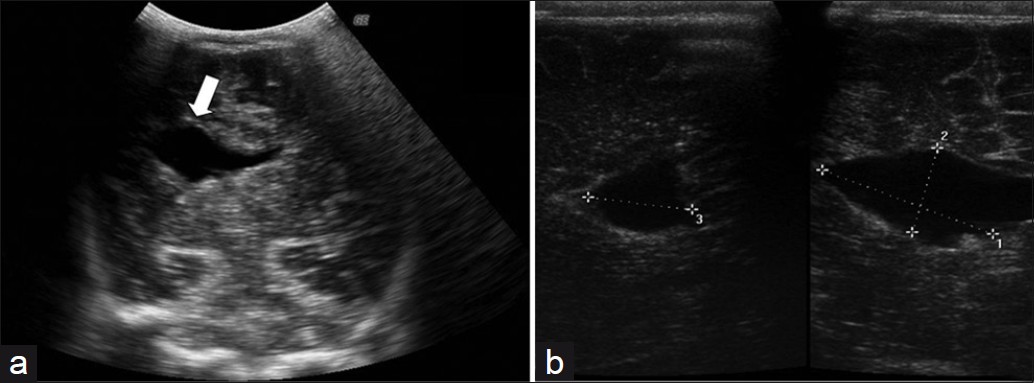
- A postnatal trans-fontanel 2DUS, shows (a) the location of the cleft in the cerebral hemisphere (white arrow) and (b) its measurements in the axial (right) and sagittal (left) planes. 2DUS: two-dimensional ultrasound.
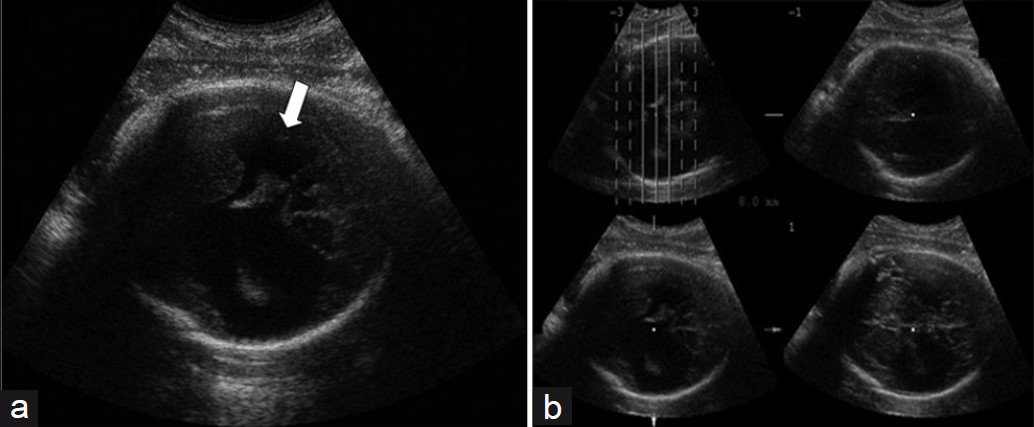
- (a) Prenatal 2DUS reveals bilateral schizencephaly with a cleft going up to both hemispheres (white arrow). (b) 3DUS in TUI mode (four axial sequential planes, with 8.0 mm intervals), clearly shows that the cleft goes up to the subarachnoid space, thus confirming that it is Type II. 2DUS: two- dimensional ultrasound; 3DUS: three-dimensional ultrasound; TUI: tomographic ultrasound imaging.
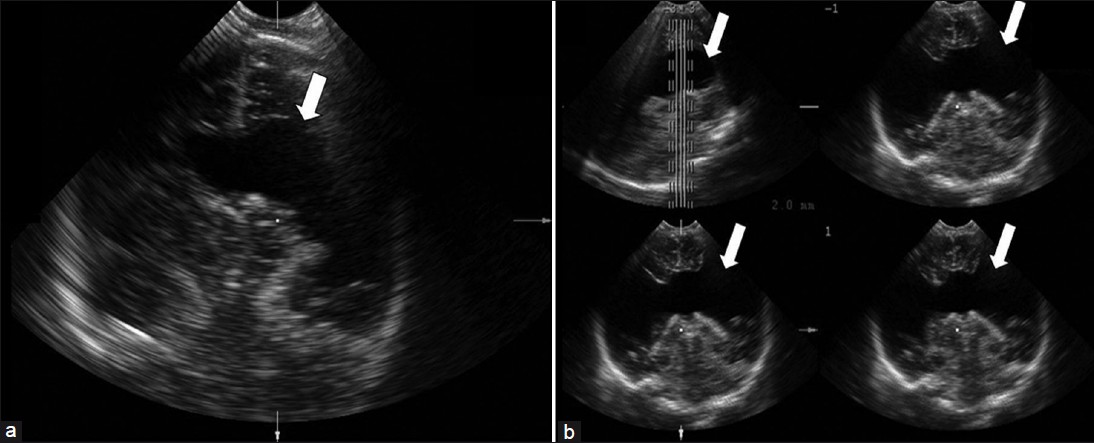
- (a) Postnatal trans-fontanel 2DUS, making it obvious that the cleft extends to the subarachnoid space (Type II) (white arrow). (b) Trans-fontanel 3DUS in TUI mode (four sagittal sequential planes, with 2.0 mm intervals) demonstrate that the cleft extends bilaterally to the subarachnoid space, thus proving that it is Type II (white arrows). 2DUS: two-dimensional ultrasound; 3DUS: three-dimensional ultrasound; TUI: tomographic ultrasound imaging.
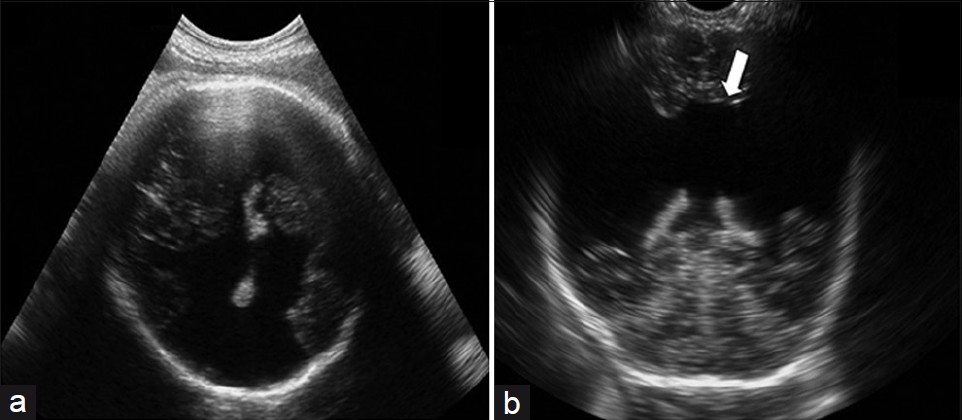
- (a) Prenatal and (b) postnatal 2DUS findings in a case of extensive bilateral schizencephaly, reaching the subarachnoid (Type II) space, with widespread destruction of the brain parenchyma (white arrow). 2DUS: twodimensional ultrasound.
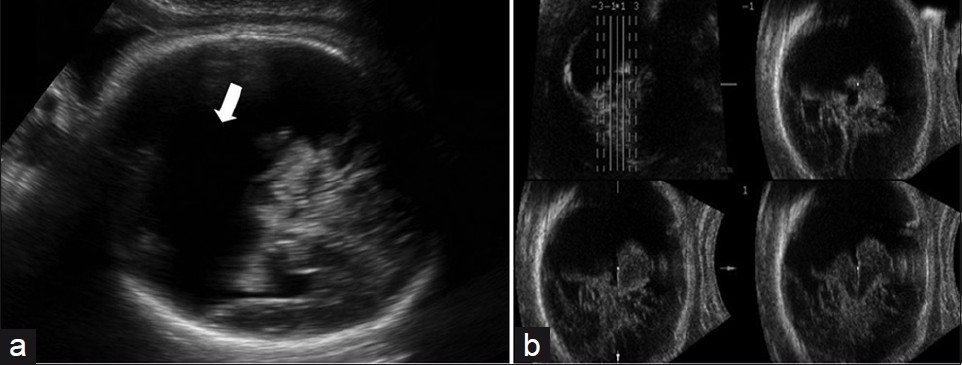
- (a) Prenatal 2DUS in a case of extensive bilateral schizencephaly, with a large posterior cystic cavity, suggesting holoprosencephaly (white arrow); (b) 3DUS in TUI mode (four coronal sequential planes, with 3.0 mm intervals) proved that this was schizencephaly Type II, because of non-fusion of the thalami. 2DUS: two-dimensional ultrasound; 3DUS: three-dimensional ultrasound; TUI: tomographic ultrasound imaging.
There were no closed-lip schizencephaly (Type I) cases among our series.
DISCUSSION
Schizencephaly is usually associated with other malformations of the central nervous system, such as, ventriculomegaly, polymicrogyria, pachygyria, heterotopias, and lissencephaly. It may also be associated with the absence of the cavum septum pellucidum, agenesis of the corpus callosum, and optic nerve hypoplasia.[1] The clinical manifestations of schizencephaly include different levels of retardation of neuropsychomotor development and seizures, and is usually associated with the extent of the lesions.[4] Therefore, an accurate prenatal diagnosis is important to enable counseling of parents and in some cases, to suggest termination of the pregnancy. The differential diagnosis is done to distinguish this disorder from holoprosencephaly, hydranencephaly, and bilateral arachnoid cysts.[6]
Two-dimensional ultrasonography may lead to a prenatal diagnosis, indicating a suspicion of a closed or open-lip cleft.[7] However, this communication is often not easily identified. The birth deficiency monitoring program of California found that the schizencephaly rate was 1:70,000 live births, although prenatal suspicion was only raised in 63 cases.[8] It is often necessary to do other imaging examinations, such as, magnetic resonance imaging (MRI),[5] but this technique is still expensive and restricted to a few reference centers. Moreover, it is contraindicated for patients with metallic implants or individuals who are claustrophobic. Likewise, MRI and computed tomography (CT) have been used postnatally to prove the suspected obstetric ultrasonographic diagnosis. Oh et al.,[9] found that postnatal MRI made it possible to outline the gray matter that encased the cleft and to show its communication with the lateral ventricle. In addition, a MRI made it possible to identify other lesions associated with the central nervous system (CNS), with greater accuracy. Similar to our study, Oh et al.,[9] also evaluated six cases of schizencephaly, which were also all Type II. In only one of their cases was it impossible to view the cleft using 2DUS. For this case, a MRI was required. Thus, it has been proven that 2DUS is the main prenatal diagnostic method for schizencephaly - A method that is lower in cost with greater accessibility than a MRI.
Magnetic resonance imaging makes it possible to evaluate almost all fetal cerebral malformations, and this is of particular importance in diagnosing cortical developmental disorders, which are poorly imaged using 2DUS. These malformations include lissencephaly, heterotopias, polymicrogyria, and schizencephaly.[10] However, in our study, by using 2D and 3D US pre- and postnatally, we were able to diagnose all the six cases of schizencephaly.
Huang et al.,[5] described the use of 3DUS in rendering mode for characterizing the cleft type, but the cleft type diagnosis had already been made using 2DUS. Lee et al.,[11] described difficulty in identifying brain clefts in examinations done at the beginning of the second trimester, using abdominal 2DUS. They also reported that it might be easier to see the cleft via vaginal ultrasonography,[11] but this route was not used for our group of patients. In the case described by Lee et al.,[11] abdominal 3DUS in the TUI mode was used, but this technique was not efficient for identifying the cleft. In our cases, 3DUS in multiplanar and TUI mode were essential for indicating the connection between the cleft and the ventricles or subarachnoid space. Regarding postnatal trans-fontanel ultrasonography, there was only one previous description using 3DUS. Araujo Júnior et al.,[6] described a case of schizencephaly that had been suspected in the twenty-ninth week of pregnancy, which was confirmed postnatally by means of a cranial CT and trans-fontanel 3DUS. 3DUS in rendering mode allowed greater accuracy in assessing the degree of ventriculomegaly, which clearly demonstrated cleft communication with the subarachnoid space, besides allowing the parents to have a better comprehension of the cerebral malformations and the prognosis for the newborn. In our study, we used trans-fontanel 3DUS in multiplanar and TUI modes, which were important for confirming the types of schizencephaly, in cases that were suspected prenatally.
In summary, we present here a pictorial essay on cases of schizencephaly that had been suspected prenatally and were confirmed postnatally by 2D and 3D US. 3DUS in multiplanar and TUI modes may be important for confirming the schizencephaly type suspected by prenatal 2DUS, especially in cases with smaller clefts, and for differential diagnosis with other cerebral malformations like holoprosencephaly.
Source of Support: Nil
Conflict of Interest: None declared.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2012/2/1/30/96546
REFERENCES
- Schizencephalies: A study of the congenital clefts in the cerebral mantle. II. Clefts with hydrocephalus and lips separated. J Neuropathol Exp Neurol. 1946;5:169-206.
- [Google Scholar]
- Nonlissencephalic cortical dysplasias: Correlation of imaging findings with clinical deficits. Am J Neuroradiol. 1992;13:95-103.
- [Google Scholar]
- Schizencephaly: Antenatal and postnatal assessment with color-flow Doppler imaging. Can Assoc Radiol J. 1994;45:193-200.
- [Google Scholar]
- Prenatal diagnosis of schizencephaly with septo-optic dysplasia by ultrasound and magnetic resonance imaging. J Obstet Gynaecol Res. 2008;34:674-9.
- [Google Scholar]
- Postnatal evaluation of schizencephaly by transfontanellar three-dimensional sonography. J Clin Ultrasound. 2008;35:351-5.
- [Google Scholar]
- Schizencephaly: Heterogeneous etiologies in a population of 4 million California births. Am J Med Genet A. 2005;137:181-9.
- [Google Scholar]
- Schizencephaly: Pre- and postnatalimaging with a review of the clinical manifestations. Radiographics. 2005;25:647-57.
- [Google Scholar]
- Prenatal imaging of congenital malformations of the brain. Semin Ultrasound CT MR. 2011;32:167-88.
- [Google Scholar]
- Prenatal diagnostic challenges and pitfalls for schizencephaly. J Ultrasound Med. 2009;28:1379-84.
- [Google Scholar]






