Translate this page into:
Penile Metastases of Recurrent Prostatic Adenocarcinoma without PSA Level Increase: A Case Report
Address for correspondence: Dr. Antonio Pierro, Radiology Department, Fondazione di Ricerca e Cura “Giovanni Paolo II,” Largo A. Gemelli, 1-86100 Campobasso, Italy. E-mail: apierrojonico@libero.it
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
We report a case of penile metastases from recurrent prostatic adenocarcinoma that was the first sign of a widespread metastatic disease in the absence of any increase in prostate-specific antigen (PSA) level. In April 2011, an 80-year-old man presented to our Radiotherapy Unit with multiple palpable hard nodules in the penis, dysuria, and moderate perineal pain, 7 years after he had received radiotherapy for prostate cancer. Nodules in the penis had appeared in February 2011. The ultrasound and magnetic resonance (MR) imaging suggested the diagnosis of multiple penile metastases. A total body computed tomography scan revealed a systemic spread of the disease, with multiple metastases in the liver, bones, and lungs. PSA level was 0.126 ng/ml. A fine needle aspiration biopsy of the liver lesion was undertaken, and the histopathologic examination revealed the prostatic origin of the metastases, so androgen deprivation therapy was started. The diagnosis of metastases should be considered in a patient with prior history of prostate malignancies presenting with solid nodules in the penis, even if the PSA level is low.
Keywords
Malignant priapism
penile metastasis
penile nodules
perineal pain
prostate cancer
INTRODUCTION

Metastases to the penis are rare, with less than 400 cases reported in the literature.[1] Malignant priapism, urinary retention, penile nodules, perineal pain, dysuria, and hematuria are the most frequently observed clinical signs of metastases to the penis.[2]
Pelvic organs, particularly the bladder, prostate, and rectum, are the primary sites of origin of the tumors that most commonly metastasize to the penis. Other primary sites of metastases are rare.[1]
We report a case of penile metastases from recurrent prostatic adenocarcinoma with normal prostate-specific antigen (PSA) levels.
CASE REPORT
In January 2004, a 73-year-old male patient was diagnosed with prostatic carcinoma, stage T2N0M0. Initial total PSA was 6.7 ng/ml. Trans-rectal ultrasound showed prostatic carcinoma confined to the right prostatic gland without deformation of the prostatic capsule. This was confirmed by MRI imaging performed with an 8-channel pelvic phased-array surface coil (1.5 T system, GE Signa Excite HD, GE Medical Systems, Milwaukee, WI, USA). Magnetic resonance imaging (MRI) demonstrated [Figure 1] the presence of a nodular area of altered signal, mildly hypointense on T2-weighted imaging, in the paramedian right lobe of the prostate, plus a similar area in the peripheral zone of paramedian left lobe. The prostatic capsule was intact and the seminal vesicles were symmetrical. The prostatic biopsy confirmed adenocarcinoma in right and left median lobes with Gleason score of 3 + 3. Prostate biopsy was performed with ultrasound guidance. Neoadjuvant hormonal therapy with Luteinizing hormone releasing hormone (LHRH) analogs was prescribed for 3 months, and thereafter, from September to November 2004, the patient underwent concomitant hormone and radiotherapy. A prophylactic dose of 45 Gy was delivered to pelvic lymph nodes using 3D-conformal irradiation, followed by a dose escalation to 56 Gy to the seminal vesicles and the prostate gland, and 73.8 Gy to the prostate only, using intensity-modulated technique (IMRT). The fractionation dose was 1.8 Gy/day for all the volumes. Plans were optimized with Plato treatment planning system (Nucletron B.V., Veenendaal, The Netherlands). Three-dimensional conformal radiotherapy (3D-CRT) plan for lymph-node irradiation was achieved with the box technique using four beams at 15 MV (0°, 90°, 180°, and 270°) collimated with standard multileaf collimator (MLC) (2 × 40 leaves, width 1 cm at the isocenter). IMRT plans were generated using the ITP/Plato inverse planning algorithm with step and shoot modality and 6 MV beam energy. A class solution based on five coplanar beams consisting of the gantry angles 36°, 108°, 180°, 252°, and 324° was selected. The patient completed the radiotherapy course without interruptions, showing Grade 2 and Grade 1 genitourinary and gastrointestinal acute toxicity, respectively. No adjuvant hormonal therapy was prescribed.
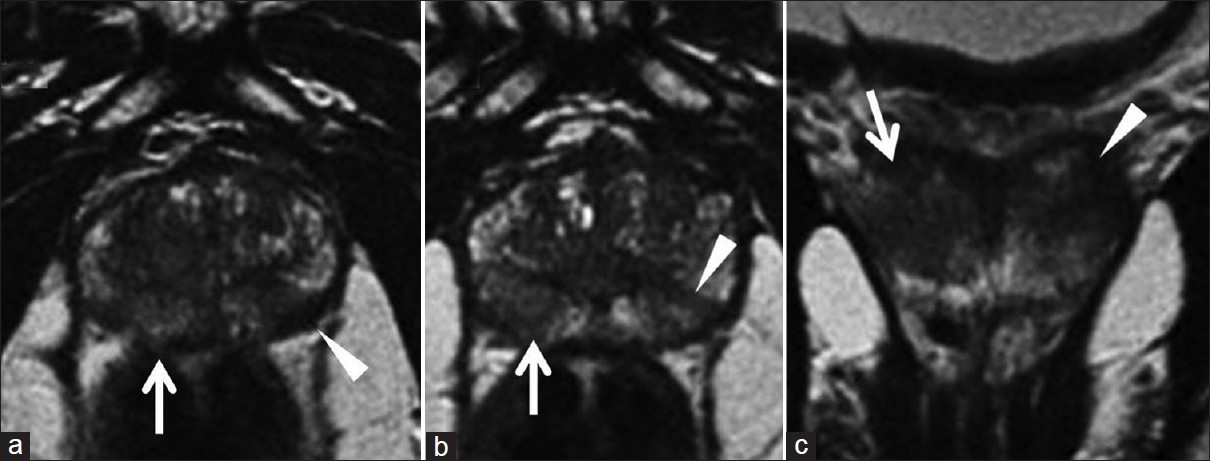
- MRI T2-weighted images in (a, b) axial and (c) coronal views show two areas of altered signal, with oval-like morphology and hypointensity, respectively, localized in the peripheral region of the right (white arrow) and the left gland (white arrowhead).
One-year later, rectoscopy documented Grade 1 proctitis without clinical symptoms. Total PSA was 0.1 ng/ml for 3 years after the end of treatment, when a biochemical increase to 0.43 ng/ml was documented, with a further rise of total PSA to 0.94 and 1.6 ng/ml in the following 2 years. A Grade I late intestinal toxicity after 5 years was recorded.
The patient was lost to follow-up for 2 years, until April 2011, when he came back to our Radiotherapy Unit with the appearance of multiple palpable hard nodules over the penis, dysuria, and moderate perineal pain. The nodules in the penis had appeared in February 2011 and the patient had been admitted to another hospital for the checkup. The ultrasound images [Figure 2] at the time of admission revealed the presence of hypoechoic solid nodules, with intra-lesional vascularization near the dorsal surface of the bilateral corpora cavernosa in the echo-color Doppler examination. MRI images (T2-weighted images at high resolution) also showed solid nodules strictly adhering to the corpora cavernosa [Figure 3]. CT [Figure 4] scan confirmed the presence of metastases to the cavernous bodies in a context of disseminated disease, with multiple pulmonary, bone, and liver metastases. PSA level was 0.126 ng/ml. A fine needle aspiration biopsy of liver lesion was taken, and the histopathologic examination revealed the prostatic origin of the metastases. So, androgen deprivation therapy was started in March 2011. A month later, given that the androgen deprivation therapy did not provide any clinical benefit, the patient was placed on metronomic therapy with cyclophosphamide and palliative radiotherapy for the painful bone metastasis, but he died after 2 months.
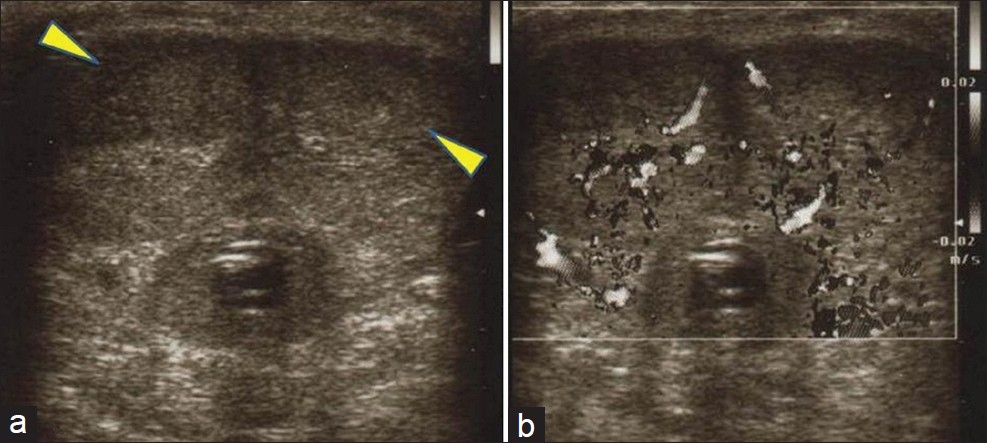
- Gray-scale ultrasound images (a, b) show hypoechoic areas (yellow arrowhead), with intra-lesional vascularization on color flow Doppler examination, near the dorsal surface of the both corpora cavernosa.
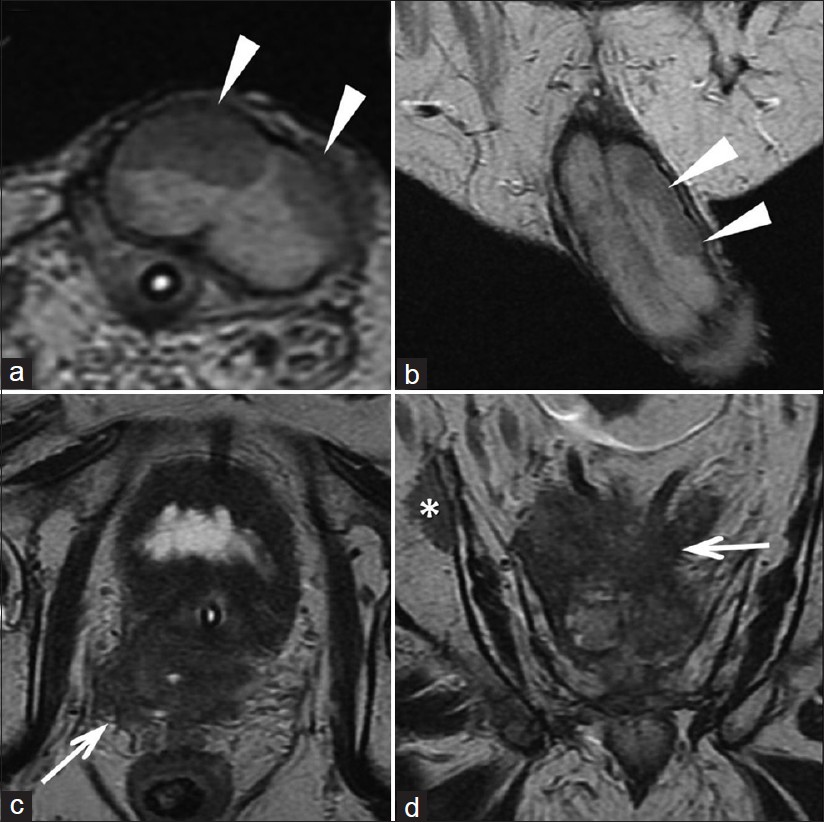
- The T2-weighted images (a: axial and b: coronal) show the presence of nodules adherent to both cavernous bodies (white arrowheads). The T2- weighted images (c) and (d) show the loss of the normal architecture of the prostate due to solid tissue (recurrence of disease) which invades the region of the seminal vesicles and infiltrates the mesorectum (white arrows). Image (d) shows a bone metastasis (asterisk).
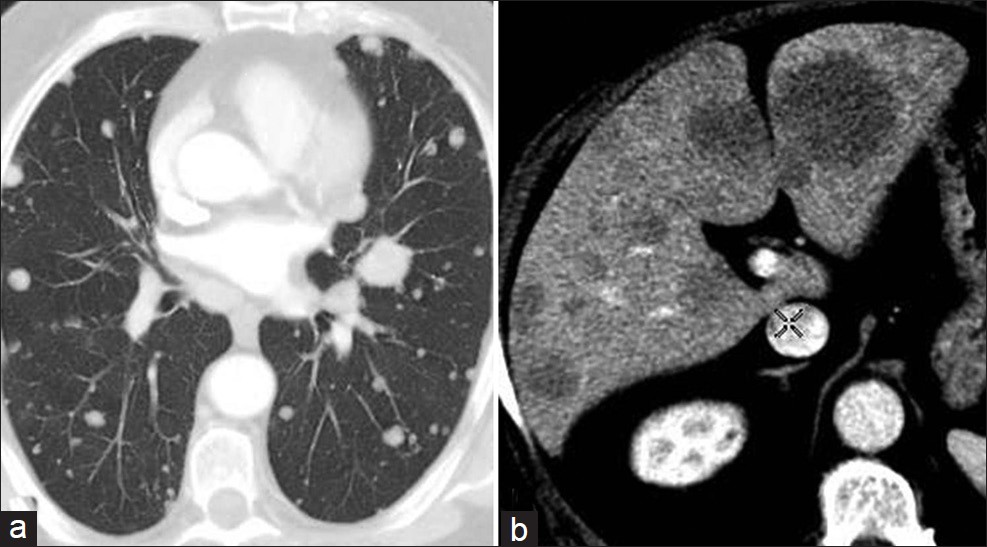
- CT images through the (a) lung and (b) liver demonstrate multiple metastatic lesions.
DISCUSSION
The atypical features of our case were both the presence of penile metastases and the presence of a widespread disease despite a low level of PSA (0.12 ng/ml).
Metastases to the penis are rare, and biopsy of nodules is confirmatory.[3] Our patient also had liver, lung, and bone metastases, so it was decided to perform a needle biopsy of the liver lesion. Progression of prostate cancer in the presence of low or undetectable PSA is uncommon.[4] In our patient, the PSA level was 0.12 ng/ml. Patients who experience disease progression with low PSA levels usually have Gleason scores >7, atypical histologic variants, particularly small cell and ductal cancers, and locally advanced tumors.[4] None of these features was present in our patient.
Median survival time of only 6 months has been reported for patients with penile metastases.[5] Our patient died of metastatic disease 2 months after the appearance of penile metastases. Prostate cancer progression without increase of PSA level may reflect a clonal shift in the original tumor that may express the features of small cell prostate cancer, including biologic aggressiveness and lack of PSA production. This subset of prostate cancer may show a completely different biologic behavior than that of the garden-variety adenocarcinoma of the prostate.[6] This manifests not only with disease progression unaccompanied by PSA elevation, but also with a more aggressive nature and atypical metastatic sites.[7]
Given the poor prognosis of this condition, usually the treatment of penile metastases is considered only a stop-gap measure to improve the quality of life of patients.[8] Surgery, radiotherapy, hormone therapy, or chemotherapy may all represent a viable therapeutic option depending on the general clinical condition of the patient, the type and extent of the primary tumor, the presence of widespread metastatic disease, and the type of symptoms. Our patient received metronomic chemotherapy and radiotherapy for the painful bone metastases. Unfortunately, he died after 2 months from the onset of penile metastases.
CONCLUSIONS
The radiologist must be familiar with the rare possibility of metastasis to the penis in patients with prostate cancer, even if PSA levels are low, as this condition is associated with a poor prognosis and is often a sign of disseminated metastatic disease.
Source of Support: Nil
Conflict of Interest: None declared, Not submitted to Congress; No funding;
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2012/2/1/44/99178
REFERENCES
- Penile metastasis from other malignancies.A study of ten cases and review of the literature. Urol Int. 2006;76:118-21.
- [Google Scholar]
- Priapism secondary to penile metastasis: A report of two cases and a review of literature. J Surg Oncol. 1998;68:51-9.
- [Google Scholar]
- Prostate cancer progression in the presence of undetectable or low serum prostate-specific antigen level. Cancer. 2007;109:198-204.
- [Google Scholar]
- Metastatic tumors to the penis: A report of 17 cases and review of the literature. Int J Surg Pathol. 2011;19:597-606.
- [Google Scholar]
- Progression from adenocarcinoma to small cell carcinoma of the prostate with normalization of prostate-specific antigen (PSA) levels. Scand J Urol Nephrol. 1996;30:509-12.
- [Google Scholar]
- Small cell and anaplastic prostate cancer: Correlation between CT findings and prostate-specific antigen level. Radiology. 1998;208:735-8.
- [Google Scholar]
- Penile metastasis of prostatic adenocarcinoma: Report of two cases and review of literature. World J Surg Oncol. 2003;1:16.
- [Google Scholar]






