Translate this page into:
Not Every Fat-Containing Lesion on Mammogram is a Benign Finding: Case Report and Review of Breast Malignant Phyllodes Tumor with Heterologous Liposarcomatous Differentiation
* Corresponding author: Dr. Fabiana Policeni, Department of Breast Imaging, UIHC, Iowa City 52246, IA, USA. fabianachacur@hotmail.com
-
Received: ,
Accepted: ,
Abstract
Phyllodes tumors (PTs) are a rare fibroepithelial tumor of the breast. Histopathological confirmation of stromal and epithelial component is required for making the diagnosis of PT, and stromal component carries an increased risk of sarcomatous transformation. We present imaging findings of a histologically proven case of malignant PT with heterologous liposarcomatous transformation of the breast.
Keywords
Fibroepithelial neoplasm
heterologous liposarcomatous differentiation
phyllodes tumors

INTRODUCTION
Phyllodes tumors (PTs) are a rare fibroepithelial tumor of the breast which accounts for <1% of breast tumor and approximately 2%–3% of the breast fibroepithelial neoplasms. PTs are common in 40–50 year age group and usually benign with malignant potential in up to 30% of cases.[1,2] The diagnosis of PTs requires histopathological confirmation of both epithelial and stromal components. Stromal component carries the high risk for the malignant transformation to usually fibrosarcomatous and rarely heterologous sarcomatous differentiation. Malignant transformation of epithelial elements is very rare. Complete surgical removal with negative margin is the definitive treatment to avoid local recurrence.[3,4] According to the World Health Organization (WHO), PTs are classified into benign, borderline, and malignant based on the histological features.[5] Benign PT mimics fibroadenoma and their differentiation from malignant PT (MPT) is difficult on core biopsy. Similarly, differentiation of the MPTs from spindle cell metaplastic carcinoma and primary breast sarcoma can also be challenging. MPTs carry a poor prognosis with an overall risk of local recurrence rates of 10%–40% and distant metastasis of 20%.[6] Fatty lesions seen on mammograms and ultrasound tend to be either hamartoma or lipoma, with a malignant lesion thought of as a distant differential. Patients with liposarcomatous differentiation, like in our case, may also develop as malignant differentiation of stromal components of PTs. The finding of such a malignant heterologous element in PTs places the tumor into a malignant category.[7] The prior reports of these such masses mention their rarity; however, they lack the discussion of imaging findings including correlation with breast magnetic resonance imaging (MRI).
We present a histologically proven case of MPT with heterologous liposarcomatous transformation of the breast and that was found on a screening mammogram. This was explored using other imaging modalities such as an ultrasound, breast MRI, and positron emission tomography (PET)–computed tomography (CT) scan. This case demonstrates that all macroscopic fat-containing lesions on mammography are not benign. This rare case of MPT with heterologous liposarcomatous differentiation mimicking an intracystic tumor on sonography is presented with a review of the literature, pertinent imaging findings, and histopathological diagnostic criteria.
CASE REPORT
A 66-year old female presented for a screening mammogram at the outside hospital which showed a 2.8 cm mixed density well-circumscribed mass with subtle areas of fat density in the left breast at the 3 o’clock position [Figure 1]. Diagnostic ultrasound was performed and showed a well-circumscribed predominantly hyperechoic mass [Figure 2]. The only pertinent history was of breast carcinoma in her mother after the age of 50 years. Ultrasound-guided core biopsy was performed, and pathology showed a cellular neoplasm composed of variably sized adipocytes with large, hyperchromatic, atypical nuclei and numerous lipoblasts. Epithelial and stromal elements required for making a diagnosis of the PT were not present in the specimen, and definitive classification was deferred till excisional evaluation of the neoplasm.
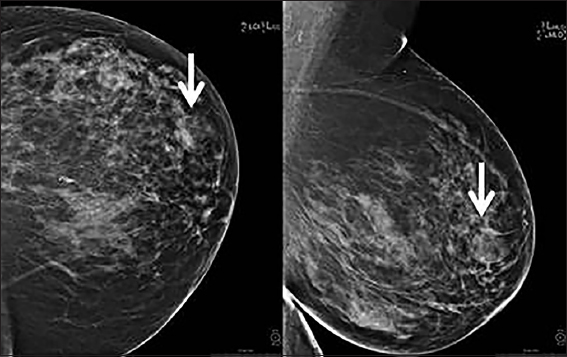
- A 66-year-old female with abnormal screening mammogram showing a mixed density circumscribed mass with areas of fat density in the left breast at the 3 o’clock position (arrow).
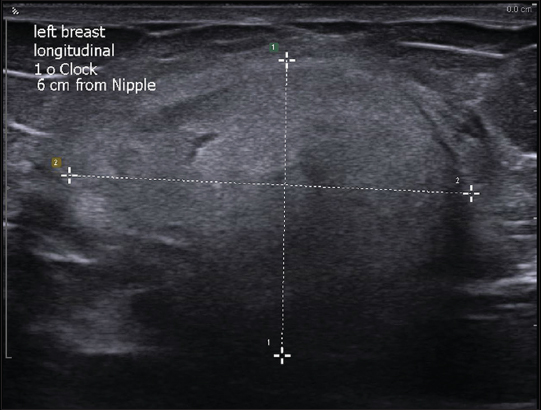
- A 66-year-old female with external ultrasound showing well-circumscribed hyperechoic mass in the left breast.
At this point, the patient was referred to our hospital for further workup and a second opinion. Breast MRI was performed for surgical planning and showed irregular enhancing breast mass. This also showed areas of T1 hyperintensity suppressing on short-tau inversion recovery, likely composed of fatty tissue [Figure 3]. PET scan was also performed to evaluate distant metastases and showed the area of mildly increased fluorodeoxyglucose uptake in the left breast with no areas of lymphadenopathy or avid distant metastasis [Figure 4]. Imaging findings were consistent with aggressive fat-containing lesions such as liposarcoma, MPTs with liposarcomatous differentiation, and metaplastic liposarcomas. The mass was scored as breast imaging-reporting and data system 5. For confirmation, the patient underwent left breast lumpectomy with additional margins and immediate oncoplastic reconstruction of the left breast. Resection specimen [Figures 5 and 6] showed a fibroepithelial lesion with increased stromal mitotic rate (up to 10 mitoses per 10 high-power fields [HPFs]) and heterologous liposarcomatous differentiation. Margins were negative for the in situ and invasive carcinoma. The negative Murine double minute2 (MDM2) and cyclin-dependent kinase4 (CDK4) stains made the diagnosis of well-differentiated liposarcoma (WDL) less likely. Oncological treatment in the form of radiotherapy or chemotherapy was not conducted.
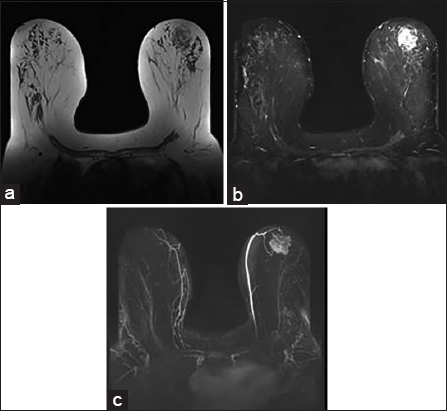
- A 66-year-old female with breast magnetic resonance imaging shows heterogeneous mass with areas of T1 hyperintensity (a) suppressing on short-tau inversion recovery (b) in the left breast. Postcontrast maximum intensity projection image shows irregularly enhancing mass (c).
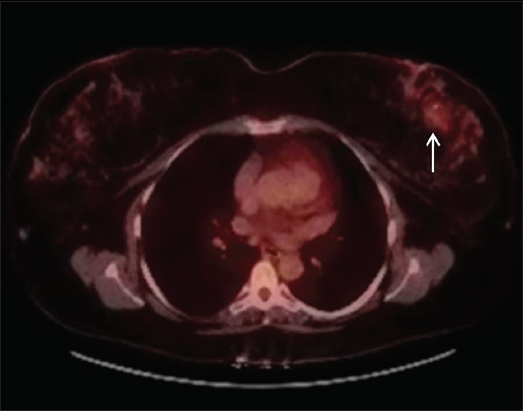
- A 66-year-old female with malignant phyllodes tumor on biopsy of the left breast mass with positron emission tomography–computed tomography scan showing area of mild uptake in left breast (arrow) with no lymphadenopathy or metastasis.
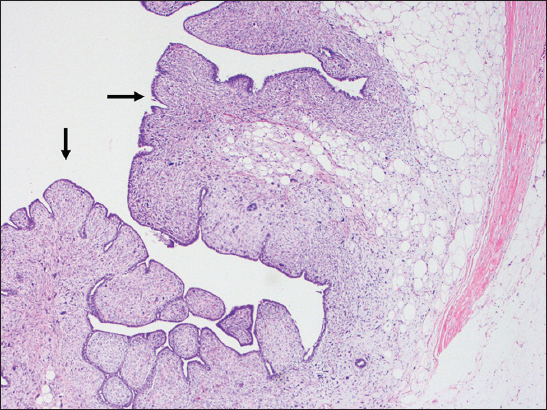
- A 66-year-old female with biopsy of the breast mass. Tumor pathology slides showing a fibroepithelial lesion showing “leaf-like” architecture (arrows) of the phyllodes tumor (H and E, ×4).
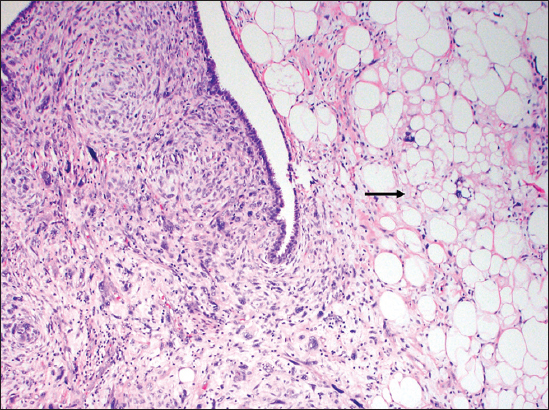
- A 66-year-old female with biopsy of the breast mass. Tumor pathology slides showing heterologous liposarcomatous differentiation with variably sized, hyperchromatic, atypical adipocytes (arrow), numerous lipoblasts and high mitotic rate (H and E, ×10).
DISCUSSION
PTs are uncommon fibroepithelial neoplasms of the breast. PTs are graded as per the WHO into benign, borderline, and malignant based on histological parameters, that is, the degree of stromal cellularity and atypia, mitotic count, stromal overgrowth, and surgical margin. Among all surgical margins, status is the most important.[5,6] These histological features are helpful but not always accurate predictors of tumor behavior in all cases.[8]
Benign PTs closely resemble fibroadenoma and show mildly increased stromal cellularity with minimal nuclear atypia (mitoses of ≤4/10). MPTs show marked stromal cellularity and atypia, with at least 10/10 HPFs mitotic activity. Grading of PT is of clinical significance to predict the clinical behavior. Benign PTs have a risk of local recurrence, while the MPTs have a higher risk of metastasis. Regardless of the histological parameters, malignant heterologous element such as liposarcoma, chondrosarcoma, or osteosarcoma consigns the tumor into the malignant category.
PTs with heterologous liposarcomatous differentiation in the breasts are extremely rare lesions which can be spontaneous from the stromal parenchyma or can differentiate from PTs. Assessing on multiple modalities for concerning fatty lesions is essential for diagnosis. A study by Inyang et al., evaluated five cases of MPTs with regions identical to well-differentiated liposarcoma (WDL) for MDM2 and CDK4 gene expression and amplification using immunohistochemistry and fluorescence in situ hybridization. Despite indistinguishable morphology, all cases of MPTs with WDL-like liposarcomatous differentiation were negative for MDM2 and CDK4, supporting different underlying pathogenesis, like our case.[9]
Mammogram has been described as showing well-circumscribed fat-containing masses in the setting of PTs, which can mimic benign masses such as hamartomas, fat necrosis, galactocele, and fibroadenomas. The role of imaging is to differentiate benign from malignant fat-containing lesions such as liposarcomas and MPT with liposarcomatous differentiation. Fat necrosis can show varying imaging appearance and may mimic malignant lesion with areas of low signal intensity on T1-weighted and T2-weighted (T2W) images corresponding to blood products. Hamartomas also show varying imaging appearances depending on the fat-parenchyma component. On US (Ultrasound), liposarcomas are heterogenous with areas of fat as hyperechogenicity. MRI demonstrates intratumoral fat with diffuse enhancement. Mammograms with correlating ultrasonographic findings are generally enough to make the diagnosis, and MRI can be valuable to further confirm the presence of fat.[10]
Ultrasound will generally show complex solid or cystic masses with areas of mixed echogenicity that may or may not be well circumscribed. Clinical suspicion and presentation again may aid in diagnosis. MRI can help to diagnose and differentiate the benign from MPTs which is not well described in the literature.[11] Yabuuchi et al., correlated the magnetic resonance findings of 30 benign, intermediate, and MPTs with histological grade. They demonstrated areas of stromal hypercellularity as low apparent diffusion coefficient signal on diffusion-weighted images and hypo-to-isointense signal on T2W images.[11] We found similar mass with early enhancement with delayed washout in an irregularly shaped mass. Our case is likely one of the few cases in the literature showing MRI findings of MPT with heterologous liposarcomatous differentiation in the breast.
Once the diagnosis is made, PET–CT scan and CT chest, abdomen, and pelvis are typically done to assess for metastatic disease. Wide surgical excision with at least 10 mm of negative margins is the mainstay treatment for MPTs, while a conservative approach can be conferred to benign PTs. Adjuvant radiation and less commonly chemotherapy can be utilized depending on metastatic disease burden and positive surgical margins.
CONCLUSION
We present a case of a fatty breast mass found on screening mammogram, which was excised and proved to be MPT with heterologous liposarcomatous differentiation. Our case highlights that not all fatty lesions are benign. Our case is one of the first few cases, demonstrating MRI findings of MPTs with heterologous liposarcomatous transformation of the breast. This should always be included in the differential with a history of insidiously growing breast mass and concerning imaging findings. It is extremely important to make the preoperative correct diagnosis because of its malignant potential and tendency to recur in MPTs. Additional modalities can be performed, and the patient can get adequate management of tumor which is surgery.
Declaration of patient consent
Patient consent was not obtained because no patient identity is being disclosed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- In-situ and invasive carcinoma within a phyllodes tumor associated with lymph node metastases. World J Surg Oncol. 2004;2:46.
- [Google Scholar]
- Surgical treatment of phyllodes tumors of the breast:Retrospective review of 172 cases. J Surg Oncol. 2005;91:185-94.
- [Google Scholar]
- Recurrent cystosarcoma phyllodes:A clinicopathologic study of 32 cases. Cancer. 1976;38:1402-6.
- [Google Scholar]
- “Fibroepithelial Tumours,” in World Health Organization Classification of Tumours of the Breast and Female Genital Organs. Lyon, France: IARC Press; 2003. p. :99-103.
- World Health Organization Classification of Tumours of the Breast. Lyon: IARC Press; 2012.
- Primary pleomorphic liposarcoma of breast:A rare case report. Indian J Pathol Microbiol. 2011;54:124-6.
- [Google Scholar]
- Predicting clinical behaviour of breast phyllodes tumours:A nomogram based on histological criteria and surgical margins. J Clin Pathol. 2012;65:69-76.
- [Google Scholar]
- Heterologous liposarcomatous differentiation in malignant phyllodes tumor is histologically similar but immunohistochemically and molecularly distinct from well-differentiated liposarcoma of soft tissue. Breast J. 2016;22:282-6.
- [Google Scholar]
- Imaging of fat-containing lesions of the breast:A pictorial essay. J Clin Ultrasound. 2013;41:424-33.
- [Google Scholar]
- Phyllodes tumor of the breast:Correlation between MR findings and histologic grade. Radiology. 2006;241:702-9.
- [Google Scholar]






