Translate this page into:
Intracranial Pial Arteriovenous Fistulae: Diagnosis and Treatment Techniques in Pediatric Patients with Review of Literature
Address for correspondence: Dr. Lakshmi Sudha Prasanna Karanam, Department of Neurointervention, King Edward Memorial Hospital, Pune - 411 011, Maharashtra, India. E-mail: drklsp@gmail.com
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Pial arteriovenous fistulae (AVF) are rare vascular lesions comprising single or multiple arterial feeders draining directly into the venous channel without intervening tangle of blood vessels as in brain AV malformations. In our present paper, we describe three cases of pial AVF in the pediatric age group with different presentations treated successfully with endovascular and surgical methods. Two patients underwent treatment for pial AVF by the endovascular technique and one by surgical clipping. The treatments were successful with good clinical outcome. We did not encounter any peri-procedural complications in any of the cases. Pial AVF is a rare entity and high degree of suspicion and adequate clinical and imaging knowledge is required to make the diagnosis. Exclusion of the fistula from the cerebral circulation should be done at the earliest to prevent devastating complications that result during the natural course of the disease.
Keywords
Coiling
endovascular
pial fistula
INTRODUCTION
Pial arteriovenous fistulae (AVF) are rare vascular lesions and account for 1.6% of intracranial vascular malformations.[1] This differs from brain AV venous malformation, which has an intervening nidus between artery and the draining vein.
In pial fistula, the artery directly communicates with the vein with no intervening tangle of vessels and hence there is more pressure gradient which makes this lesion more vulnerable to rupture and cause symptoms leading to poor prognosis for patients with pial AVF.
These lesions should be identified carefully at the earliest and should be treated immediately either by endovascular or surgical modality because of the associated high mortality seen in untreated cases.[2] In our present report, we describe three cases of pial AVF in the pediatric age group treated by endovascular and surgical methods with good clinical outcomes.
CASE SERIES
Case 1
A 14-year-old male child presented to the Emergency Room with sudden onset headache, vomiting, and brief loss of consciousness. Computed tomography (CT) scan of the brain showed diffuse subarachnoid hemorrhage [Figure 1]. Digital subtraction angiogram (DSA) showed infratentorial pial AVF with arterial feeder from the left posterior cerebral artery and enlarged venous varices draining into vertebral venous plexus [Figure 2]. Because of the poor natural history and the location of the lesion, we decided to use the endovascular technique to obliterate the fistula. 5F guiding catheter was placed in the left vertebral artery. An Echelon 10 microcatheter over Transcend 014 microwire was taken selectively into the distal arterial feeder and position of the fistula was confirmed. Subsequently, multiple microcoils were placed to reduce the flow to the fistula. Postcoiling angiogram showed complete obliteration of the fistula [Figure 3]. To prevent venous thrombosis, heparin was given for 3 days post procedure. The periprocedural course was uneventful and the patient was discharged on the 5th day post operation. On 1 month and 3 months clinical follow-up, his neurological examination was normal with no clinical complaints. One year control angiogram showed stable occlusion of the fistula with coil mass.

- Case 1: 14-year-old male presented with severe headache diagnosed with infratentorial pial arteriovenous fistula. Computed tomography scans of the brain (a and b) show intraventricular hemorrhage in lateral ventricles (arrows point to the bleed) and (c and d) show intraventricular hemorrhage in the third and fourth ventricles (arrows) with diffuse subarachnoid hemorrhage.
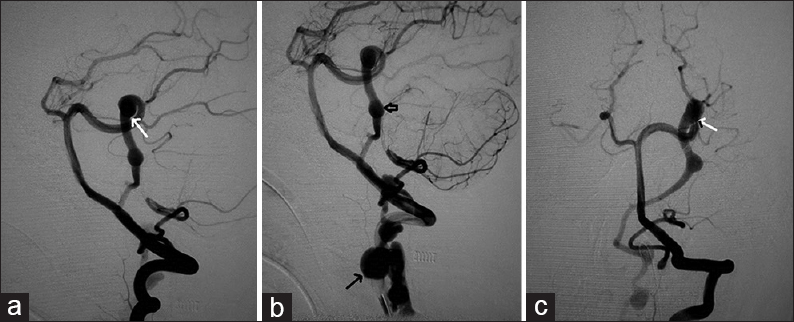
- Case 1: 14-year-old male who presented with severe headache diagnosed with infratentorial pial arteriovenous fistula. (a-c) Digital subtraction angiogram of left vertebral artery shows the pial fistula supplied from left posterior cerebral artery feeder (arrows point to the feeder) draining into venous varix (short arrow) and vertebral venous plexus (arrow in b).
- Case 1: 14-year-old male who presented with severe headache diagnosed with infratentorial pial arteriovenous fistula underwent treatment by endovascular method. Postprocedure vertebral angiogram (a and b) shows complete obliteration of the fistulous component with normal PCA (arrows) and (c) shows the coil mass in situ (arrows).
Case 2
A 15-year-old male child presented with severe headache and brief loss of consciousness. CT scan of brain showed left frontoparietal bleed [Figure 4a] and subsequent CT angiogram showed entangled blood vessels fed by left anterior cerebral artery with venous varices [Figure 4b and c]. DSA revealed high flow pial fistula supplied by the anterior cerebral artery with tortuous enlarged venous varices draining into the superior sagittal sinus [Figure 5a]. Because of the high flow fistula and presence of large venous varices, endovascular route was selected for obliteration of the fistula. The fistula was accessed with Echelon 10 microcatheter and Transcend 014 microwire combination, and subsequently, multiple coils were used to obliterate the fistulous communication [Figure 5b]. Heparin was given for 3 days to prevent venous thrombosis. The procedure was uneventful and the patient was discharged on the seventh postoperative day. On 1 month and 3 months clinical follow-up, the patient did not have any neurological deficit. One year control angiogram showed total obliteration of the fistula [Figure 5c].
- Case 2: 15-year-old boy presented with headache and loss of consciousness diagnosed with pial arteriovenous fistula. Computed tomography scan of the brain (a and b) shows the left parietal bleed (arrow points to the bleed and edema). (c) Computed tomography angiogram shows entangled vessels (arrows) from anterior cerebral artery.
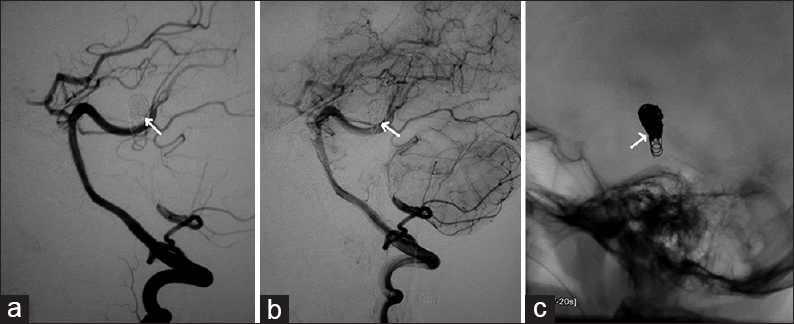
- Case 2: 15-year-old male with left parietal bleed diagnosed with pial arteriovenous fistula. (a) Digital subtraction angiogram of left internal carotid artery in lateral view shows the pial arteriovenous fistula with feeder (arrow) from the anterior cerebral artery and an enlarged venous varix. (b) Post-coiling angiogram shows exclusion of the fistula from the circulation (arrow points to the coil mass). (c) Control angiogram shows stable occlusion of the fistula on oblique view (arrow).
Case 3
A 12-year-old male child presented with generalized tonic-clonic seizures. CT scan brain [Figure 6] showed a large hematoma in the right temporal parietal region with surrounding edema and mass effect. DSA showed a fistulous communication from the middle cerebral artery feeder draining into the Sylvian vein with no intervening nidus [Figure 7]. Because of the presence of the hematoma and mass effect, surgical option was preferred as a better treatment over the endovascular option. Through the right pterional approach, after the evacuation of the hematoma, the feeder was located at the site of fistulous communication and a surgical clip was placed disconnecting the fistulous connection. Postprocedure CT scan showed totally resolved hematoma with the clip at the fistulous site [Figure 8]. His post procedural course was uneventful and he was discharged on the seventh postoperative day. On 1 month and 3 months clinical follow-up, the patient did not have any neurological deficit. Follow-up DSA was done to document total obliteration of the fistula [Figure 9].
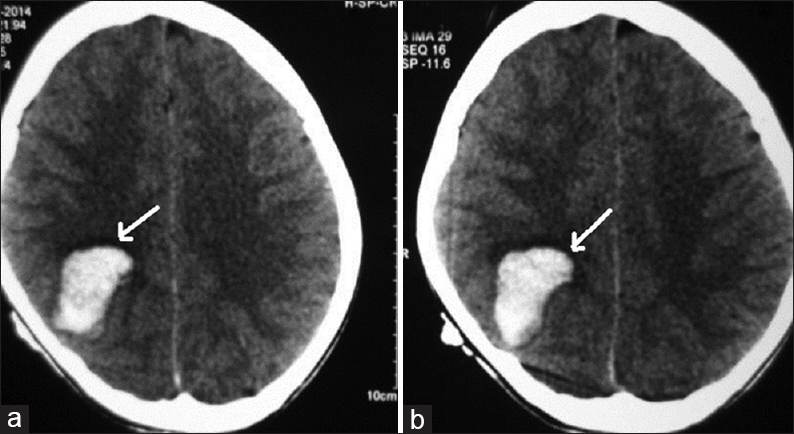
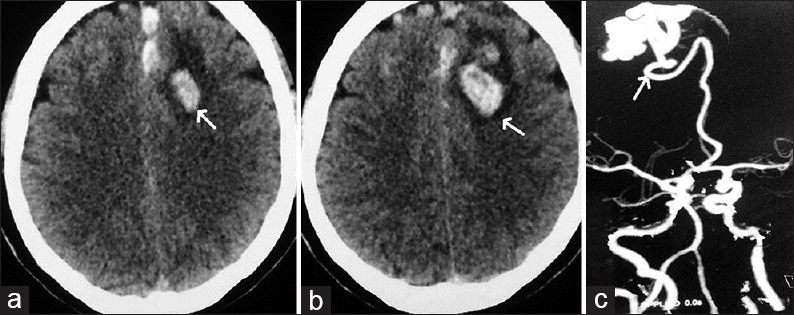
- Case 3: 12-year-old male child presented with generalized tonic-clonic seizures diagnosed with pial arteriovenous fistula. (a and b) Computed tomography scan brain shows a large hematoma in the right temporoparietal region (arrows).
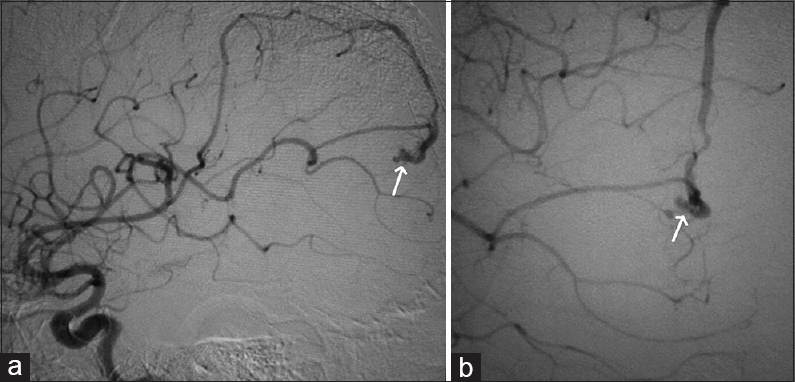
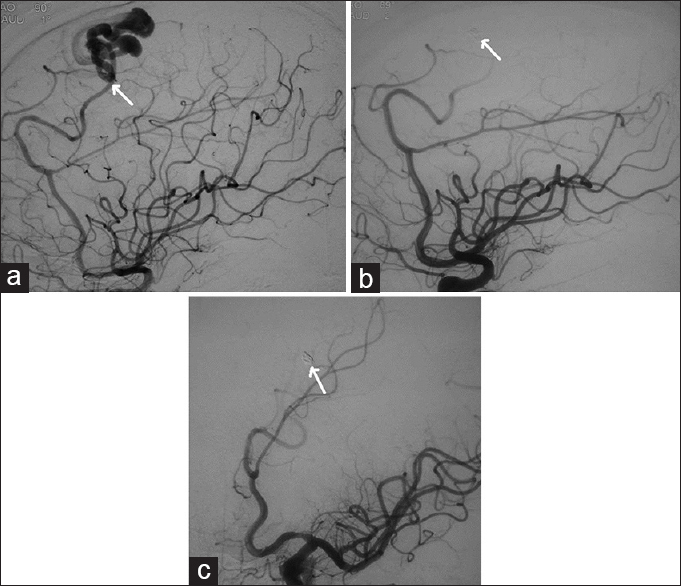
- Case 3: 12-year-old male child who presented with generalized tonic-clonic seizures was diagnosed with pial arteriovenous fistula and right temporoparietal hematoma. (a and b) Digital subtraction angiogram of right internal carotid artery demonstrate the feeder from the middle cerebral artery draining into the vein (arrows point to the feeding artery and vein).
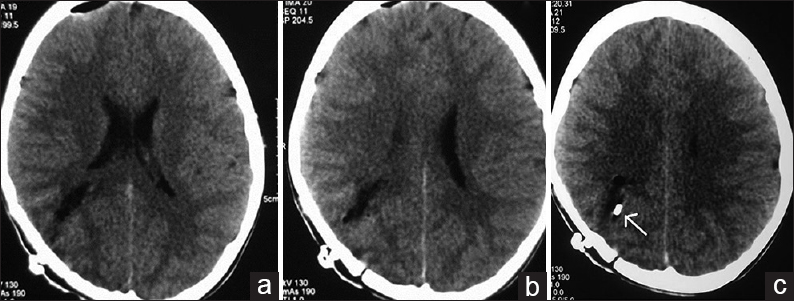
- Case 3: 12-year-old male child who presented with seizures was diagnosed with pial arteriovenous fistula underwent surgical ligation. (a and b) Postoperative computed tomography scan brain show complete resolution of the hematoma. (c) The scan shows the ligation (arrow).
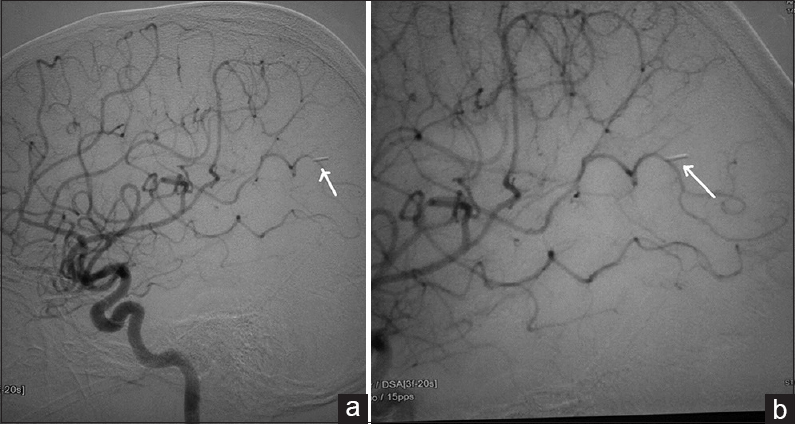
- Case 3: 12-year-old male child diagnosed who presented with seizures was diagnosed with pial arteriovenous fistula underwent surgical ligation. Postoperative angiogram (a) shows the obliteration of fistula (arrow). (b) The position of clip (arrow).
DISCUSSION
Pial AVF accounts for 1.6% of intracranial vascular malformations and has a poor natural history.[13] The underlying pathophysiology of this entity remains unclear and various theories have been proposed. Abnormal angiogenesis is one of the key factors in the formation of pial AVF. Congenital Pial AVF may have syndromic association with Rendu-Osler-Weber disease and Klippel-Trenaunay-Weber syndrome.[4] Pial AVF can be caused by cortical venous thrombosis or hereditary hemorrhagic telangiectasia.[5] The clinical presentation of these patients varies, usually because of increased intracranial pressure and may cause headache, seizures, intracranial hemorrhage, and neurological deficits. We did not find any other associating anomalies in our cases. On cross-sectional imaging, dilated vessels may be seen on the surface of the brain. In some cases, asymmetric dilatation of the pial feeding artery may be seen at the level of the circle of Willis. They differ from dural AVFs, in that they derive their arterial supply from pial or cortical arteries and are not located within the dura mater. In case of dural AVF, dilated venous pouches are usually seen outside the brain parenchyma. If untreated, these fistulae are associated with 63% mortality.[25] Hence, early treatment either by surgical or endovascular route to obliterate these AVF should be done to prevent life-threatening intracranial bleed and death.
Hoh et al., demonstrated that disconnection of the shunt without resection of the malformation was sufficient to achieve the closure of the fistulous communication and treat the disease.[6] Venous varices are associated with high turbulent flow because of arteriovenous shunting. Endovascular treatment is a safe and simple option; however, it may be difficult in cases of multiple arterial connections. Careful analysis of the angiogram is essential because if embolization is too proximal, new arterial supply may be recruited. Endovascular treatment can be implemented in stages to reduce the risk for postoperative hemorrhage by preventing normal perfusion pressure breakthrough in high-flow lesions. Venous varices were seen in two of our cases with high flow fistula in whom we used the endovascular technique to obliterate the fistula. All our cases had single feeder supply to the fistulous communication. Surgical treatment could be difficult in cases of deep-seated, critical areas, and small AVF. Pial AVF can be present co-existing with AVM or any other intracranial vascular lesions.[7]
Yang et al., in their review of literature on pial AVF described an obliteration rate of 86.5% with endovascular treatment and 96.8% with surgical treatment.[8] Residual fistula after endovascular therapy can be treated by surgery. Periprocedural complications associated with the treatment of these lesions either by surgical or endovascular methods include venous thrombosis and intracerebral hemorrhage due to abrupt closure, altered hemodynamics interfering with the normal cerebral auto regulation.[910]
Hemodynamic alteration may result in edema, infarct, and bleed after resection. In our patients, we did not encounter any periprocedural complications. It is important to medicate these patients with heparin after treatment to prevent thrombosis. In our first two patients, we used heparin for 3 days maintaining ACT >250 to prevent the thrombosis. In our last case treated by surgery, we did not give heparin.
Endovascular technique is challenging because of high flow and large size of the afferent and efferent vessels.[10] Various authors describe the use of agents such as onyx and glue in treatment of these lesions. Inadvertent migration of these agents in these high flow lesions into the veins may result in immediate hemorrhage by venous outflow obstruction.
CONCLUSION
Pial AVF is a rare cerebral vascular lesion. Because of the high flow and poor prognosis, exclusion needs to be done at the earliest. The treatment strategy in each individual case has to be made by the team of neurosurgeons and neurointerventionists to identify the best effective method based on patient-specific presentation and angio-architecture.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/2/175083
REFERENCES
- Transarterial occlusion of solitary intracerebral arteriovenous fistulas. AJNR Am J Neuroradiol. 1989;10:747-52.
- [Google Scholar]
- Endovascular embolization of congenital intracranial pial arteriovenous fistulas. Neuroimaging Clin N Am. 1992;2:309-17.
- [Google Scholar]
- Giant aneurysm arising from a single arteriovenous fistula in a child. Case report. J Neurosurg. 1984;60:1085-8.
- [Google Scholar]
- Cerebral arteriovenous fistula in the Klippel-Trenaunay-Weber syndrome. Dev Med Child Neurol. 1988;30:245-8.
- [Google Scholar]
- Endovascular treatment of brain arteriovenous fistulas. AJNR Am J Neuroradiol. 2009;30:851-6.
- [Google Scholar]
- Surgical and endovascular flow disconnection of intracranial pial single-channel arteriovenous fistulae. Neurosurgery. 2001;49:1351-63.
- [Google Scholar]
- Angiographic architecture of intracranial vascular malformations and fistulas - Pretherapeutic aspects. Neurosurg Rev. 1986;9:253-63.
- [Google Scholar]
- Cerebral arteriovenous malformations: Considerations for and experience with surgical treatment in 166 cases. Clin Neurosurg. 1979;26:145-208.
- [Google Scholar]
- Pial arteriovenous fistula in children as presenting manifestation of Rendu-Osler-Weber disease. Neuroradiology. 1995;37:60-4.
- [Google Scholar]






