Translate this page into:
Congenital Absence of the Right Pericardium: Embryology and Imaging
Address for correspondence: Dr. Chi Wan Koo, Department of Radiology, Mayo Clinic, 200, 1st Street Southwest, Rochester - 55905, Minnesota, United States. E-mail: koo.chiwan@mayo.edu
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Though congenital pericardial absence is often asymptomatic, complications can be life threatening. To date, few short case reports, primarily from the pre-CT and MR era, describe congenital absence of the right pericardium. We present a more comprehensive discussion of the embryologic derangements causing such defects and offer an up-to-date review of characteristic radiologic findings. Recognition of characteristic imaging findings of congenital pericardial absence is crucial in guiding diagnosis and management.
Keywords
Absence
congenital
embryology
imaging
pericardium
right pericardium
INTRODUCTION

Isolated congenital pericardial absence is rare with a reported frequency of 0.01–0.04%.[1] Cases of complete bilateral pericardial absence, partial left pericardial absence, and right pericardial absence comprise one-quarter of cases, with complete left pericardial absence accounting for the remainder. Right pericardial absence is extremely uncommon, with fewer than 30 cases reported.[1] The prevalence of these entities may be underestimated due to a lack of standardized diagnostic criteria.
Many cases of congenital pericardial absence are incidentally detected.[2] However, herniation and strangulation of the heart or lungs through the pericardial defect can be life threatening. To date, majority of literature focuses on the more prevalent left-sided absence. Our aim is to discuss the embryology of right pericardial absence and review its characteristic radiologic findings.
Embryology
The pericardium is composed of avascular outer fibrous and inner serous layers of mesothelial cells. The heart invaginates the serous layer as it grows, forming the continuous visceral and parietal layers with the visceral layer lining the heart and the parietal layer lining the outer fibrous pericardium. The potential space between the visceral and parietal layers is filled with fluid, reducing friction when the heart beats. In addition to the heart, the pericardium also encases the pulmonary arterial trunk, proximal ascending aorta, and portions of the left pulmonary veins, protecting these structures from pathologic processes. Moreover, the pericardium serves to fix the heart in position.[3]
At the end of the 4th week of embryo development,[2] pleuropericardial folds develop and grow medially toward the midline [Figure 1]. By the end of the 5th week, the pleuropericardial folds fuse, partitioning the thorax and two partially formed pleural cavities.
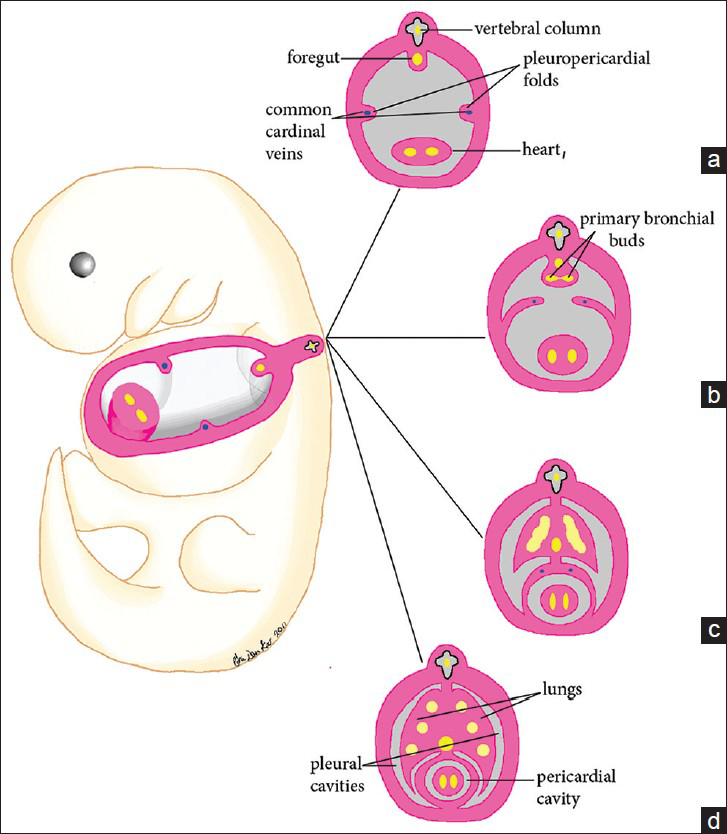
- Illustration of normal pericardial development from the fourth to sixth week of gestation. (a) At 4 weeks of gestation, the laterally positioned pleuropericardial folds are developed. (b) During the 5th week of gestation, the pleuropericardial folds grow toward the midline while the root of each fold migrates ventrally. (c) At the end of the 5th week, the pleuropericardial folds fuse, partitioning the thoracic cavity into a pericardial cavity and two partially formed pleural cavities. Note that union of the pleuropericardial folds and the root of the lungs also occurs during this time. (d) The lungs continue to extend anteriorly to the front of the heart.
Congenital defects of the pericardium are generally considered to be a consequence of failure of pleuropericardial membrane closure during development. One theory hypothesizes that the developing heart stretches the enveloping pericardium during development. If the pleuropericardial membranes fail to fuse before the heart enlarges, a pericardial defect is generated, with the defect size depending upon the differing growth rates of the heart and the pericardial membrane. An alternative theory proposes that a traction induced tear may develop in the pleuropericardial membrane, with a resultant pericardial defect. Kaneko et al.'s case of pericardial absence associated with a divided phrenic nerve,[4] passing both ventrally and dorsally to the defect, illustrates this conjecture. A third theory attributes the developmental failure of the pericardial membrane to premature atrophy of the cardinal veins (duct of Cuvier) supplying the pleuropericardial folds.[5] This theory explains the rarity of right pericardial defect as the right cardinal vein remains as the superior vena cava. Potentially, a certain percentage of pericardial defects are attributable to each of the above mechanisms.
IMAGING
Radiography
Right-sided pericardial defects may manifest radiographically as characteristic cardiac silhouette abnormalities due to herniation of pulmonary and cardiovascular structures through the defect.[26] If the superior aspect of the right pericardium is absent, the interposed lung between the right pulmonary artery and the aorta produces a relative lucency with right pulmonary artery margin prominence [Figures 2a and b]. If the inferior aspect of the right pericardium is absent, the interposed lung may create lucency between the right inferior cardiac border and the right hemidiaphragm.[7] In instances of cardiac tissue herniation through a right pericardial defect, a prominent bulge of the right heart border may be seen [Figure 3a].[2]
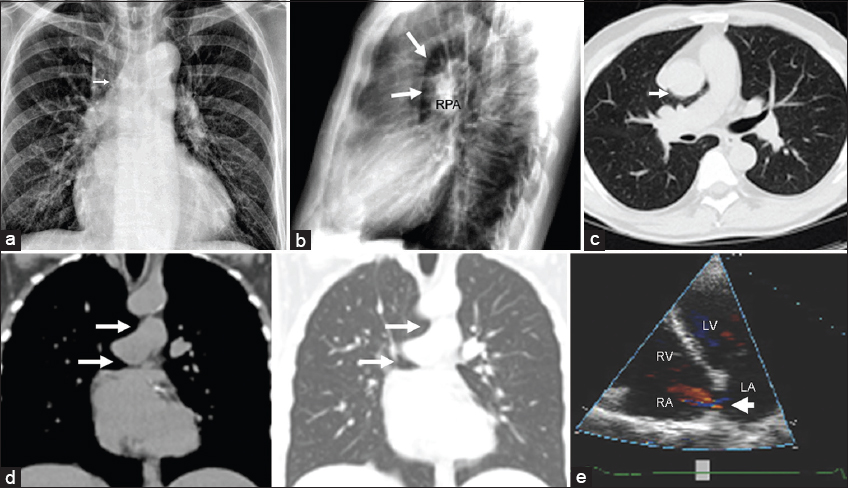
- 77-year-old man with dyspnea. (a) PA chest radiograph reveals a slightly more horizontal ascending aorta due to partial herniation of the ascending thoracic aorta through a pericardial defect (arrow). (b) Lateral radiograph demonstrates abnormal lucency between the ascending aorta and the right pulmonary artery (arrows) resulting in an unusually dense-appearing and prominent right pulmonary artery (RPA). (c) Transaxial CT imaging with lung window confirms this lucency which represents the lung interposed between the aorta and the pulmonary artery through a partially absent right pericardium (arrow), producing the characteristic “retroaortic air” sign. (d) Coronal reformatted images with soft tissue and lung windows demonstrate air insinuating beneath both the aorta and the pulmonary artery (arrows). The right lung resembles the head and body of an open-mouthed dolphin. (e) Right pericardial absence has been associated with other congenital cardiac defects. Echocardiography demonstrates a secundum type atrial septal defect with left to right shunting (arrow).
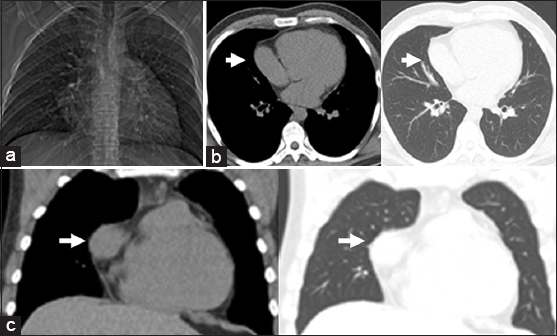
- 26-year-old man evaluated following an environmental exposure. (a) Scout view demonstrates a bulging right heart border. (b) Transaxial and (c) Coronal CT images confirm partial right pericardial absence. Part of the right atrial appendage extends laterally through the partial defect (arrow).
Computed tomography
Computed tomography techniques, especially improved high spatial resolution imaging and electrocardiogram (ECG) gating, can delineate the pericardium and demonstrate its absence. Characteristic CT findings of right pericardial absence include visualizing herniation of right heart structures, such as the right atrial appendage [Figure 3b], through a defect in the pericardium. Interposed lung parenchyma herniating through the defect can also be seen [Figure 2c], producing “retroaortic air” anterior to the pulmonary artery in a characteristic configuration.
In the era prior to cross-sectional imaging, diagnostic pneumothorax or diagnostic thoracotomy was recommended to confirm suspected cases of pericardial absence.[78] With advances in imaging techniques, cross-sectional imaging can now confirm suspicion of pericardial absence raised on radiographs, obviating invasive diagnostic procedures as illustrated in Figures 2d and 3c.
Magnetic resonance imaging
Cardiac magnetic resonance imaging (CMR) is becoming the modality of choice for establishing congenital pericardial absence.[8] CMR can demonstrate pericardial defects by the absence of the preaortic pericardial recess or the pericardium itself.[9] In addition to revealing the actual pericardial defect, functional CMR can demonstrate excessive cardiac displacement, exaggerated cardiac volume differences between diastole and systole, and associated congenital cardiac anomalies. Thirty percent of patients with congenital pericardial absence have associated cardiac defects, with atrial septal defect, bicuspid aortic valve, patent ductus arteriosus, and tetralogy of Fallot being the most common anomalies. Pericardial defects may also be present with other syndromes.[9]
Echocardiography
Pericardial defects are not typically directly visualized with echocardiography. Atypical orientation and position of the heart within the chest as well as abnormal wall motion are suggestive of the diagnosis. Echocardiography is also useful in detecting associated congenital cardiac anomalies [Figure 2e].
Clinical consequences
Generally, individuals with pericardial absence are asymptomatic. Symptoms typically result from mechanical complications arising from smaller defects. Herniation of cardiac structures can lead to strangulation.[89] Paroxysmal non-exertional chest pain is the most commonly described symptom,[10] thought to be related to herniation of the right atrial appendage,[2] right atrium, and/or ventricle.[8]
In addition to herniation-related ischemia, other symptoms such as dyspnea and syncope may be related to compression of great vessels, traction on pleuropericardial adhesions, or postural distention of vessels. Symptomatic cardiac tissue herniation or strangulation typically requires surgical liberation of the fibrous ring and pericardioplasty. Xenograft pericardioplasty[2] and defect enlargement by direct excision[8] have also been performed to treat symptomatic, partial defects of the right pericardium. The role of prophylactic surgical repair of incidentally detected defects in asymptomatic individuals remains controversial.[9]
CONCLUSION
Advancements in diagnostic imaging will improve detection of congenital pericardial absence. Increased awareness and recognition of characteristic radiologic findings by diagnostic imagers is crucial in guiding management and preventing complications in this population.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2015/5/1/12/152338
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Angina pectoris associated with a pericardial defect. J Thorac Cardiovasc Surg. 1971;61:461-5.
- [Google Scholar]
- CT and MR imaging of pericardial disease. Radiol Clin North Am. 2004;42:587-601. vi
- [Google Scholar]
- Complete left pericardial defect with dual passage of the phrenic nerve: A challenge to the widely accepted embryogenic theory. Pediatr Cardiol. 1998;19:414-7.
- [Google Scholar]
- Congenital pericardial deficiency with other anomalies: Report of a case with necropsy findings. Yale J Biol Med. 1935;8:19-22.
- [Google Scholar]
- Congenital absence of the right pericardium associated with herniation of the right atrium. Mil Med. 1970;135:557-9.
- [Google Scholar]
- Partial right-sided congenital pericardial defect with herniation of right atrium and right ventricle. Chest. 1979;76:484-6.
- [Google Scholar]
- Congenital absence of the pericardium: Case presentation and review of literature. Int J Cardiol. 2005;98:21-5.
- [Google Scholar]
- Isolated congenital absence of the pericardium: Clinical presentation, diagnosis, and management. Ann Thorac Surg. 2000;69:1209-15.
- [Google Scholar]






