Translate this page into:
Biliary Anastomotic Leakage following Orthotopic Liver Transplant: The Use of Primovist (Gadoxetate Disodium) as an Intravenous MR Cholangiography Contrast Agent for Biliary Leakage
Address for correspondence: Dr. Gavin Low, Department of Radiology and Diagnostic Imaging, University of Alberta, 2A2.41 WMC, 8440-112 Street, Edmonton, Alberta T6G 2B7, Canada. E-mail: timgy@yahoo.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
We present a case of postoperative biliary anastomotic leakage demonstrated on intravenous contrast-enhanced MRI performed with Primovist (Gadoxetate Disodium). Primovist has been widely used for its combined extracellular and hepatocyte-specific properties in the characterization of liver lesions. Another recognized, but less widely utilized property of Primovist is its ability to provide morphologic and functional assessment of the biliary system. In our case, Primovist-enhanced MR imaging demonstrated the presence and location of an active biliary leakage which was associated with free spillage into the peritoneum.
Keywords
Bile leak
liver transplantation
magnetic resonance imaging
primovist
INTRODUCTION

Bile leaks are a recognized complication of liver transplantation (commonly due to hepatic arterial thrombosis/stenosis or inadequate surgical technique) and may manifest as newly developing intra-abdominal free fluid or collections. However, the presence of fluid or collections is non-specific in the early postoperative period, as these may represent surgical trauma including hematomas or seromas. Primovist (Gadoxetate Disodium; Bayer-Schering, Berlin, Germany) is an intravenous magnetic resonance imaging (MRI) contrast agent which demonstrates hepatocyte uptake and biliary excretion. In this case, we highlight the clinical utility of Primovist-enhanced MRI in demonstrating the presence and location of a postoperative bile leak following liver transplantation.
CASE REPORT
A 35-year-old woman underwent orthotopic liver transplantation for autoimmune hepatitis-induced cirrhosis associated with secondary portal hypertension. Her decompensated liver disease had been complicated by portal gastropathy, recurrent ascites with the development of spontaneous bacterial peritonitis, hydrothorax, and secondary bacterial empyema. Transjugular intrahepatic portal systemic (TIPS) shunt had been performed as a “bridge to transplantation.” Liver transplantation was performed with standard venous reconstruction, single artery anastomoses, and biliary reconstruction via retrocolic Roux-en-Y hepaticojejunostomy. Hepaticojejunostomy was preferred to primary duct-to-duct anastomosis due to perceived anastomotic tension at the time of surgery which prevented the latter technique. The procedure was uneventful with two standard surgical drains positioned around the anastomotic site. Although postoperative recovery appeared to be rapid with early extubation, after transfer to the ward, large volume of bile-stained ascites was discovered from the surgical drains and the patient experienced increasing upper abdominal pain, a low-grade fever, and rising serum bilirubin levels.
MRI of the liver was performed according to our institution's liver transplant protocol with dedicated hepatic artery angiography (confirming normal postoperative hepatic arterial caliber and patency) and standard magnetic resonance cholangiography (MRC) sequences to assess the biliary tree. At the discretion of the reporting radiologist, intravenous Primovist (10 ml of 0.025 mmol/kg of body weight) was administered as a contrast agent with delayed images obtained at 10 min and 45 min following injection. On pre-contrast imaging, a low T1 and high T2 signal fluid collection was present in the left perihepatic space indicating a biloma [Figure 1]. The biliary anastomosis was well identified between the common hepatic duct and the jejunal loop of the Roux-en-Y hepaticojejunostomy. Following Primovist administration, a high-grade bile leak was confirmed at the surgical anastomosis with extravasation of high T1 signal bile from the biliary tree, bypassing the jejunal loop, pooling within the perihepatic space biloma, left subphrenic space, and extending down the left paracolic gutter into the left side of the pelvis [Figures 2 and 3].
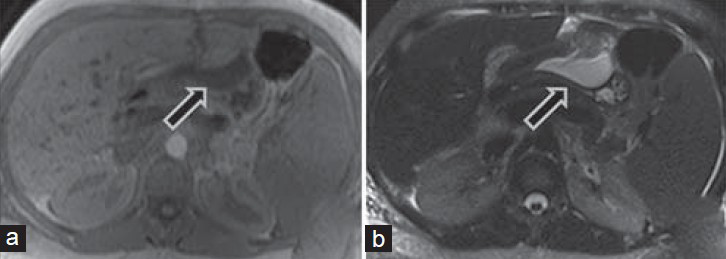
- 35-year-old woman underwent orthotopic liver transplantation for autoimmune hepatitis-induced cirrhosis associated with secondary portal hypertension and was diagnosed with postoperative biliary anastomotic leakage. Corresponding axial (a) T1-weighted and (b) T2-weighted sequences of the upper abdomen show left perihepatic fluid collection (arrow) with a low T1 signal and high T2 signal.
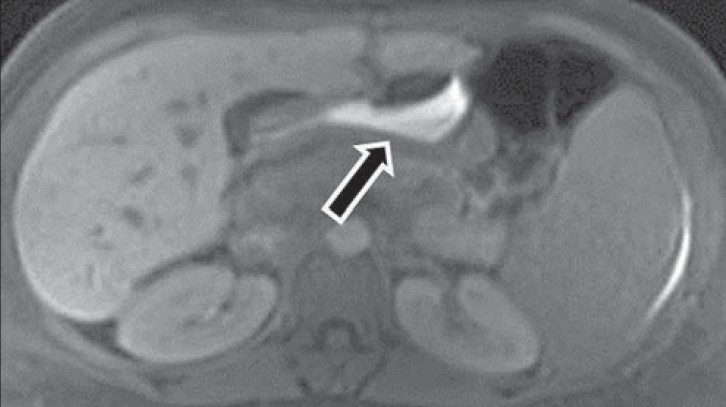
- 35-year-old woman underwent orthotopic liver transplantation for autoimmune hepatitis-induced cirrhosis associated with secondary portal hypertension and was diagnosed with postoperative biliary anastomotic leakage. Axial T1 image 45 min post-Primovist injection shows high T1 signal bile pooling (arrow) within the left perihepatic space.
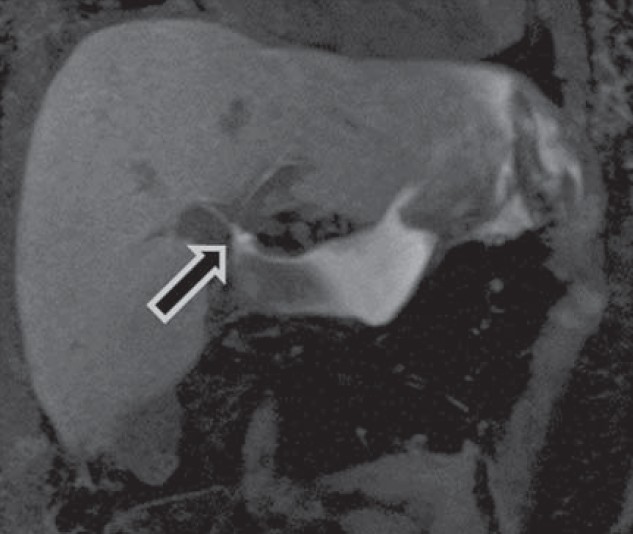
- 35-year old woman underwent orthotopic liver transplantation for autoimmune hepatitis-induced cirrhosis associated with secondary portal hypertension and was diagnosed with postoperative biliary anastomotic leakage. Coronal T1 MIP image 45 min post-Primovist injection shows high T1 signal bile pooling from biliary tree at the anastomotic site (arrow) and within the left perihepatic and subphrenic spaces.
The patient was subsequently taken back to the operating room on the 10th postoperative day where a bile leak was surgically confirmed with disruption of the anterior wall of the anastomosis. A precipitating etiology for the biliary anastomotic leakage could not be identified. The presence of a patent, normal caliber hepatic artery excluded hepatic arterial thrombosis or stenosis as an underlying etiology. Abdominal washout and revision of the hepaticojejunostomy allowed the bile leak to be successfully repaired.
DISCUSSION
The incidence of bile leak following orthotopic liver transplant has been reported to be between 5 and 10%.[1] Clinically, the diagnosis is suggested by the passage of bile through a surgical drain or abdominal scar along with fever, jaundice, and abdominal pain, but debate exists over the best imaging modality to make the diagnosis. Endoscopic retrograde cholangiography (ERC) is regarded as the Gold Standard, although this is an invasive test and not without inherent complications such as hemorrhage, pancreatitis, and infection. Furthermore, ERC was precluded in our case due to altered anatomy from the hepaticojejunostomy formation. Non-invasive investigations such as ultrasound, computed tomography (CT), and MRI (both with and without Gadolinium contrast agents) allow demonstration of indirect evidence of biliary leakage such as perihepatic collections, acute onset of free intraperitoneal fluid, and biliary dilatation, but they lack the specificity to adequately confirm the direct site of leakage.[23]
Primovist-enhanced MR imaging is a powerful non-invasive tool for evaluating the biliary system. Approximately 50% of the injected dose of Primovist is taken up by the hepatocytes and excreted into the biliary system. This permits exquisite opacification of the bile ducts during the hepatospecific delayed phase (approximately 10–120 min post-injection). Furthermore, the images may be displayed on a workstation using 3D-multiplanar reconstructions for optimal interrogation and problem solving. As we have shown, Primovist-enhanced MR imaging provided good visualization of the biliary tree with demonstration of the exact site of bile leakage, and confirmed active extravasation into the peritoneum. These encouraging findings merit more detailed investigation through controlled studies involving larger numbers of patients. Castellanos et al., found that Primovist had 100% accuracy in excluding a bile leak in 23 patients with clinical suspicion for a bile leak.[1] In addition, it should be noted that while 10 min and 45 min delayed images may be sufficient to detect a high-grade bile leak, a longer delay time (e.g. 60–90 min) may be necessary for excluding a low-grade leak.[4]
CONCLUSION
In conclusion, our case highlights the clinical potential of Primovist-enhanced MR imaging as a diagnostic tool for excluding post-transplant bile leaks. As biliary related issues are a frequent complication of liver transplantation, and as MRI is a rapid non-invasive test, we believe that Primovist-enhanced MR imaging represents a highly promising technique available to radiologists that can be used as a substitute for more invasive methods to evaluate this complication.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/75/148271
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Biliary complications after orthotopic liver transplantation: A review of incidence and risk factors. Transplant Proc. 2012;44:1545-9.
- [Google Scholar]
- Advantages of gadobenatedimeglumine-enhanced MR cholangiography in the diagnosis of post-liver transplant bile leakage. Diagn Interv Imaging. 2013;94:443-52.
- [Google Scholar]
- Biliary MR imaging with Gd-EOB-DTPA and its clinical applications. Radiographics. 2009;29:1707-24.
- [Google Scholar]
- Early phase detection of bile leak after hepatobiliary surgery: Value of Gd-EOB-DTPA-enhanced MR cholangiography. Abdom Imaging. 2012;37:795-802.
- [Google Scholar]






