Translate this page into:
Adrenal Lesions: Spectrum of Imaging Findings with Emphasis on Multi-Detector Computed Tomography and Magnetic Resonance Imaging
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The adrenal gland is a common site of a large spectrum of abnormalities like primary tumors, hemorrhage, metastases, and enlargement of the gland from external hormonal stimulation. Most of these lesions represent nonfunctioning adrenal adenomas and thus warrant a conservative management. Multi-detector computed tomography (CT) and magnetic resonance (MR) imaging are still considered highly specific and complementary techniques for the detection and characterization of adrenal abnormalities. Radiologist can establish a definitive diagnosis for most adrenal masses (i.e., carcinoma, hemorrhage) based on imaging alone. Imaging therefore can differentiate malignant lesions from those benign and avoid unnecessary aggressive management of benign lesions. The article gives an overview of the adrenal lesions and their imaging characteristics seen on CT and MR imaging.
Keywords
Adrenal
adrenocortical carcinoma
computed tomography
Adenoma
magnetic resonance imaging
INTRODUCTION

The adrenal glands are a common site for incidentally detected non-hyperfunctioning adenomas.[1] With technological advances in cross-sectional imaging, there is a substantial increase in the number of small adrenal masses that are incidentally detected at computed tomography (CT) and magnetic resonance (MR) imaging.
Cross-sectional imaging readily characterizes benign adrenal masses, such as lipid-rich adenomas, myelolipomas, adrenal cysts, and adrenal hemorrhage as they have characteristic diagnostic imaging features that show the presence of lipid, intralesional fat, water, or blood. A small minority of adrenal masses are not easily characterizable on cross-sectional imaging and often remain indeterminate. These include lipid-poor adenomas, adrenal metastases, adrenal carcinomas, and phaeochromocytomas.
CT and MR imaging are the preferred diagnostic imaging techniques used for the detection and characterization of adrenal lesions with reported specificity of identifying the mass as an adenoma being close to 100%, though the sensitivity is lower.[2] Positron emission tomography (PET) is generally performed in patients with cancer and it yields information about the biochemical processes that may precede gross anatomic changes.[3]
Despite adrenal glands being a relatively frequent site of metastatic disease, even in patients with known malignancy only 26-36% are metastatic with the high risk of contraindications to the radical curative surgery or radiotherapy; in fact most of these lesions represent nonfunctioning adrenal adenomas and thus warrant conservative management.[1]
The main goal of imaging is to firmly establish a reliable diagnosis in order to avoid unnecessary aggressive management of such lesions.[4] We present an overview of CT and MR imaging findings of benign adrenal lesions such as adenoma, myelolipoma, hemorrhage, cysts, and malignant lesions like adrenal lymphoma, pheochromocytoma, adrenal metastasis, and adrenocortical carcinoma.
IMAGING TECHNIQUE
Computed tomography
CT generally represents the primary modality for both detection and characterization of adrenal masses. Multi-detector row CT can produce isotropic data sets while simultaneously delivering exceptional temporal resolution with excellent z-axis coverage. Such data sets can be manipulated to generate high-quality multiplanar reformations that can convey complex anatomic and pathologic information. CT protocol includes unenhanced scan of the upper abdomen to readily detect areas of fat, hemorrhage, or necrosis, followed by administration of contrast medium that provides additional information on lesion characteristics based on its perfusion pattern.[25]
The overlapping venous phase enhancement patterns of adenomas and non-adenomas preclude reliable use of this phase of contrast enhancement for differentiating between adenomas and nonadenomas.[6] For this reason delayed image series is acquired at a variable time of 10-15 minutes to perform adrenal washout analysis. The absolute percentage washout (APW) and relative percentage washout (RPW) can be derived by using a set formula.[146]
Histogram analysis is used to measure the number of negative-attenuation pixels within region of interest (ROI);[1] this method is based on the concept that about 97% of adenomas contain negative pixels and it can be used to differentiate adenomas from non-adenomas showing high specificity (100%) and increased sensitivity when thresholds of more than 10% negative attenuation pixels are used with unenhanced scans.[78]
Magnetic resonance imaging
MR imaging is currently considered an accurate imaging technique for the identification and characterization of adrenal masses. By using chemical shift sequences, MR imaging has sensitivity to demonstrate even small quantities of lipid, due to the unique difference in resonance frequency between protons in water and triglycerides molecules.[49]
Most benign adrenal lesions, such as adenoma and myelolipoma, contain fat and typically exhibit significant signal loss (i.e., appear darker) on out-of-phase (OP) images (2.2 ms, at 1.5 T), due to the cancellation of signal between lipid and water protons within a voxel. By contrast, malignant adrenal masses, such as metastases, primary carcinomas, and pheocromocytomas, usually do not contain fat and thus show no perceivable signal loss on OP images.
Current imaging protocols of MR chemical shift imaging (CSI) allows the acquisition of the in-phase (IP) and OP sequences simultaneously in a single breath hold. The loss of signal intensity can be assessed with both a visual and quantitative analysis.
Adrenal-to-spleen chemical shift ratio (ASR) or the adrenal sensitivity index (SII) are the quantitative methods to measure the signal loss on MR CSI using internal references; they can be derived by the use of specific set formulae.[49]
The liver and the muscle should not be used because of the frequent incidence of hepatic steatosis and the possible muscle fatty infiltration, respectively, which also results in signal loss on the OP sequences. Unfortunately, there still remains a substantial overlap between the signal intensities seen in adenomas and in metastasis limiting its clinical applicability in distinguishing adenomas from malignant masses.
The diffusion of 3T MRI system has made it possible to develop new sequences that can help in distinguishing adenomas from non-adenomas; however, up till now it has not demonstrated a significant contribution with respect to the use of CSI.[10]
NORMAL ANATOMY AND IMAGING APPEARANCE (COMPUTED TOMOGRAPHY AND MAGNETIC RESONANCE IMAGING)
The adrenal glands are situated in the suprarenal position in the retroperitonium. The glands weigh approximately 5.0 g each, on average, and measure approximately 30.0 mm in width, 50.0 mm in length, and up to 10.0 mm in thickness. The right adrenal gland is immediately posterior to the inferior vena cava and lies anterosuperior to the upper pole of the kidney; the left adrenal gland lies posterolateral to the aorta, posterior to the pancreas and it is anteromedial to the upper pole of the kidney. The glands are very vascular and receive arterial supply from the superior, middle, and inferior suprarenal arteries, which are branches of the inferior phrenic arteries, abdominal aorta, and renal arteries, respectively.
The axial view of adrenal glands shows each gland to consist of a body and two limbs, medial and lateral, respectively.
The shape of the glands is determined on the section it is best seen. The right gland in most cases appear slightly linear or has an inverted “V” configuration. The left gland has a triangular, inverted “V” or “Y” configuration.
The typical radiologic appearance of an inverted Y-shaped, V-shaped adrenal depends, in part, on the presence of a kidney in the renal fossa. In fact, when ipsilateral kidney is congenitally absent the adrenal gland will be present in the majority of cases and it does not have the typical splayed limbs showing a longer, slender or disc shape appearance, the so called “lying down adrenal”; this definition has been ascribed to the elongated appearance of the adrenal not normally molded by the adjacent kidney.
Anatomic anomalies of the adrenal gland may occur. Because the development of the adrenals is closely associated with that of the kidneys, either agenesis of an adrenal gland is usually associated with ipsilateral agenesis of the kidney, and fused adrenal glands with a fused kidney.
Normally, the entire adrenal gland is surrounded by a capsule of connective tissue; adreno-hepatic fusion (AHF) is defined as adhesion of the liver and right adrenal cortex and close intermingling of the respective parenchymal cells with partial or complete absence of the fibrous capsule dividing the two organs.
Despite advancements in CT technology that have made it possible to acquire thin data set, lack of the fat plane at CT might not necessarily suggest the presence of AHF since the fat plane between the normal right adrenal gland and the right liver is not always visible at CT.
BENIGN ADRENAL LESIONS
Adrenal adenoma
Adenoma is the most common adrenal lesion and accounts for a large majority of adrenal masses (so called, “incidentalomas”) detected incidentally. Based on function, adenoma may be classified as non-hyperfunctioning (normal hormone levels) or hyperfunctioning (primary hyperaldosteronism, Cushing syndrome, and hyperandrogenism). Lesions are usually ovoid masses smaller than 3 cm with well-defined margins.[4]
Due to presence of fat, adenoma generally shows CT numbers less than 10 HU on unenhanced CT scan [Figure 1a].[2] Unfortunately, in a limited number of cases (10-40%), adenoma contains only a small amount of lipid (i.e., lipid poor adrenal adenoma) and thus may be difficult to differentiate from malignant lesions due to higher attenuation at unenhanced CT scan.
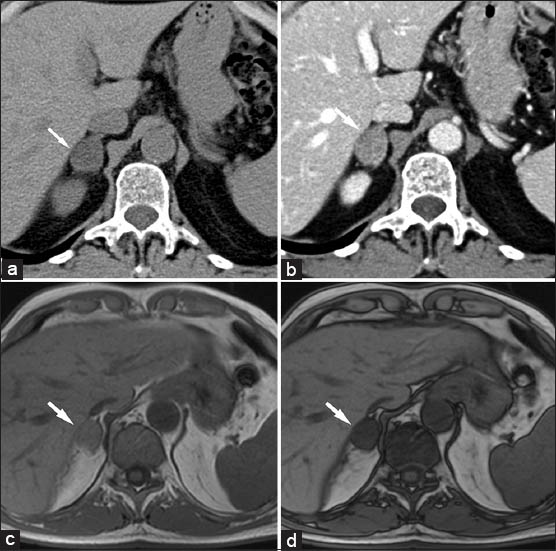
- 75-year-old male without history of malignancy with incidentally discovered adrenal adenoma. (a) Transverse nonenhanced computed tomography (CT) scan shows well-defined mass (arrow) with low attenuation to the liver parenchyma. (b) Dynamic contrast-enhanced CT scans obtained at the same level. Adenoma (arrow) exhibits rapid and homogeneous enhancement. (c) Corresponding transverse in-phase (160/4.9) and (d) Out-of-phase (160/2.5) T1-weighted gradient-echo magnetic resonance images show the, Lesion (arrow) with significant signal drop-off on out-of-phase magnetic resonance image due to the high lipid content which is diagnostic of adenoma.
When performing an histogram analysis, threshold of more than 10% negative attenuation pixels is recommended to obtain increased sensitivity (specificity 100%; sensitivity between 70% and 91%).[18]
Unlike most malignant adrenal lesions, adenoma typically shows rapid and intense enhancement after contrast administration [Figure 1b], followed by significant wash-out (APW >60%, RPW >40%) on delayed images obtained with either CT or MR imaging.[15]
Signal loss on OP MR images [Figure 1c and d] is the key imaging finding of adrenal adenoma.[49]
Adrenal adenomas have an ASR of 70 or less and the SII is >16.5%.[4]
Rounded foci of altered signal intensity may be seen within an adenoma owing to cystic changes, variation in vascularity or hemorrhage; the appearance of blood products at MR imaging varies with their stage of evolution.[11]
Adrenal myelolipoma
Myelolipoma is a rare incidentally discovered benign tumor composed of mature adipose tissue and hematopoietic elements. In a CT scan detection of a fatty component (-30 to -90 HU) within an adrenal mass [Figure 2a] is virtually diagnostic of myelolipoma.[12] After contrast administration, myelolipoma typically show bright enhancement of the highly vascularized myeloid component. At MR imaging, diagnosis typically relies on significant signal loss on frequency-selective fat-saturated (T2-weighted) images [Figure 2b and c].
- 60-year-old male with incidentally discovered right adrenal myelolipoma. (a) Transverse non-enhanced computer tomography scan shows well-defined mass (arrow) with heterogeneous density due to the presence of focal areas of fat attenuation (arrowhead). (b and c) Corresponding transverse T1-weighted gradient-echo magnetic resonance images (160/4.9) (b) without and (c) with fat-suppression. The fatty component of the myelolipoma (arrowhead) shows a decrease of signal intensity on fat-suppressed image.
Larger lesions may occasionally be difficult to differentiate from retroperitoneal lipomatous tumors, such as retroperitoneal liposarcoma [Figure 3].
- 60-year-old male with atypical left adrenal myelolipoma simulating retroperitoneal liposarcoma. Transverse contrast-enhanced CT scan shows a large predominantly fat-containing lesion (arrows) originating from the medial and lateral arms of the gland.
Adrenal hemorrhage
Adrenal hemorrhage is a common complication of a difficult delivery or meningococcal septicaemia during the neonatal period. In adults, adrenal hemorrhage is infrequent and may be caused by different etiologies, including: Abdominal blunt trauma, adrenal vein thrombosis, anticoagulation therapy, and stress. During the acute phase, hematoma causes an adrenal enlargement with increased attenuation on nonenhanced CT (50-90 HU). Over a period of time, hematoma progressively reduces in size and attenuation decreases, until a water-density collection may persist in the more advanced stages.
An adrenal hematoma may calcify after 1 year. A chronic organized hematoma demonstrating hypodense fluid attenuation and a thin peripheral rim is considered a pseudocyst.[4]
At MR imaging, signal intensity significantly varies according to the age of hematoma (acute <7 days, subacute 1-7 weeks, or chronic after 7 weeks).[413]
Adrenal cyst
Adrenal cyst is an uncommon incidentally discovered lesion. Based on different etiologies, four types of cysts have been identified: Endothelial cyst (hemangioma or lymphangioma), epithelial cyst, pseudocyst, and parasitic cyst (hydatid disease). On CT examination, simple cyst generally appears as a round well-defined nonenhancing water-density adrenal mass with no soft-tissue component and no internal enhancement. On MR imaging, simple cystic lesions are generally hypointense on T1-weighted images and highly hyperintense on T2-weighted images. Sometimes, cysts may be complex (i.e., hemorrhage), thus showing atypical imaging appearances.
Pseudocysts
Adrenal pseudocysts are unilocular or multilocular cystic lesions arising within the adrenal gland, surrounded by a fibrous tissue wall lacking a recognizable lining layer. The etiology is not clear, but adrenal pseudocysts are believed to arise from organization of a prior hemorrhagic or infectious process. On MR images, a complicated pattern may be seen with evidence of septations, blood products, or a soft-tissue component secondary to hemorrhage or hyalinized thrombus.
Peripheral curvilinear calcification may be present, however, it is difficult to appreciate it at imaging.
Adrenal schwannoma
Adrenal schwannomas are benign nerve sheath tumors thought to arise from Schwann cells associated with phrenic nerve, the vagus nerve, and the sympathetic trunk innervating the glands.
CT appearence of schwannoma typically consist in a well-circumscribed, homogenous mass; its long-standing schwannoma may have heterogeneous aspects due to the presence of cystic degeneration and calcifications. On MRI, the schwannoma shows low signal intensity on T1-weighted images and heterogeneous high signal intensity on T2-weighted images. Contrast enhancement characteristics are not specific since they may have variable homogenous or heterogeneous enhancement.
Adrenal hemangioma
Hemangioma of the adrenal gland is a nonfunctional benign tumor, quite infrequent and generally asymptomatic. It affects people between the ages of 50 and 70 years; women are affected twice as often as men. At microscopic analysis, hemangiomas are usually cavernous and rarely capillary. The tumor is well delimited and encapsulated. Multiple cavities are seen at the periphery, whereas complex central modifications involve necrosis, calcification, fibrosis, thrombosis, and hemorrhage. There are no characteristic signs on radiological studies and the majority of adrenal hemangiomas are diagnosed postoperatively also because of the low frequency and the lack of specific symptoms. However, the evidence at postcontrast CT acquisition of a mass with heterogeneous enhanced areas in a centripetal pattern can suggest the presence of hemangioma. Nonenhanced CT scans generally reveal a hypoattenuating mass, varying in size from a few centimeters to as large as 25 cm3. The larger masses frequently have areas of calcification representing dystrophic calcification or phlebolits.
On T1-weighted MR images it shows heterogeneous low signal; central intrinsic hyperintensity may be present due to hemorrhage. On T2-weighted MR images hemangioma shows marked high signal intensity except in the central fibrotic areas. After intravenous contrast administration they enhance similarly to hemangiomas elsewhere and similar enhancement patterns on both CT and MRI are seen.
Adrenal ganglioneuroma
Adrenal ganglioneuromas are rare benign tumors originating from the neural crest tissue of the sympathetic nervous system. Ganglioneuromas are generally asymptomatic even if they reach a large size. The tumor appear as well-defined masses that are oval, crescentic, or lobulated. Whereas the tumor tend to surround major blood vessels at CT it is possible to demonstrate this relationship. Calcifications may be present; no significant enhancement is usually seen.
Adrenal infections
Adrenal infections are an important but under-recognized clinical entity. The adrenal gland can be infected by a myriad of pathogens that may directly or indirectly cause tissue damage and alteration in endocrine function. Pathogens as tuberculosis, histoplasmosis, and blastomycosis may involve the adrenal gland leading to adrenal insufficiency in case of their extensive involvement. In the case of adrenal infection the patient outcome may be significantly improved by early recognition and intervention. Typically, these infections result in bilateral enlargement of the adrenal glands. In chronic cases, the glands may atrophy and calcify. Granulomatous infections may leave small, faint, diffuse calcifications within a normal-sized or atrophic gland. The adrenal glands are the fifth most common site of extrapulmonary tuberculosis after the liver, spleen, kidneys, and bones. In patients with tuberculosis an Addisonian type clinical picture may be observed. The CT appearance of adrenal involvement is characterized by the evidence of enlarged glands associated with large, hypoattenuating necrotic areas, with or without dot-like calcification
MALIGNANT ADRENAL LESIONS
Adrenal metastases
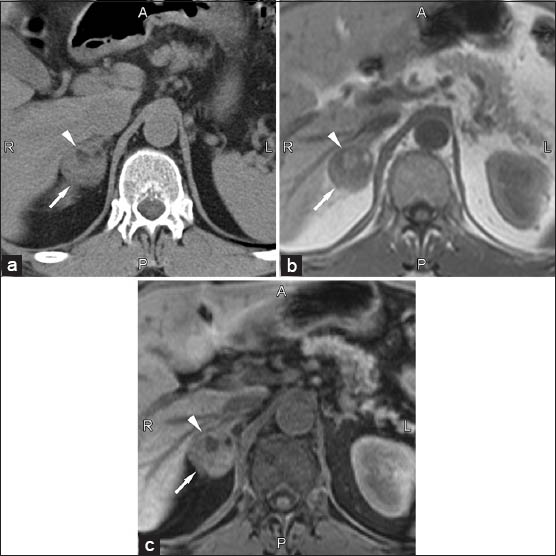
Adrenals are a common site of metastatic dissemination from other primary cancer sites. Most lesions show lobulated and irregular margins, with heterogeneous density due to areas of necrosis and hemorrhage. On MR imaging, the key imaging findings are absence of signal drop off on OP MR images and hyperintensity relative to the liver and spleen on T2-weighted images [Figure 4a and b].[8] After contrast agent administration, lesions typically show heterogeneous and prolonged enhancement on CT and MR imaging [Figure 4c].[46]
- 50-year-old female with lung cancer with typical right adrenal metastases. (a) Transverse in-phase (160/4.9) and (b) Out-of-phase (160/2.5) T1-weighted gradient-echo magnetic resonance images show the lesion (arrow) with no signal loss on out-of-phase images. (c) Transverse T1-weighted fat saturated contrast-enhanced magnetic resonance image (4.5/1.9) obtained at the same level show the lesion (arrow) has marked and heterogeneous enhancement due to a central area of necrosis (arrowhead).
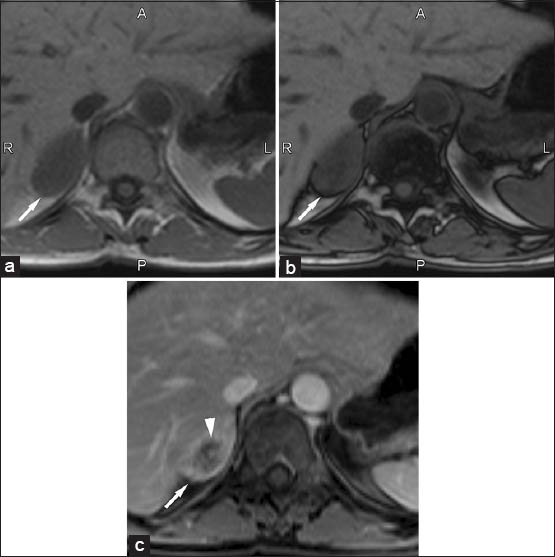
They also show slower washout at delayed imaging (APW <60%, RPW <40%) than adenomas. Unfortunately, not all metastatic lesions show typical imaging findings [Figure 5a and b], and, thus, CT-guided percutaneous biopsy is still necessary for undetermined cases.
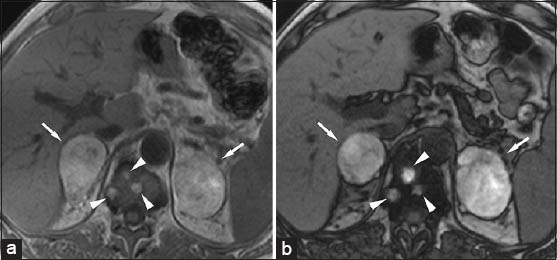
- 46-year-old male with melanoma presenting with adrenal insufficiency with bilateral adrenal metastases. (a) Transverse in-phase (160/4.9) and (b) outof-phase (160/2.5) T1-weighted gradient-echo magnetic resonance images-due to the high content of melanin, the lesion (arrow) shows high signal intensity on in-phase image that may simulate a high lipid content mass. However, no signal loss is observed on out-of-phase image. Note multiple metastases into the vertebral body (arrowheads).
Adrenocortical carcinoma
Adrenal carcinoma is a rare tumor, with a higher prevalence in the female population.[11]
Functional tumors were more common in females (7:3), whereas nonfunctional tumors were more often found in males (3:2). Nonfunctional tumors (estimated in about 40%) are seen with much greater frequency in older patients, regardless of sex.
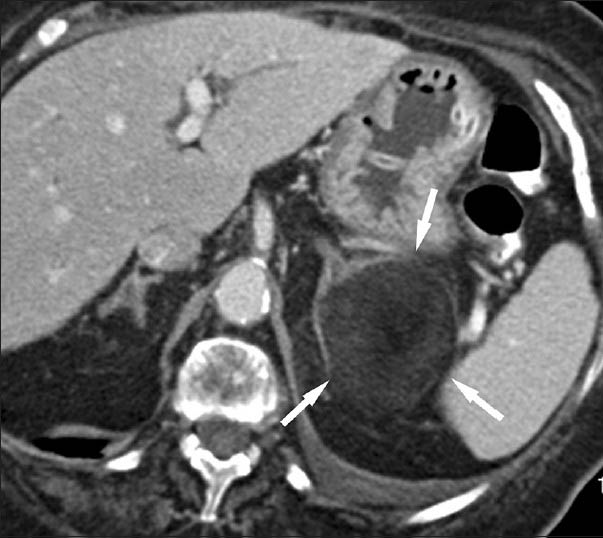
Most lesions are discovered as large palpable masses at physical examination, or at cross-sectional examinations performed for other purposes. The mean tumor size at diagnosis is 13 cm (range 3-28 cm). Adrenal carcinoma is readily detected on CT or MR imaging as large, solid, irregularly shaped mass. Due to the presence of necrosis, calcifications, and hemorrhage, lesions typically show heterogeneous density before and after contrast medium injection [Figure 6]. Vascular invasion of the renal vein and inferior vena cava, as well as metastatic dissemination to the liver, lung, and retroperitoneal lymph-nodes are frequently detected at imaging. Hyperfunctioning tumors can also be detected. When small, it is sometimes difficult to differentiate them from lipid-poor adenomas.
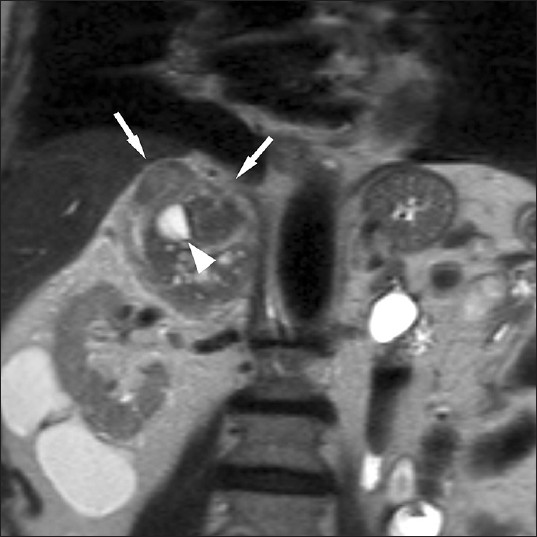
- 85-year-old male with abdominal pain diagnosed with right adrenal carcinoma. Coronal contrast-enhanced computed tomography scan shows a large, lobulated, right upper quadrant mass with heterogeneous enhancement (arrows) and nonspecific central areas of necrosis (arrowhead).
Pheochromocytoma
Pheochromocytoma is a rare tumor that arises from chromaffin cells of adrenal medulla.
Most lesions produce an excess of catecholamine that are responsible for the typical signs and symptoms related to this neoplasm, such as episodic hypertension, headache, anxiety, palpitation, and tachycardia. On CT and MR imaging, the best diagnostic imaging clue is early and bright enhancement after contrast injection and marked hyperintensity on T2-weighted MR images (the so-called “light bulb sign”) [Figure 7].[14] Unfortunately, a wide spectrum of imaging appearances has been described due to the presence of hemorrhage, necrosis, and calcifications. Wash-out characteristic are also variable and pheochromocytomas may mimic adenomas (i.e., APW >60%, RPW >40%).
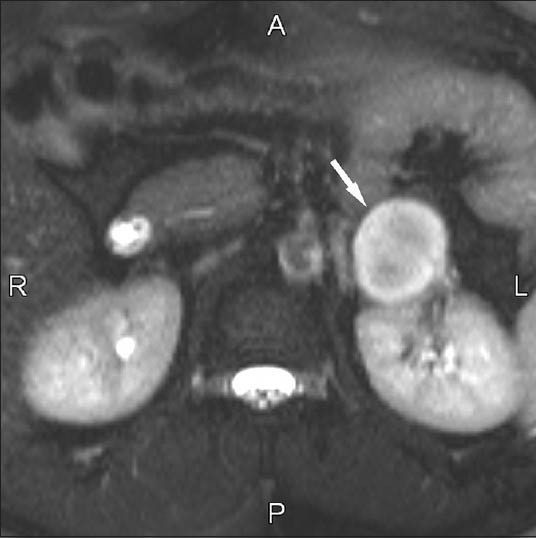
- 55-year-old male with hypertension with left adrenal pheochromocytoma. Transverse fat-saturated T2-weighted half-Fourier acquisition single-shot turbo spin-echo sequence (∞/90) shows a markedly hyperintense well defined lesion (arrow).
Adrenal lymphoma
Adrenal glands may be occasionally involved in patients with diffuse nonHodgkin disease.
On CT and MR imaging, adrenal lymphomas show nonspecific findings and are usually seen as bilateral large soft tissue masses (40-60 HU) with mild progressive enhancement after contrast medium administration [Figure 8]. Imaging characteristics may resemble other malignancies.
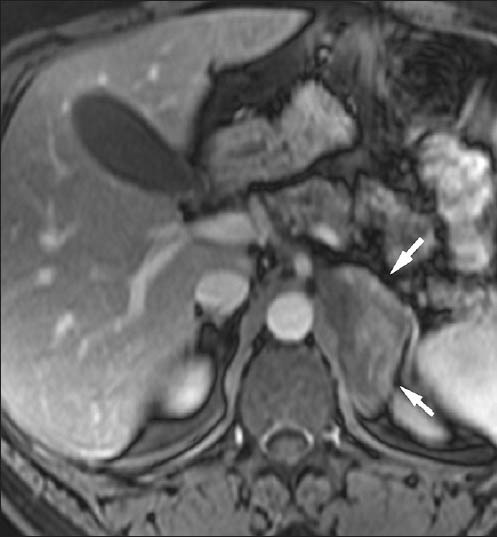
- 43-year-old male with left adrenal lymphoma. Transverse in-phase (4.5/1.9) T1-weighted gradient-echo magnetic resonance image with fat suppression shows a large left adrenal lesion (arrow) with mild and heterogeneous enhancement. Differential diagnosis with adrenal metastases may not be possible with imaging findings.
Neuroblastoma
Neuroblastoma is a poorly differentiated neoplasm derived from neural crest cells and it represents the second most common pediatric abdominal mass (after Wilms tumor), in children (40% arises from adrenal glands). This tumor is generally silent, however, it may invade adjacent organs causing paraneoplastic syndrome. It has been called “the great mimicker” because of its myriad clinical presentations related to the site of the primary tumor, metastatic disease, and its metabolic tumor by-products.
On CT, neuroblastoma appears as a lobulated, soft-tissue mass, either homogeneous or heterogeneous, which is caused by hemorrhage, necrosis, and/or calcification. MR imaging is useful for evaluating the location, extent and spread of neuroblastoma.
Neuroblastoma generally shows heterogeneous low signal intensity on T1-weighted images, high signal intensity on T2-weighted images and enhancement after administration of contrast material. Calcifications are reported in approximately 85% of patients, but they are difficult to discern on MR images. Areas of intra-tumoral hemorrhage and cystic changes may be present.
Vascular encasement and compression of the renal vessels, splenic vein, inferior vena cava, aorta, celiac artery, and superior mesenteric artery may occur even if vascular invasion is infrequent.
Ganglioneuroblastoma
Similar to neuroblastomas, ganglioneuroblastomas also arise from the neural crest. Ganglioneuroblastoma tends to be smaller and more well-defined than neuroblastoma at diagnosis.
It usually shows intermediate signal intensity on T1-weighted images and heterogeneously high signal intensity on T2-weighted images, with heterogeneous moderate enhancement after administration of contrast material.
CONCLUSION
The characterization of an adrenal lesion has been extensively investigated in radiology. Due to the high spatial resolution, rapid acquisition times, and multiplanar capabilities, mutlidetector CT is the primary modality for the detection and characterization of adrenal masses, especially in the oncologic population. Attenuation values on precontrast CT scan and the enhancement pattern after dynamic infusion of contrast medium are key features to differentiate adenomatous from nonadenomatous adrenal lesions.
Chemical shift MR imaging currently represents a problem solving modality for lesions that exhibit atypical imaging findings at CT. The proton MR imaging spectroscopy has been proposed as a complementary methodology for the study of adrenal gland, particularly for the differential diagnosis of adrenal lesions (benign lesion vs. malignant lesion).[15]
Analyzing the ratio of metabolites (i.e., colin, creatinine, and lipids) by spectroscopic exam, can help one collect additional information about the nature of the lesions identified by MRI.
Nevertheless, although the results obtained so far are encouraging, it is worth noting that they only refer to an initial experience and, consequently, it is necessary to acquire further statistical data to validate the accuracy of the methodology. In a limited number of cases, percutaneous biopsy is still necessary to definitely characterize atypical adrenal lesions.[15]
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2013/3/1/61/124088
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Characterization of adrenal masses using unenhanced CT: An analysis of the CT literature. AJR Am J Roentgenol. 1998;171:201-4.
- [Google Scholar]
- ACR Committee on Appropriateness Criteria. ACR Appropriateness Criteria on incidentally discovered adrenal mass. J Am Coll Radiol. 2006;3:498-504.
- [Google Scholar]
- Distinguishing benign from malignant adrenal masses: Multi-detector row CT protocol with 10-minute delay. Radiology. 2006;238:578-85.
- [Google Scholar]
- Adrenal masses: Characterization with combined unenhanced and delayed enhanced CT. Radiology. 2002;222:629-33.
- [Google Scholar]
- Adrenal masses: CT characterization with histogram analysis method. Radiology. 2003;228:735-42.
- [Google Scholar]
- Lipid-poor adenomas on unenhanced CT: Does histogram analysis increase sensitivity compared with a mean attenuation threshold? AJR Am J Roentgenol. 2008;191:234-8.
- [Google Scholar]
- Characterization of adrenal tumors by chemical shift fast low angle shot MR imaging: Comparison of four methods of quantitative evaluation. AJR Am J Roentgenol. 2003;180:1649-57.
- [Google Scholar]
- Effectiveness of a three-dimensional dual gradient echo two-point Dixon technique for the characterization of adrenal lesions at 3 Tesla. Eur Radiol. 2012;22:259-68.
- [Google Scholar]
- Adrenal masses: MR imaging features with pathologic correlation. Radiographics. 2004;24(Suppl 1):S73-86.
- [Google Scholar]
- Imaging of nontraumatic hemorrhage of the adrenal gland. Radiographics. 1999;19:949-63.
- [Google Scholar]
- Adrenal masses: Characterization with in vivo proton MR Spectroscopy-initial experience. Radiology. 2007;245:788-97.
- [Google Scholar]






