Translate this page into:
Vascular Access for Placement of Tunneled Dialysis Catheters for Hemodialysis: A Systematic Approach and Clinical Practice Algorithm
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The role of interventional radiology in the overall management of patients on dialysis continues to expand. In patients with end-stage renal disease (ESRD), the use of tunneled dialysis catheters (TDCs) for hemodialysis has become an integral component of treatment plans. Unfortunately, long-term use of TDCs often leads to infections, acute occlusions, and chronic venous stenosis, depletion of the patient's conventional access routes, and prevention of their recanalization. In such situations, the progressive loss of venous access sites prompts a systematic approach to alternative sites to maximize patient survival and minimize complications. In this review, we discuss the advantages and disadvantages of each vascular access option. We illustrate the procedures with case histories and images from our own experience at a highly active dialysis and transplant center. We rank each vascular access option and classify them into tiers based on their relative degrees of effectiveness. The conventional approaches are the most preferred, followed by alternative approaches and finally the salvage approaches. It is our intent to have this review serve as a concise and informative reference for physicians managing patients who need vascular access for hemodialysis.
Keywords
End-stage renal disease
hemodialysis
tunneled dialysis catheters
vascular access
INTRODUCTION

According to the United States Renal Data System, in 2008, there were more than 382,000 patients with end-stage renal disease (ESRD) in the USA. Over 90% of these patients were treated with hemodialysis.[1] Renal replacement therapy is needed, with renal transplantation being the treatment of choice for ESRD.[23] However, not all patients are suitable candidates for a kidney transplant and it takes time to find a kidney match. Hemodialysis (in-center or at-home) and peritoneal dialysis (continuous or intermittent) are the alternate options for patients awaiting renal transplantation.[45]
ACCESS OPTIONS FOR HEMODIALYSIS
There are three major types of vascular access: Arteriovenous fistula (AVF), arteriovenous graft (AVG), and tunneled dialysis catheters (TDCs). The non-tunneled central vein catheters (CVCs) are insignificant, as they are only used as temporary access. The AVF surgically creates direct communication between an artery and a vein, usually in the non-dominant arm of the patient. AVG is similar, but uses an artificial tube to connect the artery and the vein.[6] AVFs are the preferred vascular access type for dialysis, as they have the highest long-term patency rates and the lowest morbidity and mortality rates.[7] The AVFs need at least 2 months to mature for optimal dialysis. The TDC serves as a bridge to AVF dialysis. The National Kidney Foundation Dialysis Outcomes Quality Initiative (NKF-KDOQI) recommends that less than 10% of chronic maintenance hemodialysis patients can be maintained on catheters as their permanent dialysis access [Reference Dialysis Outcomes Quality Initiative (DOQI)]. In certain clinical situations, such as the lack of suitable vessels for AVF or the inability to use these fistulae due to complications like thrombosis or bleeding, the use of TDCs has been crucial.[5]
The main complications of TDCs are infections, occlusions, and mechanical complications. Infections include bacteremia and sepsis. Occlusions include thrombosis and stenosis of the involved veins. Mechanical complications consist of hemorrhages, such as those from puncturing vessels during the procedure, and, less commonly, pneumothoraxes, now infrequently seen due to the use of real-time ultrasound (US) guidance for access.[8910]
ACCESS OPTIONS FOR PLACEMENT OF TDCS
The site of vascular access has immense clinical relevance with respect to morbidity and mortality rates. Although several different veins can be chosen for the placement of a TDC, a vein can only be used for a limited amount of time before occlusions develop.[11] Central venous stenosis remains common in dialysis patients.[12] Steady exhaustion of central venous access options is an inevitable, potentially life-threatening outcome in patients who are dependent on long-term central venous catheters. Therefore, it is necessary to find another catheter access site with enough blood flow for dialysis.[13] A non-random study by Fry et al., found median survival times to be significantly longer for a first-time tunneled catheter insertion compared to subsequent tunneled catheter insertions (647 days vs. 403 days).[14]
According to the NKF-KDOQI, the right internal jugular vein (RIJV) is the preferred vascular access site for tunneled-cuffed central venous catheters. The left internal jugular vein (LIJV), the external jugular veins, the femoral veins, the subclavian veins (SCV), transhepatic accesses, and translumbar accesses are other access options.
In this review, we discuss the advantages and disadvantages of each access option, with illustrative case histories and images.
Conventional access options
Internal jugular veins
The internal jugular veins are the preferred sites of access for the placement of TDCs. The internal jugular veins are superficial and have the largest radii of the neck veins, making them the easiest veins to access technically. The use of US guidance for placement of the catheters reduces the risk of complications.[1516] It is an accepted fact that due to their large radii, the internal jugular veins have low risk of thrombosis.[1718] Also, thrombosis in the internal jugular veins is not clinically apparent. This is mainly due to the isolation of the thrombosis from the high outflow drainage of the upper extremities, as well as due to the development of collaterals. However, access through the internal jugular veins has been associated with central venous stenosis.[19]
The RIJV is preferred over the LIJV. The RIJV provides a short, direct, and linear route into the right atrium (RA) in contrast to the perpendicular route of the left-sided access. This asymmetry is proposed to be an etiological factor in the relatively increased incidence of LIJV stenosis.[20] Since the left arm is the predominant vascular access site for AVFs in right-handed people, placing a catheter in the contralateral RIJV reduces the risk of thrombosis in the left-sided venous drainage system, which could be harmful to functioning of the graft.[21] Other advantages of an RIJV access include minimal chance of damage to the thoracic duct,[22] which is anatomically located on the left side and technical ease of puncture due to better responsiveness to Valsalva maneuver.[23] A randomized study by Sulek et al., compared 60 RIJV and 60 LIJV catheters and found that the US-guided cannulation times were significantly longer (138 s vs. 58 s), the attempts were significantly more (2.3 vs. 1.5 needle passes), and the complication rates were significantly higher (20% vs. 10%) for LIJV catheters compared with RIJV catheters.[23] Fry et al., compared median survival times and found that patients with RIJV catheters survived longer (630 days vs. 430 days).[14] Figures 1 and 2 depict patients with the placement of TDCs via the RIJV and the LIJV, respectively.

- 61-year-old male with hypertension (HTn) and hepatitis C presented with ESRD, requiring hemodialysis. The RIJV was patent. (a) Frontal fluoroscopic spot image of the upper chest shows a 0.035-inch wire (arrow) was advanced through the RIJV into the right atrium (RA). (b) Frontal fluoroscopic imaging of the upper chest shows a TDC placed via the RIJV (arrow).

- 19-year-old female with glomerulonephritis presented with ESRD, requiring dialysis. A single incision technique was chosen. (a) Fluoroscopic spot image of the right upper chest and neck shows the distal end of the needle in its course through a subcutaneous tunnel from an infraclavicular location to the site of RIJV puncture. The distal end of the needle was manually bent to create a favorable angle (arrow in a). (b) Fluoroscopic spot image of the right upper chest after insertion of a 0.035-inch guidewire shows the dilatation of the tract d (arrow in b). (c) Frontal fluoroscopic spot image after a 23-cm cuff-to-tip TDC is placed (arrow). The patient had only one incision in the infraclavicular region, avoiding scarring in the neck.
Single incision technique
A novel technique that has gained popularity recently is the single incision technique. Various reports have labeled it as a feasible and safe alternative to the conventional internal jugular vein technique. Contractor et al., successfully placed 72 TDCs with the advantages of fewer procedural steps and a superior cosmetic result.[24] Figure 2 shows a patient with a TDC placed into the RIJV using a single incision technique.
External jugular vein
When the RIJV cannot be used and the patient has or will have a left arm AVF or AVG, the use of right external jugular vein (REJV) is optimal.[25] In their experience with 49 catheters placed through the external jugular veins, Beigi et al., indicated that the external jugular veins were a safe and reliable vascular access option with no procedural complications in 81.6% of the patients and a mean duration of sufficient performance of 324 days. Thrombosis (36%), accidental removal (22%), and infection (14%) were the main complications noted at the end of 1 year.[5] As the external jugular veins have smaller diameters than the internal jugular veins, adequate flow would be a concern for dialysis. This concern was successfully addressed by Vats et al., who found that external jugular vein blood flow outcomes at 30 days and 90 days were comparable to both LIJV and RIJV historical outcomes, suggesting a role for the external jugular veins as a potential long-term access site.[25] A disadvantage of the use of the external jugular veins is that they are very superficial and have smaller radii than the internal jugular veins; hence, access using US can be more challenging.[26]
Figure 3 depicts patients with placement of TDCs via the REJV.

- 61-year-old female with HTn, hypoglycemia, and Type II diabetes mellitus (DM), ESRD, needing hemodialysis. (a) Frontal fluoroscopic spot image of upper chest shows TDC in the RIJV (arrow). Two years later, patient presented with bacteremia, requiring catheter removal. Reattempt to access the RIJV was unsuccessful. Instead, REJV was accessed and a TDC was placed. (b) Frontal fluoroscopic imaging of upper chest shows small amount of extravasation of contrast (arrow). Recurrence of bacteremia resulted in need for catheter removal. (c) Frontal fluoroscopic venogram of the upper chest reveals occlusion of the REJV (arrow). (d) Frontal fluoroscopic spot image of upper chest reveals a TDC placed via the LIJV (arrow).
Dealing with catheter malfunction and central vein stenosis
It is important to maximize the use of conventional access options, as they are associated with lower complication rates. Every attempt must be made to treat the underlying etiology in an effort to facilitate catheter exchanges, rather than seeking alternative sites.[27]
Etiology
Some of the major reasons for catheter malfunction are fibrin sheath formation, catheter kinking or malposition, and thrombosis, both acute and chronic.[28] Central vein stenosis due to endothelial injury, associated with the placement of central venous catheters, can endanger AVFs or AVGs in either extremity. Usually asymptomatic, these manifest clinically when challenged by increased flow, commonly from AVFs or AVGs. The risk factors include multiple catheters, longer duration, subclavian venous location, and placement on the left-hand side of neck.[29]
Management
Catheter fibrin sheaths can be easily disturbed by disruption during guide wire exchange or use of balloon angioplasty.[30] In case of acute thrombosis, various thrombolysis or thrombectomy techniques can be used to restore patency, albeit with a small risk of bleeding during these procedures.[31] Endovascular interventions like percutaneous angioplasty are the mainstay in the management of the more chronic central vein stenosis, with stent placement reserved for recurring lesions.[2729]
Other conventional approaches
Femoral vein
Due to their large diameters and easy access, the femoral veins are a useful option for vascular access. Zaleski et al., in their experience with 41 tunneled femoral vein catheters, concluded that femoral vein catheters are more susceptible to infection as well as occlusion, thus requiring frequent catheter care. They suggested the use of an anterolateral upper thigh site of tunnel to avoid contamination of the skin exit site by groin infection. To improve patency, they suggested the tip of the catheter should be placed above the level of the confluence of the common iliac veins.[32] Placement of femoral catheters has the distinct drawback of restricting the mobility of the patient during walking and exercise.[32]
If a femoral access is chosen, the right femoral vein (RFV) is preferred over the left femoral vein (LFV) as it allows a left-sided AVF or AVG in the leg, if needed.[2133]
Figure 4 depicts a patient with placement of TDC via a right common femoral vein.

- 47-year-old female with HTn, Type II DM, coronary artery disease presented with ESRD, requiring hemodialysis. (a) Frontal fluoroscopic spot image reveals a TDC placed via the RIJV (arrow). However, bacteremia necessitated catheter removal. US revealed occlusion of RIJV (image not shown). (b) REJV was attempted, but a digital subtraction venogram of the chest reveals outflow venous occlusions (arrow). Access via the RFV was chosen instead. (c) Frontal fluoroscopic spot image in the region of right groin shows a TDC in place (arrow). (d) Frontal fluoroscopic spot image of the abdomen shows the catheter coursing through the IVC (arrow).
Subclavian vein
The DOQI recommends avoiding the SCV when upper extremity options for peripheral hemodialysis shunts remain viable in patients with renal failure (DOQI guidelines 5 and 6).[34] Occlusion/stenosis occurs commonly and results in clinically symptomatic venous outflow obstruction in the arm, as well as the loss of future access options for upper extremity AVF creation.[2835] Another major complication is pneumothorax (seen in 0.1–15% of cases).[36]
Figure 5 depicts a patient with placement of a TDC via the right SCV (RSV).

- 57-year-old male presented with ESRD, requiring hemodialysis. (a) Frontal fluoroscopic imaging of the upper chest shows the patient had a previous TDC placement at the RIJV (arrow) that had been subsequently removed due to persistent malfunction. (b) Frontal fluoroscopic spot image of the upper chest again shows a TDC placed into the RA via the RSV (arrow).
Alternative approaches
Collateral and small veins
Mediastinal, chest wall, and thyrocervical collaterals that develop secondary to occluded jugular veins have been used for vascular access in patients with huge success.[37] There are many benefits of using collateral veins in the neck region in preference to larger veins in the lower half of the body. First, the path to the superior vena cava (SVC) or RA is relatively direct. Second, the catheter care is similar to the jugular vein TDCs, and thirdly, patients tend to find them more comfortable. However, the most convincing reason for using these veins is that the other access sites are preserved for future use. The downside is that accessibility of these veins depends on the individual patient's anatomy and sometimes may not be technically easy.[34]
An antegrade approach via access in a vein in the chest is preferred by some authors due to ease of access. However, choosing a vein with a straight course is important for adequate long-term functioning of the TDC.[34] Others favor a retrograde transfemoral approach citing the potential for increased risk of bleeding due to use of the already compromised venous return of the upper half of the body in the antegrade approach.[28] In the retrograde approach, it is of paramount importance that the small collateral vein is patent and superficial, without intervening structures such as muscle or artery, so that snaring is technically possible via a fluoroscopy-guided antegrade percutaneous puncture.[28]
Funaki et al., reported primary patency rates of 90%, 71%, and 25% after 1, 6, and 12 months, respectively, in a study of 25 catheters placed in the neck region. They, in fact, favor the use of recanalized veins over femoral veins.[37]
Translumbar inferior vena cava
The TDCs can also be placed via a translumbar approach into the inferior vena cava (IVC). The anatomic position of the IVC is reliable and due to the large radius, thrombosis is rare. Although it is technically more challenging and time consuming compared to other techniques, the procedure is relatively safe, with the only important structure in the vicinity being the ureter.[38] Complications are avoided by performing in the prone position with a longer length, yet smaller gauge needle to reach the IVC. Although fluoroscopic guidance is commonly used to access just above the iliac crest just to the right of L3 vertebral body, US may sometimes be useful in lean patients.[28] Liu et al., reported catheter patency rates of 43%, 25% 5, and 7% after 3, 6, and 9 months, respectively, in a study of 84 translumbar TDCs. The main complications were poor blood flow (40%) and catheter-related infection (36%), which led to high incidence of catheter removal. Data from this study suggest that although translumbar catheters might serve as a safe, alternate access for dialysis patients, they function well for a short term as their use is limited by frequent exchanges.[39] Biswal et al., in their study, reported catheter dislodgement due to excess adipose tissue in the tunnel area, as well as dislodgement due to movement or catching on adjacent objects to be an important cause of catheter failure.[40]
Figure 6 shows placement of a TDC in the transhepatic IVC.

- 41-year-old female with ESRD, needing hemodialysis. (a) Frontal fluoroscopic spot image of the upper chest shows a malfunctioning IJV (arrow). TDC's (b) arm and (d) coronal CT venography of chest reveals SVC stenosis (arrow in b, d), left brachiocephalic vein stenosis (white arrow in c), and collaterals (white arrow in e). Femoral veins were occluded. (f) Frontal fluoroscopic spot image of the abdomen shows a translumbar IVC TDC (arrow in f). (g and h) Axial CT abdominal images show the course of catheter through abdominal wall (arrow in g) and into the IVC (arrow in h). Patient returned due to catheter dysfunction (i and j). Frontal fluoroscopic imaging shows balloon dilation of IVC stenosis (arrow in i); as it failed, stent was placed (midline horizontal arrow in j). Incidentally noted is a Transjugular intrahepatic portosystemic shunt (TIPS) stent (oblique arrow in right hypochondrium).
Hepatic vein
In spite of being the last resort vascular access in patients with infrarenal IVC occlusion, the transhepatic approach is advantageous.[41] In 2003, Stavropoulos et al., studied 36 transhepatic catheters and reported a primary patency of just 24.3 days, the major reason being a high rate of late thrombosis.[42] Recently, Younes et al., in a study of 127 transhepatic catheters, reported a much higher patency of 87.7 days. Although catheter-related maintenance in the form of exchanges was high (mean of 7.5 exchanges per patient), they were of the opinion that transhepatic catheters provide a potentially viable long-term access for patients with exhausted access options.[43] Smith et al., opined that hepatic vein TDCs are associated with a high rate of catheter malfunction. As these catheters have a long intrahepatic path, they move with liver excursions during respiration, resulting in either proximal migration/dislodgement or distally potentially contacting the tricuspid valve or the right atrial wall.[44] It should be noted placement of hepatic vein TDCs can result in liver hemorrhage in about 29% of the cases.[44]
Figure 7 shows a patient with a TDC placed in the hepatic vein.

- 49-year-old female with metastatic breast cancer, coagulopathy, and renal insufficiency and a malfunctioning LIJV catheter. (a) Frontal fluoroscopic spot image of the chest shows TDC in LIJV (arrow). (b) Frontal digital subtraction venography after injection of contrast via the right brachial vein venography reveals right-sided central venous occlusion at the level of the RSV (arrow). Transhepatic catheter placement was decided. (c) Frontal digital subtraction venography of the right hypochondrium shows the venous access that was obtained into the hepatic vein (arrow in c). (d) Frontal fluoroscopic spot image of the abdomen shows a TDC placed via the hepatic vein (arrow), with the tip in the right atrium.
Salvage options
Uncommonly used veins
Brachiocephalic veins
The brachiocephalic approach is used in cases of bilateral jugular venous occlusions. Falk evaluated the placement of TDCs in 33 right brachiocephalic veins and 11 brachiocephalic veins, and reported a 68.2% patency rate after 30 days, with a mean survival time of 92 days for those catheters that remained patent. The author opined that though US-guided visualization of the brachiocephalic vein is a challenging aspect of the procedure, in experienced hands, 100% technical success rate can be achieved.[45]
Figure 8 shows a patient with a TDC placed in the left brachiocephalic vein.

- 54-year old female with ESRD, needing hemodialysis. (a) Frontal fluoroscopic spot image of the chest shows TDC in RIJV (arrow). (b and c) Few months later, digital subtraction venograms of the chest revealed SVC stenosis (arrow). (d) Frontal fluoroscopic spot image of the chest shows balloon angioplasty of the stenosis (arrow), which was unsuccessful. RFV TDC was placed. Few months later, patient had a catheter malfunction. (e) Fluoroscopic spot image of the upper abdomen shows venoplasty of the suprarenal IVC stenosis (arrow), which was again unsuccessful. (f) A hypertrophied anterior jugular vein was accessed. Frontal digital subtraction venography shows the anterior jugular vein (arrow) that appears to empty into the left brachiocephalic vein. (g) Frontal digital subtraction venography shows access of the left brachiocephalic vein (arrow). (h) Frontal fluoroscopic spot image of the chest shows a TDC in place.
Obturator vein
The obturator vein is a particularly challenging vein for hemodialysis due to its very small radius. The risk of using the obturator vein includes hemorrhage, infection, thrombosis, and stenosis. The obturator artery, vein, and nerve course through the obturator canal, a small arched opening in the superior aspect of the obturator foramen, a potential site of vascular or nerve injury during placement of TDCs via a obturator vein approach (Salsamendi et al., in press).[46]
Figure 9 shows the placement of a TDC via the obturator vein.
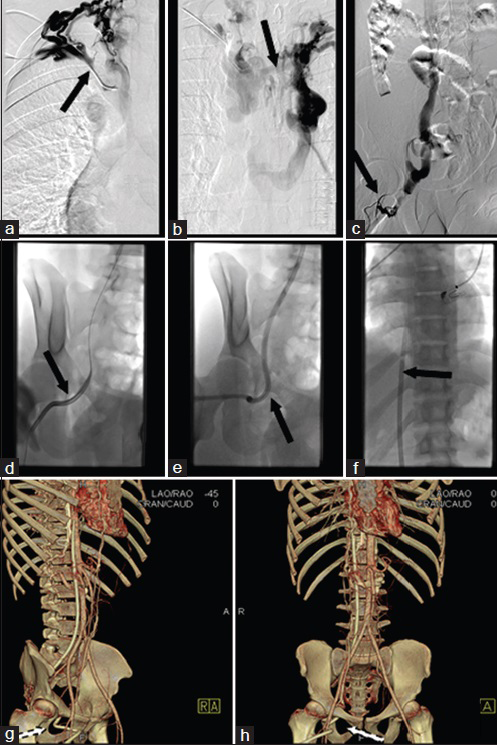
- 21-year old male with congenital renal dysplasia needing dialysis. Frontal digital subtraction venography of the chest via a right arm (a) and left arm (b) contrast injection reveals occlusion of the right (arrow in a) and left (arrow in b) brachiocephalic veins. Multiple superficial collaterals were seen in the right groin, one of which was accessed using US. (c) Frontal digital subtraction venography of the abdomen reveals a tiny collateral vein draining into right obturator vein. (d–f) Frontal fluoroscopic imaging shows dilatation of the tract (arrow in d) and advancement of the TDC (arrow in e). Tip of the catheter in the suprarenal IVC/right atrial junction (arrow in g). (g and h) 3D-CT volume reconstruction images show TDC through the obturator foramen (arrows in g, h).
External iliac vein
The external iliac vein has been described in a patient with femoral vein occlusion. However, in general, it should be avoided, as the puncture of the external iliac vein is a higher puncture as compared to that of the femoral vein, and thus has a higher risk of retroperitoneal bleeding.[47]
Renal vein
A renal vein approach has been described, but renal veins are undesirable in patients with kidney disease.[48]
Sharp recanalization technique
When the techniques for venous access described above have failed, sharp recanalization technique (SRT) can be attempted. However, it is associated with a high risk of morbidity and mortality. Although a detailed discussion of the techniques is beyond the scope of this article, a few points are noteworthy.
The ultimate goal of SRT is to find an accessible neck or chest vein in very close proximity to a remnant central vein with direct communication to the RA. Prior assessment with detailed “roadmap” venogram to image all collateral veins can be enhanced by a review of all available cross-sectional imaging studies. Based on location, distance, and orientation, the veins are then assessed for the risk of perforating vital mediastinal structures during “blind” needle puncture. Many authors have described the use of a sheathed transjugular intrahepatic portosystemic shunt needle or stiff end of a glide wire to traverse the chronic stenosis. Once access is obtained and through-and-through guide wire access is achieved, subsequent balloon dilatation is performed to enable placement of a TDC.[284950]
The Hemoaccess Reliable Outflow vascular access device
In 2008, the US Food and Drug Administration approved the Hemoaccess Reliable Outflow (HeRO) vascular access device (Hemosphere, Inc., Minneapolis, MN, USA) as a method to establish access in dialysis patients with severe central venous disease.[515253] The HeRO device is an expanded polytetrafluoroethylene (ePTFE) graft placed in the upper arm over the biceps muscle and attached to a nitinol-reinforced silicone outflow component placed in the RA via titanium connector placed subcutaneously. Thus, it bypasses central venous stenosis.
Early results suggested that the infectious complications and re-intervention rates were significantly lower for the HeRO device as compared to TDC. The secondary patency rates approximated AVGs.[5455] In 2009, Katzman et al., implanted HeRO in 36 patients. The infection rate was 0.70/1000 days with bacteremia occurring prior to HeRO's implantation, when a TDC was still present. They recorded high primary patency rates of 38.9% and secondary patency rates of 72.2%, thus suggesting a major role of HeRO device in patients who are chronically dependent on dialysis.[53]
Although initial experience demonstrated excellent patency rates, subsequent studies were not very encouraging. In 2013, based on their experience in 19 patients, Wallace et al., reported high technical success but poor patency (12-month primary and secondary patency rates of 11 and 32%, respectively) and a high rate of infection (0.5 bacteremic events per 1000 HeRO-days) that required many re-interventions. Also, a high rate of arterial “steal syndrome” (24%) was noted that required ligation.[56] More recently in 2014, Torrent et al., have reported better secondary patency rates (81.6% at 6 months and 53.7% at 12 months). However, of note was that the re-intervention rate was 2.84 procedures per HeRO vascular access device year, which resulted in a high cost of maintenance ($34,713.63 per patient/year).[57]
Thus, although the HeRO device appears effective,[515253] its use should be reserved to create a “salvage” access for those who fail all other traditional AVF and graft options and should be offered as an alternative to the leg AVG.[5657]
Surgery
When all the above options fail, a few surgical alternatives may be available. Direct right atrial catheterization[58] and extra-anatomic surgical venous bypass[59] have been described in case reports.
A SYSTEMATIC APPROACH TO THE VASCULAR ACCESS SITES OF HEMODIALYSIS
Pre-procedural workup
Apart from the available guidelines and recommendations, there are two factors that influence the choice of vascular site for access: Operator preference and patient anatomy. The operator's comfort as well as risk appetite and knowledge of the various techniques including guidewire and catheter skills are important. Familiarity of the patient's vascular anatomy is also essential. Crucial to the understanding of the next “best option” in vascular access is a detailed understanding of the clinical history and performing a thorough physical examination.[38] Figure 10 is a summary of advantages and disadvantages of various access options.

- Summary of advantages and disadvantages of various access options.
Figure 11 is a summary of key points in the pre-procedural workup when evaluating a patient for alternative sites of vascular access.

- Summary of key points in the pre-procedural workup when evaluating a patient for alternative sites of vascular access.
Vascular access planning and decision-making
The use of RIJV has become the standard of care and should be the first option, especially in anticipation of a left arm AVF or AVG. If occlusion occurs, an attempt should be made to access the REJV, rather than using the LIJV, again in anticipation of a left arm AVF or AVG. However, if the EJV cannot be used, a left-sided approach using LIJV or left external jugular vein (LEJV) should be used. The use of SCV is rare.[60]
There is controversy over which approach is the best next access. While some authors prefer the technically easier femoral vein,[32] catheterization of small supradiaphragmatic chest collaterals has gained popularity in more recent times.[3428] Exceptional catheter skills are needed in cannulating these collaterals. If these accesses fail, an infradiaphragmatic access site is the next best option. Careful attention must be paid to the site of the proposed AVG in the thigh and the catheter placed on the contralateral side. Here arises another controversy. Some authors suggest an ascending approach starting with the femoral vein followed by the translumbar IVC and then the hepatic vein.[28] The theory is that inserting catheters in the femoral vein does not jeopardize higher accesses, with the converse not being true. However, other authors have suggested the use of the translumbar IVC or the hepatic vein access first to avoid the high complication rates of femoral access sites.[38] Another point to consider is the high procedural risks associated with IVC and hepatic vein access. Again, although there is no evidence that one approach is superior to the other, in the choice between translumbar IVC and hepatic venous route, the translumbar route has been described as a “safer” option by some.[38]
If the alternative access options are exhausted, there are very few possibilities left. Salvage options are associated with a high risk of complications. Appropriate patient counseling and planning are recommended before performing these procedures, as the morbidity and mortality rates are high. Few case reports in literature describe the use of uncommonly used veins for placement of TDCs. The use of the novel HeRO device appears attractive[515253] for those who fail all other traditional AVF and graft options and should be offered as an alternative to the leg AVG.[5657] SRT of occluded veins can also be performed. Bypass surgery is the last resort, if all other options fail.[59]
CONCLUSION
In patients with ESRD, the use of TDCs for hemodialysis is an integral component in treatment. Unfortunately, the long-term use of TDC frequently leads to infections and chronic venous stenosis, depleting the patient's conventional access routes and preventing their recanalization. In such situations, the progressive loss of venous access sites should prompt a systematic approach to alternative sites to maximize the patient's survival and minimize complications.
The role of interventional radiology in the overall management of patients on dialysis continues to expand. A thorough knowledge of the various options available based on their relative degrees of effectiveness is crucial. The conventional approaches are the most preferred, followed by alternative approaches and then by salvage approaches. It is our intent to have this review serve as a concise and informative reference for physicians who are managing patients who need vascular access for hemodialysis. A handy flowchart providing a framework of the decision process is shown in figure 12.

- A handy flowchart provides the framework of the decision process.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2015/5/1/31/157858
REFERENCES
- End-stage renal disease in the United States: An update from the United States Renal Data System. J Am Soc Nephrol. 2007;18:2644-8.
- [Google Scholar]
- Systematic review: Kidney transplantation compared with dialysis in clinically relevant outcomes. Am J Transplant. 2011;11:2093-109.
- [Google Scholar]
- Kidney transplantation as primary therapy for end-stage renal disease: A National Kidney Foundation/Kidney Disease Outcomes Quality Initiative (NKF/KDOQI™) Conference. Clin J Am Soc Nephrol. 2008;3:471-80.
- [Google Scholar]
- Canadian Society of Transplantation: Consensus guidelines on eligibility for kidney transplantation. CMAJ. 2005;173:S1-25.
- [Google Scholar]
- Placement of long-term hemodialysis catheter (permcath) in patients with end-stage renal disease through external jugular vein. Adv Biomed Res. 2014;3:252.
- [Google Scholar]
- Complications of arteriovenous fistula in dialysis patients. Transplant Proc. 2006;38:1261-4.
- [Google Scholar]
- Ethical and legal obligation to avoid long-term tunneled catheter access. Clin J Am Soc Nephrol. 2009;4:456-60.
- [Google Scholar]
- Thrombotic and infectious complications of central venous catheters in patients with hematological malignancies. Ann Oncol. 2008;19:433-42.
- [Google Scholar]
- Use of real-time ultrasound guidance for the placement of hemodialysis catheters: A systematic review and meta-analysis of randomized controlled trials. Am J Kidney Dis. 2011;58:964-70.
- [Google Scholar]
- Vascular access for hemodialysis: Current perspectives. Int J Nephrol Renovasc Dis. 2014;7:281-94.
- [Google Scholar]
- Subclavian vascular access stenosis in dialysis patients: Natural history and risk factors. J Am Soc Nephrol. 1998;9:1507-10.
- [Google Scholar]
- Central vein stenosis: A common problem in patients on hemodialysis. ASAIO J. 2005;51:77-81.
- [Google Scholar]
- Factors affecting long-term survival of tunnelled haemodialysis catheters- A prospective audit of 812 tunnelled catheters. Nephrol Dial Transplant. 2008;23:275-81.
- [Google Scholar]
- Placement of hemodialysis catheters with a technical, functional, and anatomical viewpoint. Int J Nephrol 2012 2012 302826
- [Google Scholar]
- Epidemiology of hemodialysis vascular access infections from longitudinal infection surveillance data: Predicting the impact of NKF-DOQI clinical practice guidelines for vascular access. Am J Kidney Dis. 2002;39:549-55.
- [Google Scholar]
- Cross-sectional area of the right and left internal jugular veins. J Cardiothorac Vasc Anesth. 1999;13:136-8.
- [Google Scholar]
- Percutaneously inserted silastic jugular hemodialysis catheters seldom cause jugular vein thrombosis. ASAIO J. 1995;41:169-72.
- [Google Scholar]
- Venous stenosis and thrombosis associated with the use of internal jugular vein catheters for hemodialysis. ASAIO J. 1999;45:356-9.
- [Google Scholar]
- Right versus left internal jugular vein catheterization for hemodialysis: Complications and impact on ipsilateral access creation. Artif Organs. 2004;28:728-33.
- [Google Scholar]
- How I do it: Preferential use of the right external jugular vein for tunneled catheter placement. Semin Dial. 2008;21:183-5.
- [Google Scholar]
- A randomized study of left versus right internal jugular vein cannulation in adults. J Clin Anesth. 2000;12:142-5.
- [Google Scholar]
- Head rotation during internal jugular vein cannulation and the risk of carotid artery puncture. Anesth Analg. 1996;82:125-8.
- [Google Scholar]
- Single-incision technique for tunneled central venous access. J J Vasc Interv Radiol. 2009;20:1052-8.
- [Google Scholar]
- A comparison between blood flow outcomes of tunneled external jugular and internal jugular hemodialysis catheters. J Vasc Access. 2011;13:51-4.
- [Google Scholar]
- Insertion of long-term tunneled cuffed hemodialysis catheters via the external jugular vein by using a simple, safe and reliable surgical technique. J Vasc Access. 2007;8:12-6.
- [Google Scholar]
- Placement of tunnelled hemodialysis catheters across stenotic and occluded central veins. J Vasc Access. 2003;4:3-8.
- [Google Scholar]
- Percutaneous fibrin sheath stripping versus transcatheter urokinase infusion for malfunctioning well-positioned tunneled central venous dialysis catheters: A prospective, randomized trial. J Vasc Interv Radiol. 2000;11:1121-9.
- [Google Scholar]
- Thrombosis of tunneled-cuffed hemodialysis catheters: Treatment with high-dose urokinase lock therapy. Artif Organs. 2012;36:21-8.
- [Google Scholar]
- Experience with tunneled femoral hemodialysis catheters. AJR Am J Roentgenol. 1999;172:493-6.
- [Google Scholar]
- Use of the femoral vein as insertion site for tunneled hemodialysis catheters. Journal of J Vasc Interv Radiol. 2007;18:217-25.
- [Google Scholar]
- Unconventional Central Access: Catheter insertion in collateral or in recanalized veins. . 2004;21:111-7.
- [Google Scholar]
- Pneumothorax following ultrasound-guided jugular vein puncture for central venous access in interventional radiology: 4 years of experience. J Intensive Care Med. 2012;27:370-2.
- [Google Scholar]
- Imaging review of procedural and periprocedural complications of central venous lines, percutaneous intrathoracic drains, and nasogastric tubes. Pulm Med 2012 2012 842138
- [Google Scholar]
- Radiologic placement of tunneled hemodialysis catheters in occluded neck, chest, or small thyrocervical collateral veins in central venous occlusion. Radiology. 2001;218:471-6.
- [Google Scholar]
- Patency and complications of translumbar dialysis catheters. Semin Dial 2015 [Epub ahead of print]
- [Google Scholar]
- Translumbar placement of paired hemodialysis catheters (Tesio catheters) and follow-up in 10 patients. Cardiovasc Intervent Radiol. 2000;23:75-8.
- [Google Scholar]
- Percutaneous translumbar and transhepatic inferior vena caval catheters for prolonged vascular access in children. J Pediatr Surg. 1992;27:165-9.
- [Google Scholar]
- Percutaneous transhepatic venous access for hemodialysis. J Vasc Interv Radiol. 2003;14:1187-90.
- [Google Scholar]
- Transhepatic hemodialysis catheters: Functional outcome and comparison between early and late failure. J Vasc Interv Radiol. 2011;22:183-91.
- [Google Scholar]
- Use of the brachiocephalic vein for placement of tunneled hemodialysis catheters. AJR Am J Roentgenol. 2006;187:773-7.
- [Google Scholar]
- Successful obturator vein vascular access for hemodialysis catheter placement in patient with end-stage renal disease. J Vasc Access [In press]
- [Google Scholar]
- Using recanalized external iliac vein for tunneled hemodialysis catheter insertion. J Vasc Bras. 2008;7(2):171-173.
- [Google Scholar]
- Percutaneous transrenal hemodialysis catheter insertion. J Vasc Interv Radiol. 2002;13:1043-6.
- [Google Scholar]
- Sharp recanalization of central venous occlusions: A useful technique for haemodialysis line insertion. Br J Radiol. 2009;82:105-8.
- [Google Scholar]
- Sharp recanalization of central venous occlusions. J Vasc Interv Radiol. 1999;10:149-54.
- [Google Scholar]
- Modified use of the Hemodialysis Reliable Outflow (HeRO) graft for salvage of threatened dialysis access. J Vasc Surg. 2012;56:1127-9.
- [Google Scholar]
- Salvaging vascular access and treatment of severe limb edema: Case reports on the novel use of the hemodialysis reliable outflow vascular access device. Ann Vasc Surg. 2011;25:387.e1-5.
- [Google Scholar]
- Initial experience and outcome of a new hemodialysis access device for catheter-dependent patients. J Vasc Surg. 2009;50:607.e1.
- [Google Scholar]
- Long-term performance of the hemodialysis reliable outflow (HeRO) device: The 56-month follow-up of the first clinical trial patient. Semin Dial. 2010;23:229-32.
- [Google Scholar]
- Report on the Hemodialysis Reliable Outflow (HeRO) experience in dialysis patients with central venous occlusions. J Vasc Surg. 2013;58:742-7.
- [Google Scholar]
- Examining hemodialysis reliable outflow catheter performance and cost in hemodialysis access. J Surg Res. 2014;192:1-5.
- [Google Scholar]
- Successful prolonged use of an intracardiac catheter for dialysis. Nephrol Dial Transplant. 1999;14:2015-6.
- [Google Scholar]
- Surgical bypass for subclavian vein occlusion in hemodialysis patients. J Am Coll Surg. 2002;194:416-21.
- [Google Scholar]






