Translate this page into:
The Role of Computed Tomography in Predicting Left Ventricular Assist Device Infectious Complications
Address for correspondence: Dr. Carrie K Gomez, 601 Elmwood Avenue, Rochester, New York 14642, USA. E-mail: carrie_gomez@urmc.rochester.edu
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objective:
The purpose of this study is to identify early computed tomography findings around the driveline which would predict mediastinal or left ventricular assist device (LVAD) pocket abscess formation.
Materials and Methods:
A retrospective analysis was performed on 128 LVAD recipients between January 2007 and December 2011. Infectious complications were subdivided into those affecting the driveline and those resulting in abscess formation either around the LVAD pump or mediastinum. The size and location of infiltrative changes surrounding the driveline were used to predict infection propagation resulting in abscess.
Results:
Of the 128 patients, 49 (38.3%) patients developed driveline infections and 24 (18.8%) patients developed abscess. 87.5% patients who developed abscess had a preceding driveline infection. The mean time from driveline infection to the development of pump pocket abscess was approximately 7 months. In addition, patients with abscess in the pump pocket or mediastinum had preceding infiltrative changes surrounding the driveline ≥14 mm (P = 0.0001). A preperitoneal location and size of infiltrative changes ≥14 mm were correlated with a higher likelihood of abscess formation (P = 0.0002).
Conclusion:
Our study demonstrates the predictive value of infection/infiltrative changes around the driveline, which increases the risk for abscess formation in the LVAD pump pocket and/or in the mediastinum.
Keywords
Abscess
driveline
left ventricular assist device
mediastinal
pump

INTRODUCTION
Heart failure is now recognized as a major public health problem. According to the American Heart Association, there are approximately 5.1 million of Americans with heart failure and 500,000 new cases of heart failure are diagnosed each year.[12] The prognosis of heart failure is poor, with estimated rates of survival of 50% at 5 years and 10% at 10 years.[2] As the rate of heart failure continues to increase and surpass the number of donor's hearts, the need for left ventricular assist devices (LVAD) is rapidly rising. LVADs, in particular, the HeartMate II, have been used as a bridge for transplantation, as a bridge to recovery in patients with acute cardiogenic shock, and as a destination therapy for patients with end-stage heart failure not eligible for heart transplant.[345]
The HeartMate II device is a continuous axial flow device, which is implanted subdiaphragmatically into an extraperitoneal pocket (pump pocket). The LVAD pocket is placed in the left upper quadrant in the preperitoneal space, a space below the diaphragm between the posterior border of the rectus muscle and the peritoneal lining. The LVAD inflow cannula is anastomosed to the left ventricular apex, and the outflow cannula is anastomosed to the ascending aorta. The HeartMate II is motorized by an axial rotatory pump made of titanium which helps circulate blood from the left ventricle into the inflow cannula, through the rotatory pump, and out the outflow cannula into the aorta. A driveline wire traverses the abdominal wall to connect the external battery pack to the internal motor [Figure 1a and b].[567]
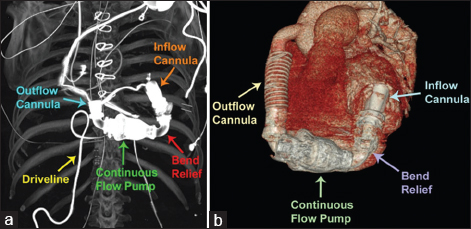
- Coronal computed tomography maximum intensity projection of the chest (a) and three-dimensional reconstruction (b) portray the components of the HeartMate II left ventricular assist device (inflow and outflow cannulas, flow pump, driveline, and bend relief).
The LVAD has been associated with multiple complications such as infection of the percutaneous driveline, abscesses, peripheral/LVAD thrombotic events, hemorrhagic complications derived from antithrombotic therapy, postoperative hematoma, kinking, rotation, and malposition of the inflow or outflow cannulas.[48]
The driveline is frequently implicated as the source of LVAD-associated infections. Superficial exit site infections can extend and involve the driveline distally.[91011] Several studies indicate the driveline infection rates for the HeartMate II continue to be as high as 20%,[9] and one study reported driveline infection rates to be as high as 30%–50%.[10] Miller et al., reported that the rate of driveline infections in recipients of HeartMate II as a bridge to transplantation was 17.5%.[12] In the Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure trial, investigators found that the prevalence of driveline and pocket infection was 28%.[13] In a more recent trial with the HeartMate II device as a bridge to transplant, the prevalence of driveline infection was 14%.[13] Moreover, among LVAD recipients who die of infections while awaiting transplantation, 29% had driveline infections.[14] Once the infection extends from the driveline and reaches the extraperitoneal space, the subsequent risk of pump pocket abscess/intra-abdominal or mediastinal abscesses increases. If the infection affects the pump pocket, more aggressive measures are needed such as prolonged intravenous therapy, extensive debridement, pump pocket irrigation drainage systems, and ultimately LVAD explantation.[11151617]
These complications can be costly and devastating to the patient; therefore, our aim is to identify early computed tomography (CT) findings around the driveline to predict mediastinal or LVAD pocket abscess formation. Such prediction may help in performing preemptive interventions to halt progression of infection and its extent into the pump pocket.
MATERIALS AND METHODS
Patient population
A retrospective analysis was performed on 128 HeartMate II LVAD recipients over a 5-year period between January 2007 and December 2011 [Table 1]. Following the Institutional Review Board approval, patients’ electronic medical records and imaging studies were examined to categorize the various infectious complications. The HeartMate II recipients from 2007 to 2011 were included in the study. The following groups were excluded from the study: HeartMate II recipients with infections from other sources, not arising or related to HeartMate II LVAD implantation; recipients with no CT imaging after receiving the HeartMate II LVAD; recipients with immediate postoperative infectious complications; and recipients without sufficient imaging follow-up including those with no imaging pertaining to the infection.
Infectious complications were categorized as driveline infection, pump pocket abscess, and mediastinal abscess. Driveline infections were defined clinically by the presence of external drainage with positive culture results. Mediastinal and pump pocket abscesses were noted when positive culture results stemmed from pocket drainage, device replacement, or removal operations. Forty-nine patients were found to have infectious complications. Of these 49 patients, 8 were removed from the dataset as 2 of the patients had no CT imaging, 3 lacked sufficient imaging follow-up, and 3 had abscesses associated with the initial surgery. The mean age of the patients included in the study was 55.5 years (standard deviation 11.9). Treatment of patients who developed driveline infection included the administration of systemic antibiotics and in some cases surgical drainage. Management of patients who developed pump pocket abscess comprised systemic antibiotics, surgical exploration/drainage, and device explantation in intractable cases.
Data from the 128 patients were analyzed to evaluate survival time and the length of time between device implantation and driveline infection, abscess, or device replacement. Kaplan–Meier survival curves were generated to evaluate 1- and 2-year survival probability estimates.
Further statistical analyses including odds ratios (ORs) and correlation coefficients were obtained to demonstrate complications that resulted in death or the need for device replacement.
Computed tomography image acquisition and interpretation criteria
CT images were obtained on Philips 16 and 64 slice CT scanners. For evaluation of driveline infections, the protocol included a noncontrast chest CT scan extending through the mid-abdomen to include the entire course of the driveline. The extent of the infiltrative change surrounding the driveline was measured by reviewing the patients’ CT images and measuring the maximum diameter of the fluid collection perpendicular to the driveline in the axial plane. The location of the infiltrative change was graded as subcutaneous or preperitoneal [Figure 2a and b]. Driveline infections were further divided into two categories based on their size: ≥14 mm or <14 mm. Preperitoneal infections ≥14 mm were considered to be worrisome infections. Contrast was used in patients with adequate renal function, and noncontrast studies were performed on patients with poor renal function and those who could not tolerate intravenous contrast.

- Axial computed tomography images at the level of the upper abdomen show the subcutaneous (a) and preperitoneal (b) infiltrative changes (yellow arrows) around the driveline.
RESULTS
Of the 128 study subjects, 49 patients developed driveline infection (38.3%) and 24 patients (18.8%) had abscess as complication. Of the 49 patients with driveline infections, 8 were excluded from the study due to lack of sufficient imaging and loss to follow-up.
The mean time from the placement of the HeartMate II LVAD to the development of driveline infection was approximately 13 months. On the other hand, the mean time from LVAD placement to abscess formation was longer, approximately 20.1 months.
The majority of the patients who developed abscess as a complication had a preceding driveline infection. In our study, 21 out of 24 (87.5%) of patients with abscess had involvement of the driveline prior abscess formation.
Moreover, the presence of infiltrative changes surrounding the driveline was an important factor to predict infection progression. It was found that 19 out of 21 patients with abscess formation had preceding infiltrative changes around the driveline measuring ≥ 14 mm (sensitivity 90.5%, specificity 80.0%; positive predictive value [PPV] 82.6%, OR 38.0, P = 0.0001). On the other hand, 2 out of 21 patients who developed abscess had preceding driveline infiltrative changes of <14 mm [Figure 3].
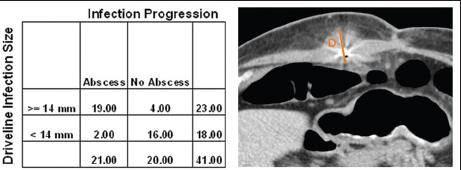
- Relationship between the driveline size of infiltrative changes and abscess formation (D: Represents the infiltrative changes around the driveline). Nineteen out of 21 patients with abscess formation had preceding infiltrative changes around the driveline measuring ≥14 mm (P = 0.0001).
We also assessed the location of the infiltrative changes around the driveline to evaluate infection spread. The location of infiltrative changes was classified as subcutaneous or preperitoneal. It was found that individuals with preperitoneal infiltrative changes were more prone to develop abscess. Eighteen out of 21 patients who developed abscess had preperitoneal infiltrative changes surrounding the driveline (sensitivity 85.7%, specificity 90%, PPV 90.0%, OR 54.0, P < 0.0001). In contrast, only 3 out of the 21 patients who formed abscess had preceding subcutaneous infiltrative changes around the driveline [Figure 4].
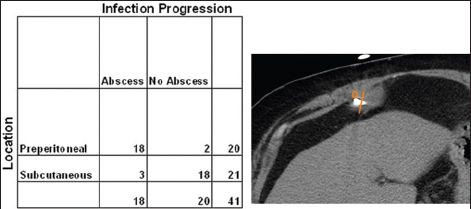
- Relationship between the driveline location of infiltrative changes and abscess formation (D: Represents the infiltrative changes around the driveline). Individuals with preperitoneal infiltrative changes surrounding the driveline were more prone to develop abscess. Note that 18 out of 21 patients who developed abscess had preperitoneal infiltrative changes surrounding the driveline (P < 0.0001).
A combination of the size and location of infiltrative changes around the driveline was also obtained to assess infection spread. A preperitoneal location and size of infiltrative changes ≥14 mm were correlated with a higher likelihood of abscess formation. Seventeen out of 21 patients who developed abscess had preperitoneal location and infiltrative change size ≥14 mm (sensitivity 81.0%, specificity 95.0%, OR 80.8, PPV 94.4% P = 0.0002) [Table 2].

Of the 128 patients, 9 patients required device replacement (7%) either due to malfunction or infection. Of these nine patients, 3 HeartMate II devices (33.3%) were replaced due to abscess.
Furthermore, 25 recipients of LVAD died due to different complications such as stroke, hemorrhage, pulmonary embolism, malposition, and infection. Of the 25 deaths, 4 (16%) were related to abscess.
Kaplan–Meier survival curve analysis determined a 1-year survival probability of 90% and a 20-month survival probability of 55% [Figure 5].
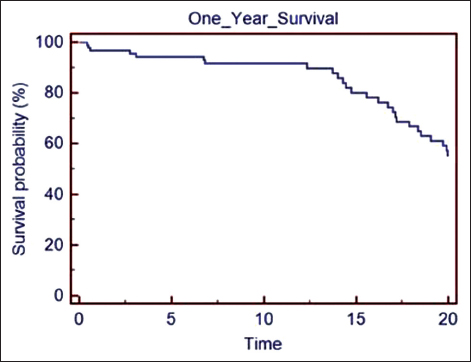
- Kaplan–Meier survival curve analysis of recipients of left ventricular assist device show a 1-year survival probability of 90% and a 20-month survival probability of 55%.
DISCUSSION
The results of this study demonstrate the benefit of utilizing CT imaging to assess infection spread. Driveline infections and abscess account for a significant amount of LVAD complications. In our study, 38.3% of patients had driveline infections and 18.8% developed abscesses. It has been proposed that LVAD therapy induces defects in the cellular and humoral immune system, making these individuals more vulnerable to infections.[181920]
Although the LVAD HeartMate II is increasingly being used, significant rates of infection have been reported. In the HeartMate II bridge to transplantation trial, 14% of patients had driveline infections. More recently, the HeartMate II destination therapy trial demonstrated higher rates of LVAD-related infection of 35%.[181920] Infectious complications result in significant morbidity and mortality. In our study, they accounted for 3 out of 9 (33.3%) devices requiring replacement and for 4 out of 25 deaths (16%).
Driveline infections occur more frequently than pump pocket infections.[20] However, since driveline infection can extend into the pump pocket and mediastinum, a better assessment of driveline infectious changes that can forecast deep LVAD infections are needed [Figure 6].
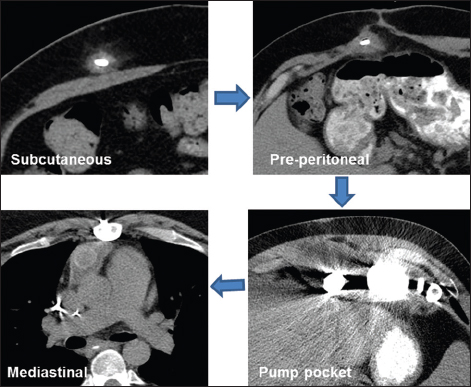
- Mechanism of spread of infection from the driveline to resultant abscess formation. Note the infection initially affects the driveline superficially in the subcutaneous tissue, then it extends into the preperitoneal space, and then it migrates up into the pump pocket and may also extend into the mediastinum.
CONCLUSION
This study shows that patients with infiltrative change size around the driveline of ≥14 mm are more prone to develop pump or mediastinal abscesses. Similarly, a preperitoneal location of infiltrative changes around the driveline predisposes to abscess formation more than just subcutaneous changes surrounding the driveline.
Thus, CT imaging could be used to measure the infiltrative change surrounding the driveline as a predictive measure for abscess formation in the pump pocket and/or mediastinum.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgment
We acknowledge Dr. Vikram Dogra for reviewing the manuscript.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/43/192835
REFERENCES
- Heart disease and stroke statistics-2013 update: A report from the American Heart Association. Circulation. 2013;127:e6-245.
- [Google Scholar]
- Driveline infections in left ventricular assist devices: Implications for destination therapy. Ann Thorac Surg. 2012;94:1381-6.
- [Google Scholar]
- Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med. 2001;345:1435-43.
- [Google Scholar]
- Clinical management of continuous-flow left ventricular assist devices in advanced heart failure. J Heart Lung Transplant. 2010;29:1081.
- [Google Scholar]
- HeartMate II left ventricular assist system: From concept to first clinical use. Ann Thorac Surg. 2001;71(3 Suppl):S116-20.
- [Google Scholar]
- Hemorrhage and thrombosis with different LVAD technologies: A matter of flow? Ann Cardiothorac Surg. 2014;3:582-4.
- [Google Scholar]
- Technique for minimizing and treating driveline infections. Ann Cardiothorac Surg. 2014;3:557-62.
- [Google Scholar]
- Current concepts in the diagnosis and management of left ventricular assist device infections. Expert Rev Anti Infect Ther. 2013;11:201-10.
- [Google Scholar]
- Use of a continuous-flow device in patients awaiting heart transplantation. N Engl J Med. 2007;357:885-96.
- [Google Scholar]
- Infection in permanent circulatory support: Experience from the REMATCH trial. J Heart Lung Transplant. 2004;23:1359-65.
- [Google Scholar]
- Results of extended bridge to transplantation: Window into the future of permanent ventricular assist devices. Ann Thorac Surg. 1996;61:396-8.
- [Google Scholar]
- Infection after implantation of pulsatile mechanical circulatory support devices. J Thorac Cardiovasc Surg. 2010;139:1632-6.
- [Google Scholar]
- Infectious complications after pulsatile-flow and continuous-flow left ventricular assist device implantation. J Heart Lung Transplant. 2011;30:164-74.
- [Google Scholar]
- Effect of left ventricular assist device infection on post-transplant outcomes. J Heart Lung Transplant. 2009;28:237-42.
- [Google Scholar]
- Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361:2241-51.
- [Google Scholar]
- Infectious complications in patients with left ventricular assist device: Etiology and outcomes in the continuous-flow era. Ann Thorac Surg. 2010;90:1270-7.
- [Google Scholar]
- Left ventricular assist device-related infections: Past, present and future. Expert Rev Med Devices. 2011;8:627-34.
- [Google Scholar]






