Translate this page into:
Squamous Cell Carcinoma of the Renal Pelvis Associated with Kidney Stones: Radiologic Imaging Features with Gross and Histopathological Correlation
Address for correspondence: Dr. Ozlem Tugce Kalayci, Department of Radiology, Izmir Ataturk Training and Research Hospital, Izmir, Turkey. E-mail: doktorozlemtugce@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Squamous cell carcinoma (SCC) of the renal pelvis is a rare neoplasm and is usually associated with long standing renal stone disease. This tumor is aggressive in nature and usually has a poor prognosis. We report a case who presented with sudden significant weight loss. During the radiologic investigation, a renal mass and staghorn calculi were detected in the right kidney. The patient subsequently underwent right radical nephrectomy. Pathological diagnosis was SCC of renal pelvis with extensive infiltration in to the renal parenchyma. The radiologic imaging features and histopathologic findings of this rare tumor are discussed in this report.
Carcinoma de células escamosas de la pelvis renal asociado a nefrolitiasis: Características radiológicas y correlación histopatológica
El carcinoma de células escamosas (CCE) de la pelvis renal es una neoplasia rara y usualmente asociada a nefrolitiasis de larga evolución. Este tumor de naturaleza agresiva usualmente tiene un pobre pronóstico. Reportamos un caso que se presentó con pérdida súbita de peso. Durante la valoración radiológica se encontró una masa renal con un cálculo coraliforme en el riñón derecho realizándose una nefrectomía radical. El diagnóstico patológico fue de CCE de la pelvis renal con infiltración extensiva al parénquima renal. Las características por Imagen y los hallazgos histopatológicos de este raro tumor son discutidos en este reporte de un caso
INTRODUCTION

Primary renal squamous cell carcinoma (SCC) constitutes less than 1% of all urinary tract neoplasms. Renal SCC is more frequently reported in the urinary bladder and male urethra than in the renal pelvis. Although this malignancy is rare in the upper urinary tracts, this diagnosis should be included in the differential diagnosis when evaluating a renal mass that is associated with renal calculi.[1–3] We report a case of incidentally detected renal SCC associated with staghorn disease in a patient having no history of urinary disease. Ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI) characteristics are discussed along with histopathologic findings.
A 63-year-old male patient presented to our institute with 10 kg weight loss in twenty days. Examination of the abdomen was unremarkable. He had a serum urea level of 12 mg/dL and serum creatinine level of 0.8 mg/dL. Hematological and biochemical parameters including renal function tests were within normal limits. He had no history of hematuria, pyuria, or prior surgery.
RADIOLOGICAL FEATURES
Ultrasound (US) evaluation showed intraparenchymal hypoechoic solid mass approximately 4.9 cm × 5.1 cm in size with lobulated contour extending to the upper pole of the kidney with absence of corticomedullary distinction [1a–b]. There was no significant vascularity in the mass on color flow Doppler imaging. There were staghorn stones in the right renal pelvicalyceal system; however, no dilatation of the renal pelvis and calyces. Renal CT revealed a big, non-functioning right kidney with staghorn calculi and a hypodense mass within the renal parenchyma extending to the upper pole of the right kidney. The metastatic work-up was negative, and the renal malignancy was diagnosed as Stage III (pT3N0M0). A contoured homogenous isointense mass lesion was detected in the upper pole, which was seen invading the renal sinus on T1-weighted imaging. T2-weighted imaging revealed a heterogenous hyperintense mass with some hypointense areas. The mass showed minimal enhancement after administration of intravenous gadolinium contrast medium. There were multiple milimetric hypointense foci in all sequences compatible with stones or calcifications [1c–1e]. Subsequently, patient underwent the right radical nephrectomy.
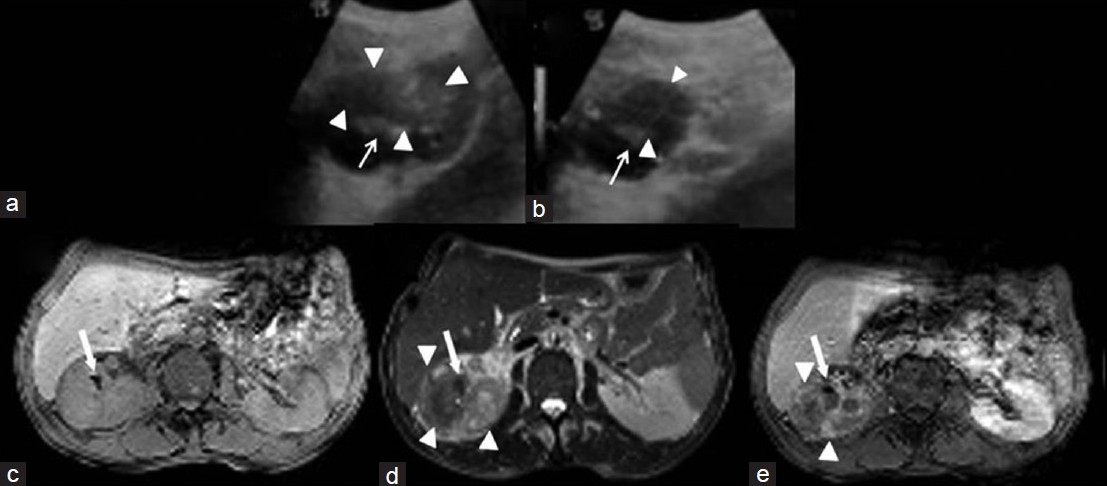
- (a) Coronal and (b) axial ultrasound (US) imaging show intraparenchymal hypoechoic heterogenous solid mass with lobulated contour in the upper-middle portion of right kidney (arrowheads). (c) Axial T1-weighted magnetic resonance image (MRI), (d) T2-weighted image (WI) show an increase in the size of the right kidney and reveal a heterogenous hyperintense mass (arrowheads). (e) Post-contrast T1-WI reveals a homogenous isointense mass lesion invading renal sinusparenchyma, and the mass shows minimal enhancement after administration of intravenous contrast medium and note hyperechoic foci on US imaging and hypointense foci in all MRI sequences compatible with stones (white arrows).
PATHOLOGICAL FEATURES
On gross examination of the nephrectomy specimen, the right kidney was found to be enlarged in size, measuring 14 cm × 11 cm × 9 cm. Cut section showed multiple yellow-brown colored staghorn stones. There was an irregular gray-white solid tumor replacing the renal parenchyma and renal pelvis. Surrounding renal parenchyma was not seen and the mass was found invading the perinephric and pelvic fat [Figure 2a and 2b]. Furthermore, in serial sections there was an irregular hemorrhagic area in the superior pole. Microscopic examination of the hematoxylin and eosin stained tissue revealed a poorly-differentiated SCC infiltrating the kidney parenchyma and perinephric-pelvic fat. Tumoral mass was invading the renal capsule, perirenal fat, and lymphovascular structures. There were a lot of necrosis within the mass [Figure 3a–c]. Pathological diagnosis was SCC of renal pelvis extensively infiltrating the renal parenchyma and perirenal fat. Most SCCs of the renal pelvis are moderately or poorly differentiated and hence the characteristics of SCC, such as keratin pearls and intracellular bridges, may not be readily apparent. SCCs are typically more invasive than most transitional cell carcinomas at diagnosis.[34]
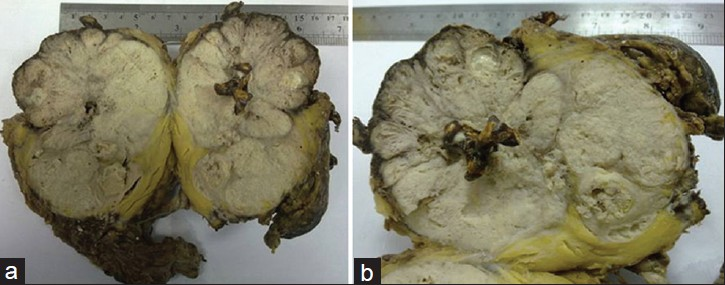
- Cut sections of the nephrectomy specimen (a and b) after the right radical nephroureterectomy show an irregular gray-white solid tumor in total kidney and multiple yellow-brown colored staghorn stones. Surrounding renal parenchyma is not visible and the mass is seen invading perinephric and pelvic fat
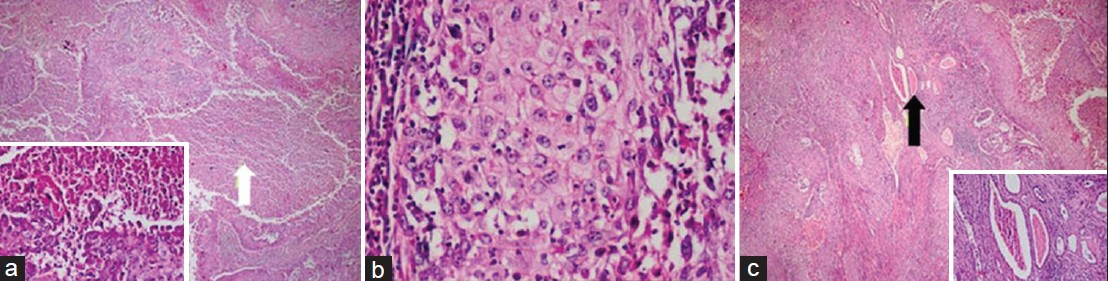
- Histopathology samples stained with H and E, ×40 (a, b, c) show poorly differentiated squamous cell carcinoma of renal pelvis, extensively infiltrating the renal parenchyma and perinephric fat. (a) Sample shows intensive necrotic areas within tumor islands (white arrow). (b) Sample reveals tumoral tissue with atypical squamous epithelial cells. (c) Image demonstrates presence of residual renal tubule cells within the tumor islands (black arrow).
DISCUSSION
Primary SCC of the renal pelvis is rare and represents only 0.5-0.8% of malignant renal tumors. In general, these tumors are highly aggressive in nature and are usually present at an advanced stage when detected and have a poor prognosis when compared to the other upper urinary tract malignancies.[15] Chronic irritation, inflammation, and infection induce squamous metaplasia of the renal collecting system, which may progress to dysplasia and carcinoma in some patients. Whether the occurrence of squamous metaplasia is due to the presence of the calculus that leads ultimately to the development of carcinoma or existence of SCC causes the formation of calculus is not clear yet.[67]
Radiologically, primary SCC of the renal pelvis may appear as a solid mass, with hydronephrosis, calcifications, or as a renal pelvic infiltrative lesion without evidence of a distinct mass. The radiologic differential diagnosis includes primary and secondary renal neoplasms and xanthogranulomatous pyelonephritis (XGP) associated with renal calculi. XGP is an uncommon form of chronic pyelonephritis, typically occurring as a result of chronic obstruction, which leads hydronephrosis, causing destruction of renal parenchyma. XGP is commonly associated with lithiasis however, rarely causes keratinizing squamous metaplasia and its manifestations closely mimic renal neoplasm, leading to misdiagnosis of malignancy.[3] In our case, there was a large infiltrative mass in the renal parenchyma without pelvicalyceal ectasia or any clinical symptoms of urinary infection. The radiologic findings of SCC are variable and may present with diffuse enlarged non-functional kidney with renal calculi, perirenal infiltration, and low density or echogenity in the renal parenchyma. The primary renal SCC cannot be distinguished from XGP or other malignant neoplasms of the kidney. A large transitional cell cancer with both renal pelvis and parenchymal involvement may present with similar CT and MR imaging findings. MR imaging features of renal SCC have been rarely described in literature. Recently, Imbriaco et al.,[8] reported renal SCC within a horseshoe kidney associated with renal stones with non-specific CT and MRI findings. The non-specific clinical and radiologic features in renal SCC may cause diagnostic confusion and histopathology is needed for confirmation.[2349]
Raghavendran et al.,[10] reported a case series of stones associated renal pelvic malignancies and they suggested that a patient with long standing stone disease and associated poor functional kidney or hematuria necessitates a screening CT. Lee et al.,[4] found that the most helpful features in CT of renal SCC was presence of enhancing extra-luminal and exophytic mass and in some cases, with an intraluminal component. They further suggested that as it is impractical to perform CT for every patient with renal stone, intravenous urography (IVU) should be carried out periodically, especially, in patients with long standing stones, and should be read as a split function test for all portions of renal parenchyma. Because the filling defects, delay in appearance of pyelogram, or renal parenchymal thickening in IVU may indicate a renal tumor despite the absence of mass effect and preservation of renal contour, warranting further studies.[1]
The fact that the urothelium normally does not have squamous cells renders the pathogenesis of these tumors interesting. The process is assumed to begin with an urothelial metaplasia resulting from a reaction to chronic irritation, leading to dedifferentiation, dysplasias, and in the end, to a SCC.[3] The relevant medical history include chronic episodes of pyelonephritis or nephrolithiasis.
Renal SCC usually presents at an advanced stage-pT3 or higher. Because of advanced stage at presentation, the prognosis is generally poor, as surgical resection is rarely curative and adjuvant chemoradiotherapy is usually ineffective. The prognosis is dismal with a 5-year survival rate of less than 10%.[39]
CONCLUSION
Primary renal SCCs are rare tumors and show a strong association with renal stones. Image features on conventional radiographic studies are non-specific. SCC should be suspected in any patient presenting with renal pelvis mass and history of renal calculi. CT and MRI play a crucial role in diagnosis and staging of these tumors.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2013/3/1/12/109725
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Incidentally detected squamous cell carcinoma of renal pelvis in patients with staghorn calculi: Case series with review of the literature. ISRN Oncol. 2011;2011:620574.
- [Google Scholar]
- Squamous cell carcinoma located in the renal caliceal system: A case report and review of the literature. Turk J Cancer. 2002;32:20-4.
- [Google Scholar]
- Rare coexistence of keratinizing squamous cell carcinoma with xanthogranulomatous pyelonephritis in the same kidney: Report of two cases. J Cancer Res Ther. 2010;6:339-41.
- [Google Scholar]
- Renal squamous cell carcinoma: CT findings and clinical significance. Abdom Imaging. 1998;23:203-8.
- [Google Scholar]
- Survival of patients with squamous cell malignancies of the upper urinary tract. Clin Med Insights Oncol. 2012;6:11-8.
- [Google Scholar]
- Squamous cell carcinoma of the renal pelvis and ureter: Incidence, symptoms, treatment and outcome. J Urol. 2007;178:51-6.
- [Google Scholar]
- Squamous cell carcinoma within a horseshoe kidney with associated renal stones detected by computed tomography and magnetic resonance imaging. Urology. 2011;78:54-5.
- [Google Scholar]





