Translate this page into:
Role of Perfusion CT Differentiating Hemangiomas from Malignant Hepatic Lesions
Address for correspondence: Dr. Jagjeet Singh, Department of Radiodiagnosis and Intervention Radiology, IGMC, Shimla - 171 001, Himachal Pradesh, India. E-mail: djagjeetsingh86@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objective:
The purpose of the study was to determine the role of computed tomography (CT) perfusion in differentiating hemangiomas from malignant hepatic lesions.
Materials and Methods:
This study was approved by the institutional review board. All the patients provided informed consent. CT perfusion was performed with 64 multidetector CT (MDCT) scanner on 45 patients including 27 cases of metastasis, 9 cases of hepatocellular carcinoma (HCC), and 9 cases of hemangiomas. A 14 cm span of the liver was covered during the perfusion study. Data was analyzed to calculate blood flow (BF), blood volume (BV), permeability surface area product (PS), mean transit time (MTT), hepatic arterial fraction (HAF), and induced residue fraction time of onset (IRFTO). CT perfusion parameters at the periphery of lesions and background liver parenchyma were compared.
Results:
Significant changes were observed in the perfusion parameters at the periphery of different lesions. Of all the perfusion parameters BF, HAF, and IRFTO showed most significant changes. In our study we found: BF of more than 400 ml/100 g/min at the periphery of the hemangiomas showed sensitivity of 88.9%, specificity of 83.3%, positive predictive value (PPV) of 57.1%, and negative predictive value (NPV) of 96.7% in differentiating hemangiomas from hepatic malignancy; HAF of more than 60% at the periphery of hemangiomas showed sensitivity of 77.8%, specificity of 86.1%, PPV of 58.3% and NPV of 93.9% in differentiating hemangiomas from hepatic malignancy; IRFTO of more than 3 s at the periphery of hemangiomas showed sensitivity of 77.8%, specificity of 86.1%, PPV of 58.3%, and NPV of 93.9% in differentiating hemangiomas from hepatic malignancy.
Conclusion:
Perfusion CT is a helpful tool in differentiating hemangiomas from hepatic malignancy by its ability to determine changes in perfusion parameters of the lesions.
Keywords
Hemangioma
hepatocellular carcinoma
liver
metastasis
perfusion
INTRODUCTION

Globally primary liver cancer is the fifth most frequently diagnosed cancer.[1] Early diagnosis is important to improve survival rates in the affected individuals. The liver has a dual blood supply and most liver diseases cause changes in blood flow (BF). Hence it is important to evaluate the hemodynamic changes to discover lesions early and assess therapeutic response.[23456] Currently, the perfusion imaging techniques with multi-slice computed tomography (CT) allow quantification of the perfusion parameters of tissues.[78]
As the liver plays an important role in blood circulation by acting as a filter, metastatic liver cancer occurs in over 75% of all terminal cancer patients.[910] Metastatic foci smaller than 10 mm are difficult to detect even with advanced imaging modalities. Thus tools are needed that provide earlier detection. Occult metastasis gives rise to detectable changes in liver BF similar to those caused by overt metastasis. CT techniques allow quantification of perfusion parameters of tissues allowing early detection of occult metastasis.[11121314]
Hepatic hemangiomas are small, asymptomatic lesions seen in all age groups often discovered incidentally in routine cross-sectional imaging. They are the second most common hepatic tumor, exceeded only by metastasis. Sometimes with conventional imaging techniques differentiation between hemangiomas and malignant hepatic lesions becomes difficult, especially in patients who have a known primary tumor.[15]
The recent development of multidetector CT (MDCT) allows fast data acquisition over a large anatomic area with isotropic voxels. The shorter scan times allow the capture of distinct phases, including the unenhanced phase, the arterial phase, and the portal venous phase. These phases provide important information concerning the enhancement patterns and hence the possibility of characterization of focal liver lesions. The improved temporal resolution of the newer faster MDCT systems also allow perfusion studies of the liver.[16171819] Perfusion imaging techniques with multi-slice CT allow quantification of the perfusion parameters of tissues and help in differentiating benign lesions from malignant lesions.[2021]
The purpose of the study was to determine the role of CT perfusion in differentiating hemangiomas from malignant hepatic lesions.
MATERIALS AND METHODS
Study design
This prospective study was approved by the institution review board. Forty-five patients (24 males, 21 females; mean age: 54 years) of which 27 cases of metastasis, 9 with hepatocellular carcinoma, and 9 with hemangiomas were included in this study. Informed consent was obtained from all the patients.
Inclusion criteria
Included in the study were patients with radiologically detected focal liver lesion on ultrasonography, indeterminate focal liver lesions on liver fine needle aspiration cytology, and focal liver lesions in known primary malignancy.
Exclusion criteria
Patients with known contrast reaction, patients with creatinine clearance less than 30 ml/min/1.73 m2, pregnant women, patients in the pediatric age group (<18 years) and patients with focal liver lesions confirmed as benign histopathologically prior to the study were excluded from the study.
Technique
CT perfusion of the liver was performed on a 64-slice CT scanner (GE light speed VCT Xte extra) with volume helical shuttle covering 14 cm of liver span. Patients were kept fasting for at least 6 h. Intravenous access (with 18 G cannula) was obtained through antecubital vein.
For initial localization of the lesion, a CT scan of the liver was obtained without contrast medium during a breath hold at the end of expiration. After the lesion localization, a 14-cm scan range for computed tomography perfusion (CTP) was selected to include the largest lesion area visible on non-contrast computed tomography (NCCT) localizing images.
A dynamic study of the liver was performed in quiet breathing. No motion correction software for abdomen was available in the MDCT scanner used. The acquisition was done in shallow and slow breathing, as it is not possible to hold the breath for acquisition time of 45 s to acquire 26 phases. Further external compression bands were used to minimize respiratory excurses. Similar protocols have been used for motion correction in other studies.[6]
A 26-phase helical CT examination of the total liver volume was acquired after rapid intravenous injection of 50 ml nonionic contrast agent at a rate of 4-5 ml/s followed by 20 ml of normal saline at a rate 4-5 ml/s using an automated injection device (Stellant, Medrad). Scanning was initiated 5 s after the start of the injection and images were acquired for a total duration of 45 s (one image per second).
All the CT examinations were performed with the following parameters: Detector collimation: 0.625-1.250 mm; Matrix: 512 × 512; Reconstruction intervals: 3.75-5.00 mm; Scan time: 50 s; Gantry rotation time: 0.4 s; Tube voltage: 80-100 kV; Tube current: 150-300 mAs/Auto mAs.
Data analysis
Images and data obtained were transferred to an image processing workstation. Perfusion maps of the liver were generated using software Perfusion CT 4 (GE) and Perfusion De-convolution technique.
To evaluate the perfusion parameters of focal lesions, unprocessed images were used. Region of interest (ROI) was drawn over the aorta and portal vein on the same slice showing peak enhancement in arterial and portal venous phases, respectively. In case of a single lesion, ROI was drawn at the center, at the edge of the lesion, whole lesion and normal-appearing background liver parenchyma for comparison. If two or more lesions were present, ROI were drawn along all the lesions of more than 20 mm in size and mean value from all the lesions were used for analysis. We selected the images in which the tumor demonstrated maximal enhancement. Two radiologists analyzed the data independently and the mean of their perfusion values was taken for further evaluation. Following parameters were evaluated: BF (in milliliters per 100 g of wet tissue per minute), blood volume (BV, in milliliters per 100 g of wet tissue), mean transit time (MTT, in seconds), permeability surface area product (PS, in milliliters per 100 g of wet tissue per minute), and IRFTO (seconds).
Functional maps were displayed in colors ranging from blue to red, blue being the lower range of display for BF, BV, PS, and hepatic arterial fraction (HAF) and red being the lower range of display for MTT and IRFTO [Figure 1].

- Functional maps. (a) CT image shows ROI drawn over aorta, portal vein, background liver parenchyma, at the periphery, center, and whole of the lesion. (b) Graph shows time intensity curves obtained. (c and d) Functional maps obtained for various perfusion parameters.
Statistical analysis
The mean, standard error of mean and range of various perfusion parameters were calculated at the center, periphery, whole lesion and in the background liver parenchyma. Comparison of various perfusion parameters at the periphery, center, and whole lesion with background liver parenchyma was done by means of unpaired t-test. A P ≤ 0.05 was considered to denote statistical significance. Sensitivity, specificity, positive predictive value (PPV) and negative predictive values (NPV) for various perfusion parameters were calculated.
RESULTS
From July 2011 to September 2012, a total of 50 consecutive patients (26 males and 24 females; mean age: 54 years) met the selection criteria and were enrolled in the study. A total of 27 patients had liver metastasis, 9 had HCC, and 9 had hemangiomas. Of the remaining five patients, three patients who had no focal liver lesions, and two patients in whom histopathological reports were inconclusive were excluded from the study. Fine needle aspiration cytology (FNAC) or histopathology was done in all cases, except in cases of HCC having alfa-feto protein level more than 400 (ng/mL), cases showing arterial phase enhancement with washout in portal venous phase fulfilling European Association for the Study of the Liver (EASL) criteria, and in cases of hemangiomas that showed no change in lesion characteristics on follow-up CT scans.
Perfusion parameters in background liver parenchyma
Perfusion parameters of background liver parenchyma were calculated in all cases. Perfusion parameters at the periphery, center, and whole lesions were compared with background liver parenchyma. Significant differences were seen in parameter values in the lesions when compared to background liver parenchyma readings. [Table 1].

Perfusion parameters in hemangiomas
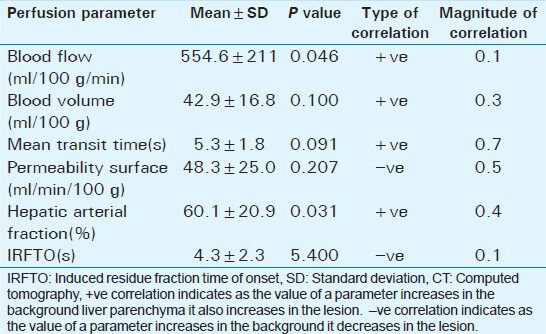
There was significant change in BF and HAF at the periphery of hemangiomas when compared to background liver parenchyma (P < 0.05). No significant change was seen in BV, MTT, PS, and IRFTO at the periphery of lesions when compared with background liver parenchyma values. BF was more than 400 ml/100 g/min in 88.9% (8 cases) of hemangiomas. HAF was more than 60% in 7 cases (77.8%) of hemangioma. The various perfusion parameters at the periphery of lesions were correlated with perfusion parameters in the background normal liver parenchyma.[Figure 2 and Table 2].
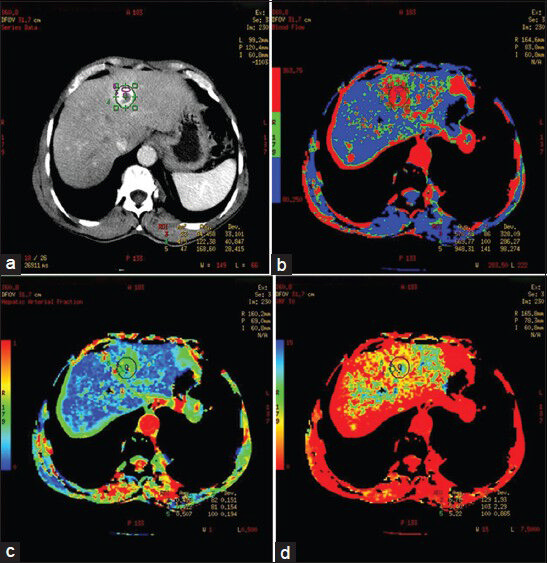
- 56-year-old male diagnosed with hemangioma, who was a follow up case of cancer of sigmoid colon. (a) Postcontrast CT image shows intense peripheral nodular enhancement of lesion. Perfusion maps at the periphery of the lesion (b) BF map shows increase in BF (948.3 ml/100 g/s), (c) HAF map shows increase in BV (50.7 ml/100 g) and d) IRFTO map shows decrease in MTT (5.2 s).
Perfusion parameters in HCC
There was significant increase in BF and PS with decrease in MTT and IRFTO at the periphery of HCC as compared with background liver parenchyma (P < 0.05). No significant change was seen in BV and HAF [Figure 3 and Table 3].

- 45-year-old female who presented with mass in right hypochondrium. diagnosed with Fibrolamellar hepatocellular carcinoma. (a) Postcontrast CT, (b) BF, (c) MTT and (d) HAF maps of the liver. Postcontrast CT shows heterogeneous enhancement of exophytic lesion in right lobe of liver with central necrosis. Perfusion maps show increased BF (407.1 ml/100 g/s), BV (41.3 ml/100 g), and HAF (33.7) at the periphery of lesion as compared with background liver

Perfusion parameters in liver secondaries
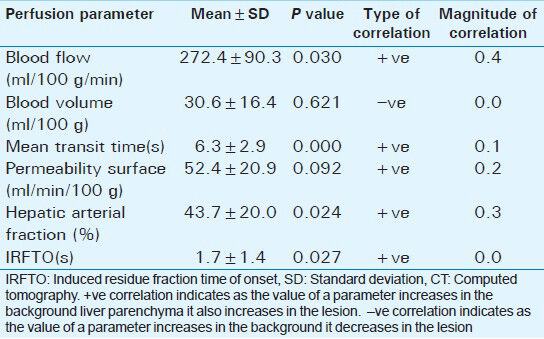
Of the 27 cases of secondaries, 20 cases were hypervascular, and 7 were hypovascular in arterial phase of unprocessed perfusion images. Hypovascular secondaries were seen in three cases of cancer of gall bladder, two cases of cancer of breast, and two cases with unknown primary, whereas hypervascular secondaries were seen in eight cases of cancer of colon, four cases of cancer of breast, three cases of cancer of lung, four cases of cancer of cervix, and one case of cancer of thyroid. All cases of secondaries except for one case with unknown primary showed significant increase in BF and HAF with significant decrease in MTT and IRFTO at the periphery as compared with background liver parenchyma (p < 0.05). No significant change was seen in BV and PS [Figure 4]. It can be concluded that distinction cannot be made between hyper– and hypovascular secondaries based on only CT perfusion parameter values and it is recommended that unprocessed images of all the 26 phases should also be taken into consideration for better characterization [Table 4].
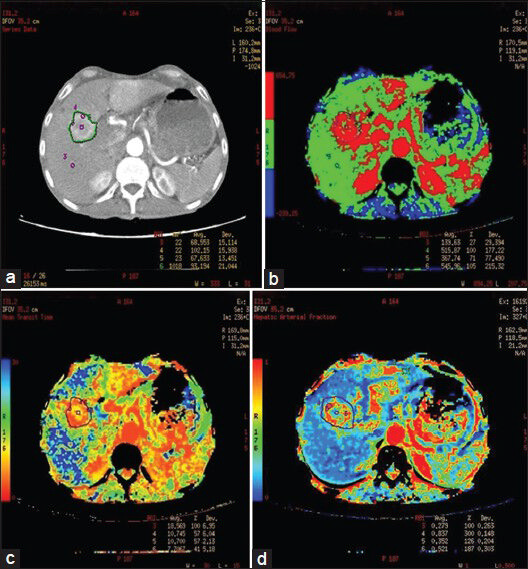
- 60-year-old male who was a known case of cancer stomach with secondaries in the liver. (a) Postcontrast CT, (b) BF, (c) MTT and (d) HAF maps of the liver. Postcontrast CT shows peripheral rim enhancement of lesion with central necrosis. Perfusion maps show increased BF (515.8 ml/100 g/s) and HAF (83.7%) with decreased MTT (10.7 s) at the periphery of lesion as compared with background liver.
DISCUSSION
We evaluated the role of CT perfusion on 64-slice MDCT in determining the change in perfusion parameters that differentiate hemangiomas from malignant hepatic lesions. We observed significant differences between CT perfusion values of malignant hepatic lesions and hemangiomas. Perfusion CT has emerged recently as a means of rapid, noninvasive assessment not only of the anatomy, but also of physiology, as is evident in its ability to reliably measure BF. MDCT is fast, economical, and widely available, all of which contribute to further interest in perfusion CT techniques and their future implementation in the clinical arena.[2223]
In the past, many investigators determined the role of CT perfusion in HCC and liver metastasis, however, very few studies have been published determining the role of CT perfusion in differentiating HCC from hemangiomas. To the best of our knowledge, there is no published study determining the role of perfusion CT in differentiating secondaries from hemangiomas.
In 2008, Zang et al., studied the role of perfusion CT in HCC and showed increase in BF, BV, and PS with decrease in MTT.[24] Perfusion values in our study are comparable with values given Zang in his study [Table 5].
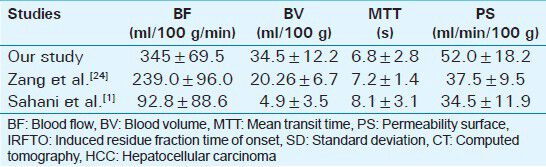
Sahani et al.,[1] performed dynamic computed tomography perfusion (CTP) in HCC and showed significant change in perfusion parameters in HCC as compared with background liver parameters. Our study also showed significant change in perfusion parameters in HCC as compared with background liver. Our perfusion values for BF, BV, and PS are higher and for MTT are lower as compared with values calculated by Sahani et al. This difference in parameters may be due to differences in technique and/or different algorithms used to obtain perfusion parameters as well as different sets of population. Sahani et al., used only aorta as input vessel, whereas we used both aorta and portal vein as input vessels. We conducted our study on patients from Asia whereas Sahani's patients were from USA.
In the past, many investigators studied the role of perfusion CT in liver metastasis and showed increase in perfusion parameters at the periphery of metastasis. Our values are comparable with those of Jiang et al.,[9] but are different from those of Guyennon et al.,[25] this difference may be due to different techniques and algorithm used for perfusion [Table 6].

Wang et al., studied perfusion parameters in hemangiomas and showed increase in HAF at the periphery of hemangiomas.[26] In our study, we observed increase in BF, BV, PS, and HAF with decrease in MTT at the periphery of hemangiomas. In our study, perfusion data was acquired for only 45 s and centripetal filling in was demonstrated in subsequent delayed contrast study done at 15 min to cover the whole liver and abdomen. Perfusion scanning was not done in delayed phase to reduce radiation dose to patient. The decrease in MTT can be explained as we compared perfusion values at the periphery of lesions, which is comprised of small capillaries as compared with dilated bigger vascular spaces in the center of the lesion. Second, most hemangiomas in our study were small and small hemangiomas shows atypical hemodynamics with diffuse enhancement in arterial phase and with transient hepatic attenuation difference in surrounding liver parenchyma due to arterio-portal shunting.[272829] Most cases of hemangiomas in our study showed early arterial enhancement with washout in surrounding liver parenchyma. Inclusion of this surrounding liver parenchyma in ROI may be reason for low MTT values. No study to date, in our knowledge, has been carried out to show MTT values in hemangiomas. Further research is required to ascertain routine use of these values.
There was significantly greater increase in BF at the periphery of hemangiomas as compared with HCC and metastasis. HAF also showed significantly more increase at the periphery of hemangiomas as compared with HCC and metastasis. IRFTO was significantly decreased at the periphery of HCC and metastasis, however, in cases of hemangiomas, no significant change was seen as compared with background liver parenchyma. No significant difference was seen in BV, MTT, and PS at the periphery of various lesions. BF, HAF, and IRFTO showed significant changes and may help in differentiating hemangiomas from malignant hepatic lesions. Wang et al.,[26] showed the significance of HAF in differentiating hemangioma from malignant hepatic lesions. There is no study, to our knowledge, that shows the significance of other perfusion parameters in differentiating hemangiomas from malignant hepatic lesions. Further research is required to evaluate the role of other perfusion parameters.
Cut-off values
In our study, BF more than 400 ml/100 g/min at the periphery of the hemangiomas showed sensitivity of 88.9%, specificity of 83.3%, PPV of 57.1%, and NPV of 96.7% in differentiating hemangiomas from hepatic malignancy. Also HAF more than 60% at the periphery of hemangiomas showed sensitivity of 77.8%, specificity of 86.1%, PPV of 58.3%, and NPV of 93.9% in differentiating hemangiomas from hepatic malignancy. Similarly IRFTO more than 3 s at the periphery of hemangiomas showed sensitivity of 77.8%, specificity of 86.1%, PPV of 58.3%, and NPV of 93.9% in differentiating hemangiomas from hepatic malignancy.
Limitations
There were a number of limitations to our study. First, the sample size in the study was small. Second, for comparison only limited data was available of CT perfusion values in hemangiomas. Third, radiation dose in CT perfusion is higher when compared with radiation dose in conventional CT. Most hemangiomas in our study were small iin size (< 2 cm).
Reproducibility of perfusion parameters
In our study, all data was analyzed by two radiologist independently. To evaluate intraobserver variability same reader analyzed each patient twice. Values are calculated by the same observer twice at diff erent times. Coefficient of variation was derived for each perfusion parameter to evaluate inter- and intra-observer variability. Coefficient of variability for intraobserver variability for various perfusion parameters are: BF - 3.2%, BV - 4.1%, PS - 4.3%, MTT - 12.4%, HAF - 3.3%, and IRFTO - 3.6%. Coefficient of variability for interobserver variability for various perfusion parameters are: BF - 7.2%, BV - 8.4%, PS - 8.8%, MTT - 25.4%, HAF - 9.2%, and IRFTO - 9.6%. Coefficient of variation is highest for MTT both in intra- and inter-observer readings. Coefficient of variation is lower in liver protocol as used in our study compared to body protocol used by other authors.[27] Our values for coefficient of variation for various perfusion parameters are comparable to values given by Chaan et al., in 2012.[30] If we choose the same area of lesion for calculation of perfusion parameters at different times, the variation in values is less and if we choose different areas for calculation of perfusion parameters at different times, the variation in values is greater. CT perfusion is a reproducible technique with acceptable intra- and inter-observer variation.
CONCLUSION
Our initial experience suggests that CT perfusion is a noninvasive, quantifiable, and feasible technique, which can be helpful in differentiating hepatic hemangiomas from malignant liver lesions by determining changes in perfusion parameters of the lesions. It should be implemented in routine preoperative and follow-up examination of patients for differentiating hemangiomas from malignant liver lesion. Single CT scan can provide both morphological and functional images. Perfusion imaging of the liver has the potential to improve detection and characterization of liver lesions beyond that enabled by conventional imaging techniques. Limitation of perfusion CT is its increased radiation exposure. Future development in this field can lead to development of low dose protocols and improve our understanding of tumor physiology.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/10/127959
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Advanced HCC CT perfusion of liver and tumor tissue: Initial experience. Radiology. 2007;243:736-43.
- [Google Scholar]
- Body perfusion CT: Technique, clinical applications, and advances. Radiol Clin North Am. 2009;47:161-78.
- [Google Scholar]
- Hepatocellular carcinoma treated with transarterial chemoembolization: Dynamic perfusion-CT in the assessment of residual tumor. World J Gastroenterol. 2010;16:5993-6000.
- [Google Scholar]
- Computer tomography perfusion in evaluating the therapeutic effect of transarterial chemoembolization for hepatocellular carcinoma. World Gastroenterol. 2008;14:5738-43.
- [Google Scholar]
- Functional CT for quantifying tumor perfusion in antiangiogenic therapy in a rat model. Radiology. 2005;237:151-8.
- [Google Scholar]
- Perfusion CT for the assessment of tumour vascularity: Which protocol? Br J Radiol. 2003;76(Spec No 1):S36-42.
- [Google Scholar]
- Hepatic perfusion parameters in chronicliver disease: Dynamic CT measurements correlated with disease severity. Am J Roentgenol. 2001;176:667-73.
- [Google Scholar]
- Quantitative perfusion map of malignant liver tumors, created from dynamic computed tomography data. Acad Radiol. 2004;11:215-23.
- [Google Scholar]
- Functional CT for assessment of early vascular physiology in liver tumors. Hepatobiliary Pancreat Dis Int. 2008;7:497-502.
- [Google Scholar]
- Opportunities for near-infrared thermal ablation of colorectal metastases by guanylyl cyclise C-targeted gold nanoshells. Future Oncol. 2006;2:705-16.
- [Google Scholar]
- Hepatic haemodynamics: Interrelationships between contrast enhancement and perfusion on CT and Doppler perfusion indices. Br J Radiol. 2002;75:17-23.
- [Google Scholar]
- Early changes in liver perfusion caused by occult metastasis in rats: Detection with quantitative CT. Radiology. 2001;218:556-61.
- [Google Scholar]
- Hepatic metastases: The value of quantitative assessment of contrast enhancement on computed tomography. Eur J Radiol. 1999;30:206-13.
- [Google Scholar]
- Imaging diagnosis of hepatic metastases of pancreatic carcinomas: Significance of transient wedge-shaped contrast enhancement mimicking arterioportal shunt. Abdom Imaging. 2008;33:437-43.
- [Google Scholar]
- Clinical application of hepatic CT perfusion. World J Gastroenterol. 2009;15:907-11.
- [Google Scholar]
- Cerebral blood flow determination by rapid-sequence computed tomography. Radiology. 1980;137:679-86.
- [Google Scholar]
- Perfusion quantitation by ultrafast computed tomography. Invest Radiol. 1992;27:18-21.
- [Google Scholar]
- Measurement of myocardial perfusion and cardiac output using intravenous injection methods by ultrafast (cine) computed tomography. Invest Radiol. 1992;27:40-6.
- [Google Scholar]
- Contrast bolus dynamic computed tomography for the measurement of solid organ perfusion. Invest Radiol. 1993;28:72-7.
- [Google Scholar]
- Feasibility of integrated CT-liver perfusion in routine FDG-PET/CT. Abdom Imaging. 2010;35:528-36.
- [Google Scholar]
- Perfusion CT for assesment of tumor vascularity: Which protocol? Br J Radiol. 2003;76:36-42.
- [Google Scholar]
- Treatment of hepatocellular carcinoma by interventional methods. Eur Radiol. 2001;11:2207-19.
- [Google Scholar]
- Epidemiology of hepatocellular carcinoma in areas of low hepatitis B and hepatitis C endemicity. Oncogene. 2006;25:3771-7.
- [Google Scholar]
- Haemodynamic study of primary hepatocellular carcinoma evolved from viral-induced cirrhosis using CT perfusion imaging. J South Med Univ. 2008;11:1986-9.
- [Google Scholar]
- Perfusion characterization of liver metastases from endocrine tumors: Computed tomography perfusion. World J Radiol. 2010;2:449-54.
- [Google Scholar]
- Application of CT perfusion imaging in discrimination of liver carcinoma and haemangiomas. Linchuang Gandan Bing Zazhi. 2006;22:455-7.
- [Google Scholar]
- Hepatic hemangiomas with arterioportal shunt: Findings at two-phase CT. Radiology. 2001;219:707-11.
- [Google Scholar]
- Hepatic cavernous hemangioma: Temporal peritumoral enhancement during multiphase dynamic MR imaging. Radiology. 2000;216:692-7.
- [Google Scholar]
- Cavernous Hemangioma of the liver: Pathologic correleation with dynamic CT findings. Radiology. 1997;203:121-5.
- [Google Scholar]
- Effect of dual vascular input functions on CT perfusion parameter values and Reproducibility in tumors and normal liver. J Comput Assist Tomogr. 2012;36:388Y393.
- [Google Scholar]






