Translate this page into:
Quantification of Cardiac Output with Phase Contrast Magnetic Resonance Imaging in Patients with Pulmonary Hypertension

*Corresponding author: Jose Ricardo Po,Borgess Heart Institute, Ascension Borgess Hospital, 1722 Shaffer St., Kalamazoo, MI, United States. josericardo.po@ascension.org
-
Received: ,
Accepted: ,
How to cite this article: Po JR, Tong M, Meeran T, Potluri A, Raina A, Doyle M, et al. Quantification of cardiac output with phase contrast magnetic resonance imaging in patients with pulmonary hypertension. J Clin Imaging Sci 2020;10:26.
Abstract
Objective:
The purpose of the study is to compare phase contrast (PC) imaging with invasive measurements of cardiac output (CO) in patients with pulmonary hypertension (PH).
Materials and Methods:
We analyzed 81 cases with PH who underwent cardiac magnetic resonance imaging and right heart catheterization (RHC). Measurement of CO and stroke volume (SV) by cardiac magnetic resonance (CMR) was performed by PC imaging of the proximal aorta (Ao) and pulmonary artery (Pa) and by RHC using the Fick and thermodilution (TD) methods.
Results:
There was good correlation in CO measurements between PC and RHC; however, there was better correlation with SV measurements; Fick-TD (r=0.85), PC-TD (Ao r=0.77, Pa r=0.79), and PC-Fick (Ao r = 0.73, Pa r = 0.78). Bland-Altman analysis of SV showed that Pa PC had slightly lower standard deviation than Ao PC; PC-Fick (Pa SD = 15.11 vs. Ao SD = 16.4 ml) and PC-TD (Pa SD = 16.99 ml vs. Ao SD = 17.4 ml) while Fick-TD had the lowest (SD = 14.4 ml). Compared to Fick, measurement of SV with Ao PC (‒4.12 ml) and Pa PC (0.22 ml) both had lower mean difference than TD (‒11.1 ml).
Conclusion:
Non-invasive measurement of CO and SV using PC-CMR correlates well with invasive measurement using RHC. Our study showed that PC-CMR had high accuracy and precision when compared to Fick. Among all the modalities, PC-CMR contributed the least amount of variation in measurements.
Keywords
Cardiac output
Stroke volume
Pulmonary hypertension
Phase contrast imaging
Cardiac magnetic resonance imaging
INTRODUCTION
Cardiac output (CO) is a strong diagnostic and prognostic indicator of cardiovascular morbidity and mortality in a variety of disease states including left-sided congestive heart failure and pulmonary arterial hypertension (PAH).[1-3] Since the practical utility of the thermodilution (TD) and Fick method has been mainstreamed by the pulmonary artery (Pa) catheter in the 1970s, it has remained the most reliable method for measuring CO. However, despite widespread adoption, confounders include the practicality of measuring oxygen consumption, valvular regurgitation, and technical variabilities. This is also an invasive test with a variety of potential complications, albeit minimal, including bleeding/hematomas, pneumothorax, and cardiac arrhythmias.[4] Finally, expertise is required to interpret the data, as structural cardiac and respiratory factors can mislead the information obtained.[5]
Thus, there have been considerable efforts to non- invasively reproduce cardiac catheterization measurements. Echocardiography has been one of these techniques to estimate CO; most commonly utilizing pulsed Doppler across the left ventricular outflow tract (LVOT) multiplied by the LVOT area and heart rate.[6] Potential problems with these techniques include assumption of a circular LVOT and requirement of parallel alignment of the pulsed Doppler signal.
Cardiac magnetic resonance (CMR) phase contrast (PC) imaging is a non-invasive, rapid, and highly reproducible technique.[7,8] CO measured through PC was previously validated in a small cohort of 23 patients referred for both CMR and right heart catheterization (RHC) for a variety of indications.[9] However, oxygen consumption was measured in this cohort for utilization in the Fick equation, which is not practically available in the routine clinical setting. In pulmonary hypertension (PH), there is often confounding variable with respect to oxygen consumption such as age, sex, respiratory rate, and body size. However, this cohort of patients with PH and especially PAH has considerable need for serial CO determinations often requiring frequent catheterizations, especially during intensive medication titration. Naturally, it follows that if CO could be derived accurately and reproducibly in this population, numerous patient, and clinician benefits would ensue. Thus, the objective of our study is to determine the accuracy of CMR compared to invasive catheterization in measuring CO in patients with PH.
MATERIALS AND METHODS
Patients
Between January 2013 and January 2017, we retrospectively identified patients with a diagnosis of PH who underwent both CMR and RHC (<4 h apart). PH was defined by a mean Pa pressure of 25 mmHg at rest measured during RHC.[10] All patients were managed by the heart failure/PH service at our institution (Allegheny General Hospital, Pittsburgh, PA). All the tests were ordered as part of routine clinical management of the subjects and as such were not part of a research protocol. The study was approved as an ongoing outcomes research protocol by our institutional review board and patients signed a general consent release. Patients were excluded if they did not have PC performed during their CMR study or if there was significant intracardiac shunting (Qp: Qs <0.8 or >1.5). There was no funding source for this study.
CMR imaging
Standard CMR (1.5 T; General Electric, Milwaukee, WI) was performed on all the subjects. Patients were given anxiolytic medications before the examination as needed. Steady‐state free precession breath‐hold cines were performed (TR/ TE/flip angle 3.2/1.6/60; FOV 350 mm, matrix 224 × 224, scan percentage 100%) through contiguous 8 mm short‐ axis slices from the atrioventricular ring to apex, and in the two‐, three‐, and four‐ chamber long‐ axis views. Transverse stacks were also performed to evaluate both extracardiac and cardiac morphology and function. Ventricular function was measured using standard cardiac imaging software (Medis QMass, Medis, The Netherlands). PC imaging was performed in the proximal ascending aorta (Ao) and main Pa. The PC images were typically acquired using retrospective ECG gating under free breathing conditions (TR/TE/flip angle 7/3/20; slice thickness 8 mm, FOV 350 mm2, matrix 256 × 256, NEX = 2). Velocity encoding was initially set to 250 cm/s and was adjusted accordingly if aliasing occurred. Complete coverage of the cardiac cycle was accomplished using view sharing to acquire 60 cardiac phases per cycle. The segmentation factor of 4 was used for PC acquisition. Flow through the proximal ascending Ao and main Pa was measured offline using standard software (Medis QFlow, Medis, The Netherlands). Regions of interest were drawn to encircle the entire cross section of the vessel in the initial frame and were propagated to the remaining frames using an automated contouring function with manual override.
RHC
Standard RHC through an internal jugular vein approach was performed on all patients using a fluid filled 7 Fr catheter and pressure transducer. Minimal sedation using midazolam and/ or fentanyl was given under the discretion of the performing physician. The catheter was advanced into the Pa using fluoroscopic guidance. Standard pressure measurements were obtained in the right heart chambers. CO using the indirect (assumed) Fick method was calculated after determination of the oxygen saturation and hemoglobin. Oxygen consumption was estimated using K × BSA (body surface area) with K of 133 ml/min used for all subjects. The arterial saturation was obtained from finger oximetry and the venous saturation was obtained from a blood sample drawn from the distal port of the Swan-Ganz catheter. CO was also measured through TD method after injecting 10 ml of cold saline in the proximal port. For TD, five injections were performed per patient and values were averaged; values that differed by >10% were discarded as outliers. All CO measurements were performed using standard catheterization equipment and software (Philips Xper Flex Cardio, Philips Health, The Netherlands).
Statistical analysis
Continuous variables were recorded as mean ± standard deviation and categorical variables as proportions. All data underwent Kolmogorov–Smirnov testing to check for normality. Analysis of variance (ANOVA) was performed on the stroke volume (SV) measurement to determine presence of significant differences among the measures. Correlation coefficients of the CO measurement were calculated. Bland- Altman analysis was performed to assess the bias and differences between pairs of measurements.[11] Analysis was performed using Microsoft Excel (Microsoft, Redmond, Washington).
RESULTS
We analyzed 81 cases involving 72 patients. There were nine patients who had repeat testing (both CMR and RHC) who were included as separate measures. The demographic and baseline characteristics are shown in Table 1. The mean age was 60.7 ± 13.51 years (range: 24–84) and 49 (60%) were female. The majority of the cases (68%) belonged to the WHO Group 1 (PAH). The mean body mass index was 31.83 ± 7.65 kg/m2. The mean left ventricular ejection fraction was normal at 60.7 ± 9% and the mean right ventricular ejection fraction was mildly reduced 48.59 ± 12.66%. Comparison of the mean CO revealed that TD had the highest mean CO (5.97 ± 1.70 L/min), followed by Fick (5.21 ± 1.39 L/min), Pa PC (5.14 ± 1.55), and then Ao PC (4.86 ± 1.47).
| Characteristic | Value |
|---|---|
| Number of cases | 81 |
| Age, y | 60.7±13.51 |
| Female, n | 49 (60%) |
| BMI, kg/m2 | 31.83±7.65 |
| WHO classification | |
| Group 1 (PAH) | 55 (68%) |
| Group 2 (PHLHD) | 15 (19%) |
| Group 3 (PHRESP) | 5 (6%) |
| Group 4 (CTEPH) | 6 (7%) |
| LVEF, % | 60.72±9.02 |
| RVEF, % | 48.59±12.66 |
| MRI hemodynamics | |
| HR, bpm | 71.20±12.64 |
| MAP, mmHg | 101.44±15.76 |
| Ao PC SV, ml | 69.05±21.48 |
| Ao PC CO, L/min | 4.86±1.47 |
| Pa PC SV, ml | 73.4±22.56 |
| Pa PC CO, L/min | 5.14±1.55 |
| RHC hemodynamics | |
| HR, bpm | 73.13±12.98 |
| MAP, mmHg | 99.26±19.64 |
| RAP, mmHg | 8.0±4.84 |
| mPAP, mmHg | 41.9±13.16 |
| Fick SV, ml | 73.17±12.98 |
| Fick CO, L/min | 5.21±1.39 |
| TD SV, ml | 84.26±27.53 |
| TD CO, L/min | 5.97±1.70 |
Ao: Aorta, BMI: Body mass index, CO: Cardiac output, CTEPH: Chronic thromboembolic pulmonary hypertension, HR: Heart rate, LVEF: Left ventricular ejection fraction, MAP: Mean arterial pressure, mPAP: Mean pulmonary artery pressure, MRI: Magnetic resonance imaging, Pa: Pulmonary artery, PAH: Pulmonary arterial hypertension, PH-LHD: Pulmonary hypertension due to left heart disease, PH-RESP: Pulmonary hypertension due to respiratory disorders, RAP: Right atrial pressure, RHC: Right heart catheterization, RVEF: Right ventricular ejection fraction, SV: Stroke volume, TD: Thermodilution, WHO: World Health Organization
Bland-Altman analysis of CO [Figure 1] showed that Ao PC versus Fick (Ao PC-Fick) had a mean CO difference of ‒0.35 L/min (SD 1.12 L/min), Ao PC versus TD (Ao PC-TD) had a mean CO difference of ‒1.12 L/min (SD 1.06 L/min), Pa PC versus Fick (Pa PC-Fick) had a mean CO difference of ‒0.07 L/min (SD 1.21 L/min), and Pa PC versus TD (Pa PC-TD) had a mean CO difference of ‒0.84 L/min (SD 1.18 L/min). Comparison of CO between Fick and TD (Fick-TD) showed a mean CO difference of ‒0.76 L/min (SD 1.05 L/min). The correlation coefficients were similar among the different comparisons of CO: PC-Fick (Ao r = 0.70, Pa r = 0.67), PC-TD (Ao r = 0.79, Pa r = 0.74), and Fick-TD (r = 0.79). The PC- TD correlations were higher than the PC-Fick correlations and were closer to the r value between Fick-TD.
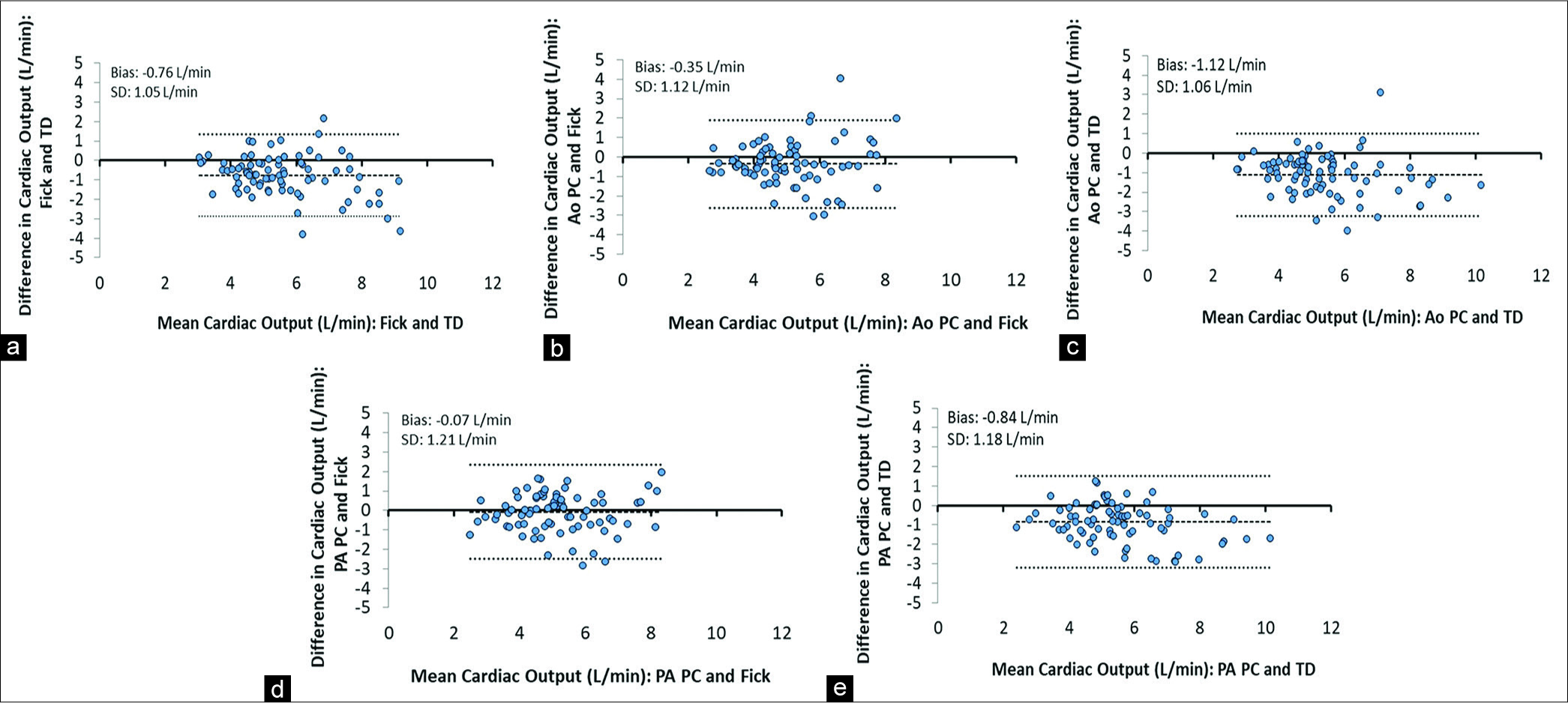
- Bland-Altman plot of difference in cardiac output; (a) Fick equation and thermodilution. (b) Aortic phase contrast imaging and Fick equation. (c) Aortic phase contrast imaging and thermodilution. (d) Pulmonary artery phase contrast imaging and Fick equation. (e) Pulmonary artery phase contrast imaging and thermodilution.
SV is the key variable in deriving CO once HR is constant. For this reason, we focused on SV measurement. Bland-Altman analysis of SV [Figure 2] showed that Pa PC had slightly lower standard deviation than Ao PC. In the SV analysis, Ao PC-Fick had a mean difference of ‒4.12 ml (SD 16.4 ml); Ao PC-TD had a mean difference of ‒15.2 ml (SD 17.4 ml); Pa PC-Fick had a mean difference of 0.22 ml (SD 15.11 ml); Pa PC-TD had a mean difference of ‒10.86 ml (SD 16.99 ml); and Fick-TD had a mean difference of ‒11.1 ml (SD 14.4 ml). There was better correlation between SV measurements compared to CO measurements; PC-Fick (Ao r = 0.73, Pa r = 0.78), PC-TD (Ao r = 0.77, Pa r = 0.79), and Fick-TD (r = 0.85).

- Bland-Altman plot of difference in mean stroke volume; (a) Fick equation and thermodilution. (b) Aortic phase contrast imaging and Fick equation. (c) Aortic phase contrast imaging and thermodilution. (d) Pulmonary artery phase contrast imaging and Fick equation. (e) Pulmonary artery phase contrast imaging and thermodilution.
Because Pa PC had consistently lower mean differences and standard deviation and higher correlation compared to Ao PC, we focused further analysis on Pa PC. To determine if other clinical factors affected SV measurement, we divided the patients into subgroups depending on the BMI, CO, right ventricular ejection fraction (RVEF), left ventricular ejection fraction (LVEF), and PH group [Tables 2 and 3]. The mean difference among all the subgroup comparisons showed that Pa PC-Fick had generally lower mean differences and very close standard deviations (mean of 0.86 ml difference among all groups) when compared to Fick-TD.
| Pa PC versus Fick | Pa PC versus TD | Fick versusTD | |
|---|---|---|---|
| All patients (n=81) | 0.78 | 0.78 | 0.85 |
| Nonobese BMI (n=35, <30 kg/m2) | 0.87 | 0.82 | 0.85 |
| Obese (n=46, ≥30 kg/m2) | 0.71 | 0.75 | 0.84 |
| Low CO (n=205, <4 L/min) | 0.79 | 0.76 | 0.83 |
| Normal CO (n=61, >4 L/min)() | 0.73 | 0.71 | 0.84 |
| Decreased LVEF (n=25, <57%) | 0.81 | 0.83 | 0.83 |
| Normal LVEF (n=56, ≥57%) | 0.77 | 0.76 | 0.86 |
| Decreased RVEF (n=42, <52%) | 0.87 | 0.79 | 0.89 |
| Normal RVEF (n=39, ≥52%) | 0.61 | 0.75 | 0.80 |
| Group 1 PH (n=55) | 0.81 | 0.86 | 0.79 |
| Nongroup 1 PH (n=26) | 0.75 | 0.69 | 0.95 |
BMI: Body mass index, CO: Cardiac output, LVEF: Left ventricular ejection fraction, Pa: Pulmonary artery, PH: Pulmonary hypertension, RVEF: Right ventricular ejection fraction
| Pa PC versus Fick (ml) | Pa PC versus TD (ml) | Fick versus TD (ml) | ||||
|---|---|---|---|---|---|---|
| Mean difference | Standard deviation | Mean difference | Standard deviation | Mean difference | Standard deviation | |
| All patients (n=81) | ‒0.22 | 15.11 | ‒10.86 | 16.99 | ‒11.1 | 14.4 |
| Nonobese BMI (n=35, <30 kg/m2) | 2.30 | 11.65 | ‒8.06 | 15.00 | ‒10.36 | 13.80 |
| Obese (n=46, ≥30 kg/m2) | ‒1.35 | 17.25 | ‒13.00 | 18.24 | ‒11.64 | 14.95 |
| Low CO (n=205, <4 L/min) | ‒5.60 | 12.74 | ‒9.90 | 10.16 | ‒4.29 | 11.77 |
| Normal CO (n=61, >4 L/min) | 2.14 | 15.42 | ‒11.18 | 18.76 | ‒13.32 | 14.55 |
| Decreased LVEF (n=25, <57%) | ‒1.60 | 14.37 | ‒9.90 | 14.90 | ‒8.29 | 15.00 |
| Normal LVEF (n=56, ≥57%) | 1.05 | 15.48 | ‒11.29 | 17.95 | ‒12.34 | 14.07 |
| Decreased RVEF (n=42, <52%) | 0.70 | 11.63 | ‒10.02 | 12.88 | ‒10.72 | 11.31 |
| Normal RVEF (n=39, ≥52%) | ‒0.28 | 18.40 | ‒11.77 | 19.32 | ‒11.48 | 17.55 |
| Group 1 PH (n=55) | 1.00 | 13.96 | ‒9.35 | 13.07 | ‒10.35 | 15.80 |
| Nongroup 1 PH (n=26) | ‒1.41 | 17.49 | ‒14.06 | 23.23 | ‒12.65 | 10.97 |
BMI: Body mass index, CO: Cardiac output, LVEF: Left ventricular ejection fraction, Pa: Pulmonary artery, PH: Pulmonary hypertension, RVEF: Right ventricular ejection fraction
DISCUSSION
The seminal study to date that validated PC in determining SV in patients with cardiac disease was a small study performed by Hundley et al.[9] A total of 23 patients underwent CMR followed immediately by RHC with Fick and TD measurements of CO. Coronary artery disease and dilated cardiomyopathy were the most common underlying disease in this study. The mean difference in SV with Ao PC-Fick was 3 ml (SD 9 ml) while Ao PC-TD mean difference was ‒3 ml (SD 11 ml). The mean SV by Fick and TD versus PC yielded an r = 0.89.
Innumerable studies have demonstrated the intrinsic value of PC in congenital heart disease in both children and adults predominantly in shunt evaluation. Less attention has been focused on PH patients as a specific group with CO and SV. Another small study by Mauritz et al. also evaluated PC in the measurement of SV in patients with PH.[12] A total of 34 patients with the WHO Group I were evaluated with CMR and RHC within 12 h. In this study, CO was performed using the direct Fick method. Fick was compared to PC of the main Pa, ascending Ao (however, in only 9 out of 34 patients), RV volume, and LV volume. The mean difference between Pa PC- Fick was ‒4.2 ml (SD 11.48 ml). Pa PC-Fick yielded r = 0.71. The above study is limited by having only 34 subjects.
Another study evaluated the use of PC in 128 PH patients (divided evenly between derivation and validation cohort) in the Assessing the Spectrum of PH Identified at a Referral Centre Registry.[13] The aim of the study was to develop a composite numerical model from CMR for non-invasive estimation of hemodynamic parameters obtained from RHC particularly mean Pa pressure. The investigators compared CO derived through TD to LV volumetry and Ao PC. In this study, the Ao PC-TD had an r = 0.70 which is lower than r = 0.79 in our study. The Ao PC-TD comparison in this study was similar to our study with a mean difference of 0.4 L/min (SD = 1.2 L/min). Ao PC-TD in our study had a higher mean difference at ‒1.12 L/min but had a slightly lower standard deviation at 1.06 L/min. The aforementioned study did not report comparisons of SV.
In our study, the mean difference in SV with PC-Fick is almost identical as the prior studies by Hundley et al. However, the standard deviation is slightly higher in our study (15.11 ml) when compared to Hundley et al. (11 ml) and Mauritz et al. (4 ml).[9,12] This difference may be explained by the fact that in our study, the indirect (assumed) Fick method was used while the two other studies, direct Fick method was used which has an inherently more accurate measurement of oxygen consumption.
The range of disagreement with Fick-TD in our study was similar to prior studies in a wide range of patients from healthy volunteers to patients with cardiopulmonary disease, including those with PH.[14-17] However, there have been concerns regarding the accuracy of TD compared to indirect Fick. Most studies that have compared Fick with TD used the direct Fick method including the original TD validation study.[14,17] There have been studies that have shown discrepancies between indirect Fick and TD. In a study of 213 RHC cases of which 91 had PAH, the mean difference with indirect Fick-TD was ‒0.39 L/min (SD 2 L/min).[18] In contrast, standard deviation between Fick and TD in our study was lower (SD 1.05 L/min), suggesting less variation in our study.
Regarding the internal accuracy of PC, we have previously published data from our laboratory showing excellent results measuring SV in vitro. Using a phantom with a pulsatile flow pump system utilizing a fluid with a viscosity similar to blood (Shelly Medical Imaging, Vancouver, Canada), there was excellent correlation of measured SV with the pump settings (r = 0.98, P < 0.001).[19] We also compared the PC measurements across the ascending Ao and the Pa in our study for further evaluation. The mean difference was ‒4.35 ml (SD 10.45 ml) in addition to r = 0.88. Our findings showed less bias and variability along with higher correlation than the comparison between Fick and TD. Compared to the aortic measurements, the Pa measurements have lower mean difference and standard deviation. The differences might be explained by lower accelerations across the Pa which minimizes technical errors.
Sources of variation in the measurements, particularly for PC, include turbulent flow, accelerated jet flow, and convergent flow. These factors are unlikely to be present in our study as there were no patients with significant valvular stenosis. Discarding outlier measurements before averaging in the catheterization laboratory could also affect the final measurements entered. Another source of difference between the invasive and non-invasive measurements was the use of sedation. We did not control for use of sedation both in the catheterization and CMR laboratories. Sedation was given under the discretion of the supervising physician for pain and anxiety management. In the overall cohort, the correlation between the SV measurements was higher compared to the CO measurements indicating that removing the heart rate as a factor decreases the variability in the measurements.
The Fick method is considered the “gold standard” in the measurement of CO. However, it is important to consider whether direct or indirect Fick is being performed. Prior studies comparing direct Fick and PC showed high precision (low standard deviation) and correlation between the two methods. However, our study which used the same PC technique showed slightly lower precision (higher standard deviation) when compared to indirect Fick.
In addition, our comparison between the two catheterization techniques (Fick-TD) had lower precision than PC-Fick. It is also critical to note that the bias is lower between PC- Fick (3 ml) compared to Fick-TD (11 ml) showing higher accuracy of PC over TD when compared to Fick. We performed further analysis with ANOVA to determine if there are significant differences in the measurements. For the Pa, there was significant difference (P < 0.01) between the three SV measurements. PC (SD 22.5 ml) and Fick (SD 23.2 ml) had similar standard deviation, but TD (SD 27.5 ml) had higher standard deviation. Using the ANOVA contrast test for relative ordering ranking, PC and Fick had lower variation then TD was (P < 0.05). For the aorta, there was significant difference (P < 0.001) between the three SV measurements. Using the ANOVA contrast test for relative ordering ranking, PC (SD 21.5 ml) was first followed by Fick (SD 23.2 ml) and finally TD (SD 27.5 ml) (P < 0.01). In this three-way comparison utilizing indirect Fick, TD, and PC, we were able to show that non-invasive PC compares to the most referenced invasive standard: Direct Fick. Our findings show that the lower precision with PC-Fick, due to the assumption of oxygen consumption, reflects a higher accuracy with PC.
To the best of our knowledge, this is the largest study to date that compared PC with indirect Fick and TD. Other smaller studies only compared PC to either Fick or TD and were performed as part of research protocols and utilized direct measurement of oxygen consumption. We also performed measurements in both the Ao and Pa in all the patients. Although Fick is more accurate through direct measurement of oxygen consumption, this process is tedious and is not performed in routine clinical practice. The patients included in our study reflect testing performed for the management of their PH and the methods used in usual “real-world” practice.
Our study demonstrates the accuracy of CMR in determining CO without the need to assume oxygen consumption or the need for rigorous invasive techniques. This is of particular importance in patients that require frequent hemodynamic assessment such as those with PH. CMR can provide non- invasive assessment of cardiac structure and function and is invaluable in assessing right ventricular morphologic changes.[20] However, CMR is unlikely to supplant RHC yet as the test of choice for the initial diagnosis of PH as measurement of pulmonary pressures and pulmonary vascular resistance by CMR is still being investigated.[13,21] Our study adds further evidence that CMR provides invaluable information as a non-invasive monitoring tool for patient with PH and may provide a more accurate assessment of CO, an otherwise invasive measurement. The implications for clinical and research considerations toward moving this high-risk population to less, not more invasive interventions are substantial.
CONCLUSION
Non-invasive measurement of CO and SV using PC-CMR correlates well with invasive measurement using RHC. Our study showed that PC-CMR had high accuracy and precision when compared to Fick. Among all the modalities, PC-CMR contributed the least amount of variation in measurements.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
Robert Biederman, MD
Honoraria: Amgen, Bracco, Lantheus, Sanofi/ Regeneron Grants: Biotronik, NIH/NHLB, United Therapeutics.
Amresh Raina, MD
Consultant: Abbot Clinical trials: Actelion, Bayer, Bellerophon, United Therapeutics
References
- Clinically significant change in stroke volume in pulmonary hypertension. Chest. 2011;139:1003-9.
- [CrossRef] [PubMed] [Google Scholar]
- Imaging right ventricular function to predict outcome in pulmonary arterial hypertension. Int J Cardiol. 2016;218:206-11.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of epoprostenol on right ventricular hypertrophy and dilatation in pulmonary hypertension. Chest. 2004;125:572-9.
- [CrossRef] [PubMed] [Google Scholar]
- Complications of right heart catheterization procedures in patients with pulmonary hypertension in experienced centers. J Am Coll Cardiol. 2006;48:2546-52.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness: The ESCAPE trial. JAMA. 2005;294:1625-33.
- [CrossRef] [PubMed] [Google Scholar]
- Feigenbaum's Echocardiography (7th ed). Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams and Wilkins; 2010.
- [Google Scholar]
- Evaluation of the precision of magnetic resonance phase velocity mapping for blood flow measurements. J Cardiovasc Magn Reson. 2001;3:11-9.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular magnetic resonance phase contrast imaging. J Cardiovasc Magn Reson. 2015;17:71.
- [CrossRef] [PubMed] [Google Scholar]
- Quantitation of cardiac output with velocity-encoded, phase-difference magnetic resonance imaging. Am J Cardiol. 1995;75:1250-5.
- [CrossRef] [Google Scholar]
- Diagnosis and assessment of pulmonary arterial hypertension. J Am Coll Cardiol. 2009;54:S55-66.
- [CrossRef] [PubMed] [Google Scholar]
- Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307-10.
- [CrossRef] [Google Scholar]
- Non-invasive stroke volume assessment in patients with pulmonary arterial hypertension: Left-sided data mandatory. J Cardiovasc Magn Reson. 2008;10:51.
- [CrossRef] [PubMed] [Google Scholar]
- Noninvasive estimation of PA pressure, flow, and resistance with CMR imaging: Derivation and prospective validation study from the ASPIRE registry. JACC Cardiovasc Imaging. 2013;6:1036-47.
- [CrossRef] [PubMed] [Google Scholar]
- Determination of cardiac output by the Fick method, thermodilution, and acetylene rebreathing in pulmonary hypertension. Am J Respir Crit Care Med. 1999;160:535-41.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of thermodilution and fick methods for measurement of cardiac output in tricuspid regurgitation. Am J Cardiol. 1992;70:538-9.
- [CrossRef] [Google Scholar]
- Comparison of cardiac output measurement techniques: Thermodilution, Doppler, CO2-rebreathing and the direct Fick method. Acta Anaesthesiol Scand. 1995;39:245-51.
- [CrossRef] [PubMed] [Google Scholar]
- Measuring cardiac output in critically Ill patients: Disagreement between thermodilution-, calculated-, expired gas-, and oxygen consumption-based methods. Cardiology. 1997;88:19-25.
- [CrossRef] [PubMed] [Google Scholar]
- Thermodilution and Fick cardiac outputs differ: Impact on pulmonary hypertension evaluation. Can Respir J. 2012;19:261-6.
- [CrossRef] [PubMed] [Google Scholar]
- Routine evaluation of left ventricular diastolic function by cardiovascular magnetic resonance: A practical approach. J Cardiovasc Magn Reson. 2008;10:36.
- [CrossRef] [PubMed] [Google Scholar]
- Role of cardiac magnetic resonance imaging in the management of patients with pulmonary arterial hypertension. J Am Coll Cardiol. 2008;52:1683-92.
- [CrossRef] [PubMed] [Google Scholar]
- Investigating cardiac MRI based right ventricular contractility as a novel non-invasive metric of pulmonary arterial pressure. Clin Med Insights Cardiol. 2014;8:45-50.
- [CrossRef] [PubMed] [Google Scholar]






