Translate this page into:
Pineal Gland Lymphoma: Case Report and Literature Review
Address for correspondence: Dr. Akshya Gupta, 601 Elmwood Avenue, Box 648, Rochester - 14642, New York, USA. E-mail: Akshya_Gupta@urmc.rochester.edu
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
A 65-year-old male presented to our institution with acute-onset headache. Imaging studies demonstrated a mass in the region of the pineal gland, with subsequent histopathology findings being consistent with large B cell lymphoma. The patient was treated with methotrexate, but ultimately did not survive. Primary central nervous system (CNS) lymphoma rarely involves the pineal gland, but should be considered in the differential diagnosis of pineal gland tumors in the appropriate clinical setting.
Keywords
Lymphoma
pineal gland
hydrocephalus

INTRODUCTION
Primary central nervous system (CNS) lymphoma is a relatively uncommon malignancy, accounting for approximately 6% of all malignant CNS neoplasms.[1] They typically occur in middle-aged and older adults, although it can be seen more commonly in younger patients with human immunodeficiency virus (HIV) or transplant recipients. More than 90% of primary CNS lymphomas are of the diffuse large B-cell variety.[1] Most of these lesions appear hyperdense on a non-contrast enhanced computed tomography (CT) scan and tend to demonstrate restricted diffusion on magnetic resonance imaging (MRI), given their high cellularity. This is the eighth reported case of CNS lymphoma involving the pineal gland in the literature.[234567]
CASE REPORT
A 65-year-old male presented with a 2-week history of worsening headache and double vision. Initial non-contrast head CT demonstrated a homogeneous hyperdense mass in the pineal gland region with mild hydrocephalus, but no calcification or hemorrhage [Figure 1]. Contrast MRI was performed, which demonstrated a homogeneously enhancing mass involving the pineal gland, with increased perfusion with corresponding low apparent diffusion coefficient values [Figure 2]. The mass was hypointense on T1-weighted images and isointense to mildly hyperintense compared to brain parenchyma on T2-weighted images. Leptomeningeal enhancement was identified near the supraoptic recess and along the cerebellar velum [Figure 3].
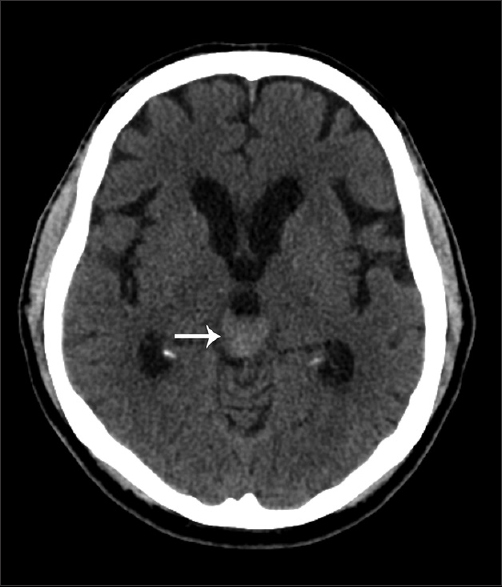
- 65-year-old male presented with headache and was later diagnosed with pineal gland lymphoma. Axial non-contrast CT of the head demonstrates a homogeneously hyperdense mass (arrow) in the pineal gland region. There are no calcifications or hemorrhage identified.
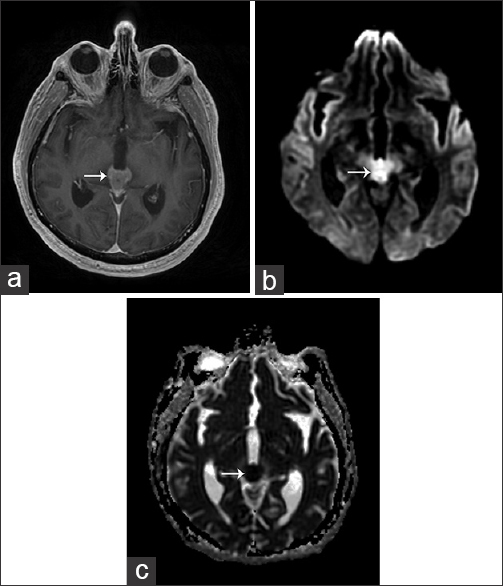
- 65-year-old male presented with headache and was later diagnosed with pineal gland lymphoma. (a) Axial T1 post-contrast MRI image of the head demonstrates a homogeneously enhancing mass (arrow) in the region of the pineal gland. (b) Axial MRI diffusion-weighted imaging (DWI) sequence demonstrates increased signal (arrow). (c) The corresponding axial apparent diffusion coefficient (ADC) map demonstrates low ADC values (arrow).
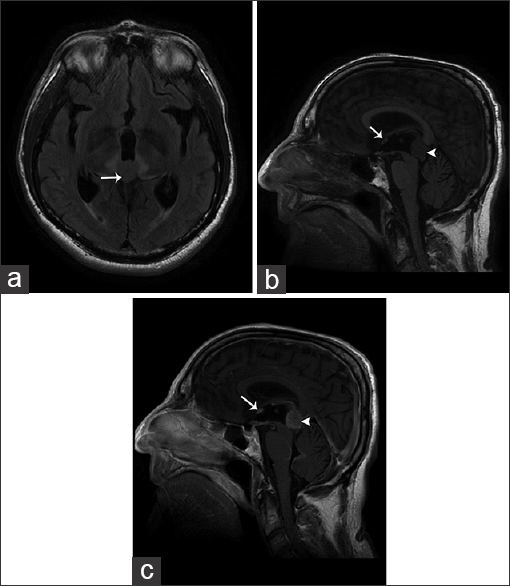
- 65-year-old male presented with headache and was later diagnosed with pineal gland lymphoma. (a) Axial T2 FLAIR MR image demonstrates the pineal gland mass (arrow) as isointense to mildly hyperintense to brain parenchyma. (b) Sagittal T1-weighted MR image demonstrates that the mass is hypointense. (c) Sagittal T1 post-contrast MR image again shows the enhancing pineal gland mass (arrowhead) as well as leptomeningeal enhancement near the supraoptic recess (arrow) and cerebellar velum.
A transventricular endoscopic biopsy was performed. Cerebrospinal fluid sampling was not obtained unfortunately. The histopathology showed a hypercellular tumor with high nuclear/cytoplasmic ratio, but no pineocytomatous or Homer Wright rosettes or papillary architecture [Figure 4]. The nuclei had coarse chromatin, notches, and occasional nucleoli. The tumor appeared to involve the pineal parenchyma. Focal necrosis was found. No rhabdomyoblasts or strap cells were found. Mitoses were noted and Ki-67 labeled the majority of cells. Tumor cells exhibited vimentin, CD45, extensive CD20, CD79a, and nuclear PAX 5 reactivity. In situ hybridization showed kappa, but no lambda light chain hybridization. Scattered small lymphocytes showed CD3 immunoreactivity. There was focal granular synaptophysin, but no neuron specific enolase (NSE), glial fibrillary acidic protein (GFAP), beta tubulin, Cam 5.2, thyroid transcription factor (TTF-1), CD99, pancytokeratin, myogenin, or neurofilament immunoreactivity. A diagnosis of large B cell lymphoma was made.
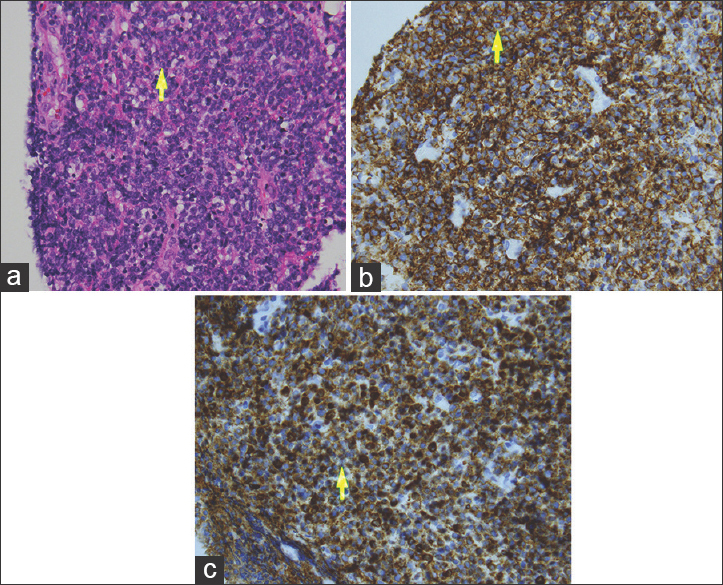
- 65-year-old male presented with headache and was later diagnosed with pineal gland lymphoma. (a) Hematoxylin and eosin staining at magnification (40×) demonstrates malignant lymphoma with coarse chromatin, nucleoli (arrow), and mitoses. (b) Diaminobenzidene chromagen at magnification (40×) demonstrates tumor cells (arrow) extensively exhibiting CD20 reactivity. (c) Diaminobenzidene chromagen at magnification (40×) demonstrates tumor cells (arrow) exhibiting extensive CD79a, but no neural, myogenic, or epithelial marker immunoreactivity.
At the time of biopsy, a ventricular drain was placed. Patient's symptoms resolved and the drain was subsequently removed after trial of clamping. Staging chest, abdomen, and pelvis CT exams revealed no other areas of lymphomatous involvement. The patient was discharged home in stable condition, but presented to the emergency department 1 week later with worsening hydrocephalus and headache. At this time, the patient underwent ventriculoperitoneal shunt placement for management of his hydrocephalus and received one cycle of high-dose methotrexate. However, he developed severe hypotension and acute kidney injury 2 months after his diagnosis and was re-admitted to the hospital, with repeat non-contrast head CT showing enlargement of the mass. Given his multiple medical comorbidities and evidence of disease progression, the patient and his family elected to pursue comfort care measures.
DISCUSSION
Primary CNS lymphoma accounts for approximately 6% of all malignant CNS neoplasms.[1] They can affect both immunocompetent and immunocompromised patients, although patients who are immunocompromised typically present at a younger age. There is a predilection for the periventricular white matter and basal ganglia, and they can present as either solitary or multiple mass lesions. Pathologically, most are high-grade B-cell lymphomas.
On non-contrast CT, most masses are hyperdense, given the high cellularity, and surrounding edema is common. Calcifications and necrosis are considered less common. Contrast enhancement is typically homogeneous, but can have a more variable appearance in immunocompromised patients. On MRI, although these lesions can appear hypointense to gray matter on T1-weighted imaging and hypointense to isointense on T2-weighted imaging, the hallmark is restricted diffusion due to the increased cellularity.[16] Enhancement is typically homogeneous.
Primary CNS lymphoma has rarely been found to involve the pineal gland, with seven cases reported in the literature. Headache was one of the most common presenting symptoms in these patients, although symptoms include cranial nerve and cauda equina syndrome, focal neurologic deficits, fever, diplopia, altered mental status, and seizure.[7] The average age at diagnosis was 40 years, and only one female patient has been reported. B-cell lymphoma has been the most common pathologic diagnosis, with cases including large B cell lymphoma, malignant B cell lymphoma, immunoblastic lymphoma, and anaplastic lymphoma kinase positive anaplastic large cell lymphoma (ALK-1 positive ALCL).[7] There has been a single case of malignant T cell lymphoma and one case providing no additional detail to the diagnosis of lymphoma. Imaging features in these cases most commonly included hydrocephalus and relatively homogeneously enhancing lesions identified on MRI.
The imaging findings in this particular case were typical of highly cellular tumor with leptomeningeal involvement, given the hyperdense mass on non-contrast CT and restricted diffusion with contrast enhancement on MRI. A histologic diagnosis is essential prior to beginning invasive treatment, as the imaging characteristics of pineal lymphoma are not necessarily pathognomonic. Other differential considerations for a tumor in the pineal region with these imaging characteristics include pineoblastoma, germ cell tumor, and metastatic disease. Pineoblastoma is a pediatric diagnosis with masses typically appearing more heterogeneous and with peripheral calcifications. Germ cell tumors are also typically diagnosed at a younger age and are associated with calcification. While metastatic disease may fit the imaging pattern and patient age seen in our case, a primary malignancy was not identified.
Our patient was treated with high-dose methotrexate, which has been shown to increase survival in patients with primary CNS lymphoma. The addition of rituximab to this regimen has also been shown to improve remission rates and progression-free survival; however, our patient decompensated and progressed prior to initiation of this therapy.[8]
CONCLUSION
Primary CNS lymphoma is an uncommon primary CNS malignancy, and rarely involves the pineal gland. However, in the appropriate clinical context and with relevant imaging findings, lymphoma should be in the differential diagnosis of a pineal gland mass. Prompt diagnosis and treatment are essential to improve survival in these patients.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2015/5/1/51/166350
REFERENCES
- Multimodality imaging of primary CNS lymphoma in immunocompetent patients. Br J Radiol. 2014;87:20130684.
- [Google Scholar]
- Spinal malignant lymphoma appearing after radiation and chemotherapy of a pineal region tumor. Surg Neurol. 1996;45:169-71.
- [Google Scholar]
- Neuroendocrine, immunohistochemical, and ultrastructural study of pineal region tumors. J Pineal Res. 1998;25:147-58.
- [Google Scholar]
- Primary cerebral ALK-1-positive anaplastic large cell lymphoma in a child. Case report and literature review. Pediatr Neurosurg. 2007;43:516-21.
- [Google Scholar]
- November 2002: A 72-year-old woman with a pineal gland mass. Brain Pathol. 2003;13:235-6.
- [Google Scholar]
- Atypical sites of lymphoma in the central nervous system. Arq Neuropsiquiatr. 2011;69:566-7.
- [Google Scholar]
- Pineal malignant B-cell lymphoma with lower cranial nerve involvement. Intern Med. 2014;53:1205-8.
- [Google Scholar]
- High-dose methotrexate with or without rituximab in newly diagnosed primary CNS lymphoma. Neurology. 2014;83:235-9.
- [Google Scholar]






