Translate this page into:
Mycotic Aneurysm of the Hepatic Artery: A Case Report and its Management

*Corresponding author: Abhinav Pravin Patel, Department of Imaging Sciences, University of Rochester Medical Center, Rochester, New York, United States. abhinav_patel@urmc.rochester.edu
-
Received: ,
Accepted: ,
How to cite this article: Patel AP, Cantos A, Butani D. Mycotic aneurysm of the hepatic artery: A case report and its management. J Clin Imaging Sci 2020;10:41.
Abstract
Cholecystectomies are one of the most common surgical procedures performed in the United States, with complications being relatively common. We submit a case of a ruptured mycotic aneurysm of the hepatic artery, a complication of a recent cholecystectomy which was successfully treated with the use of transarterial thrombin.
Keywords
Mycotic aneurysm
Hepatic artery
Thrombin
Angiography
Cholecystectomy
INTRODUCTION
Cholecystectomies are one of the most common surgical procedures performed in the United States, with over 300,000 laparoscopic cholecystectomies performed every year.[1] The complication rates of cholecystectomies range from 1.56% to 6%. Common complications include bleeding from adjacent tissues or the cystic artery, iatrogenic perforations of the gallbladder, injuries to the bile duct, and infection, including abscess formation.[2] We submit a case of a post- cholecystectomy patient who presented with a ruptured hepatic artery mycotic aneurysm from an abscess within the porta hepatis ultimately requiring endovascular treatment.
Topical thrombin is a common tool utilized by physicians in the treatment to achieve hemostasis. Its use has even expanded to the treatment of superficial pseudoaneurysm. However, there have been only a few cases of catheter-directed endovascular use of thrombin. This case presentation describes the transarterial use of thrombin in the treatment of the aneurysm.
PATIENT CASE
This case involves a 71-year-old male with a past medical history of depression, complex regional pain syndrome, and non-insulin dependent diabetes mellitus Type 2 who presented with abdominal pain and found to have symptomatic cholelithiasis requiring laparoscopic cholecystectomy followed by a complex course, including acute blood loss of approximately 350 cc intraoperatively.
He returned to the emergency department (ED) approximately 2 weeks post-discharge with complaints of worsening abdominal pain, nausea, and vomiting. Contrast-enhanced CT of the abdomen and pelvis (CT A/P) demonstrated multiple large subscapular hepatic abscesses [Figure 1a and b]. Drainage catheters were subsequently placed in two abscesses [Figure 2a and b]. On repeat, contrast-enhanced CT A/P demonstrated new focal dilatation in the right hepatic artery concerning the development of a mycotic pseudoaneurysm [Figure 3].
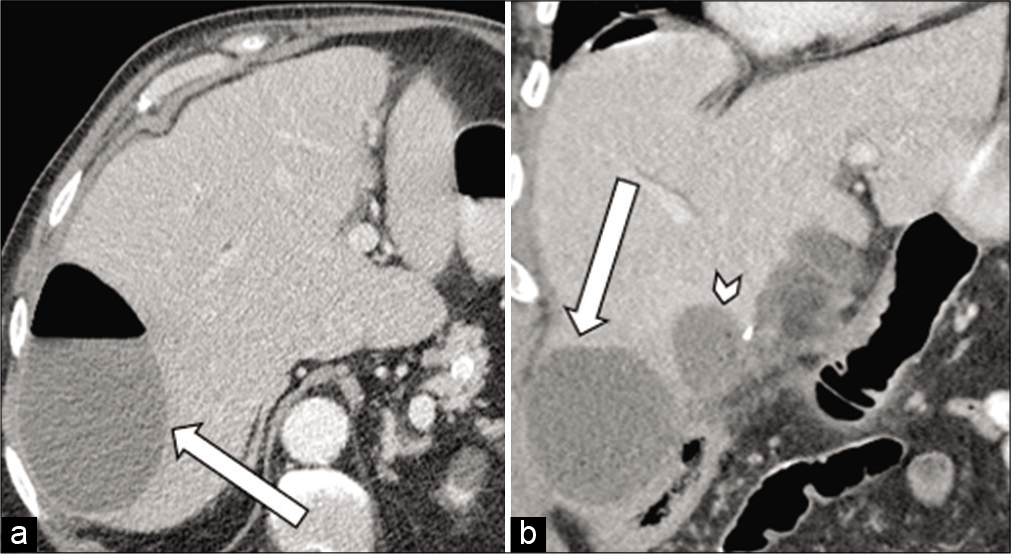
- 71-year-old male with a recent cholecystectomy who presents with abdominal pain (a) axial contrast-enhanced CT of the abdomen and pelvis demonstrates a large abscess in the right hepatic lobe (arrow). (b) Coronal contrast-enhanced CT of the abdomen and pelvis (right image) demonstrates another large abscess at the base of the right hepatic lobe (arrow) with a medial smaller abscess (arrowhead).
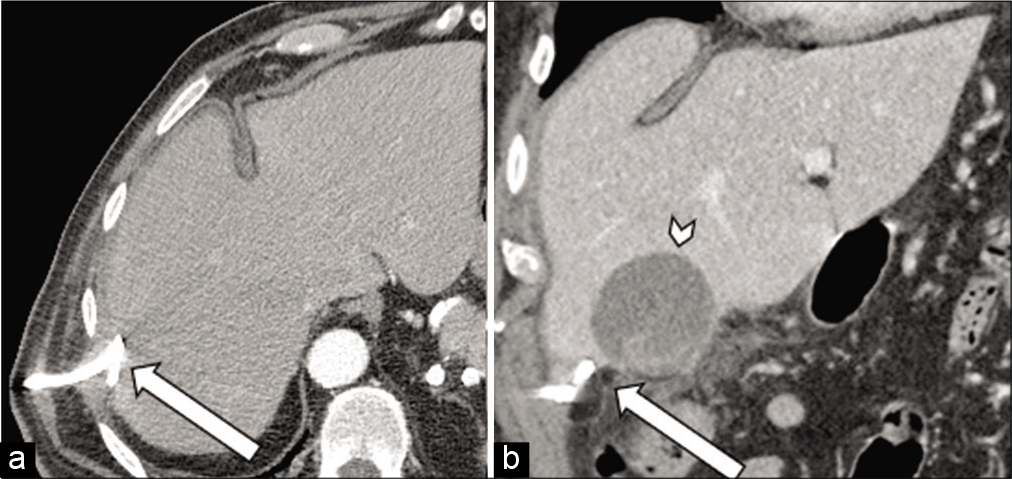
- 71-year-old male with a recent cholecystectomy complicated by multiple abdominal abscesses who presents with abdominal pain. (a) Axial contrast-enhanced CT of the abdomen and pelvis (left image) demonstrates placement of drainage catheter in the right hepatic lobe abscess (arrow). (b) Coronal contrast- enhanced CT of the abdomen and pelvis (right image) demonstrates a drainage catheter in the inferior hepatic abscess (arrow) with interval growth of the more medial abscess (arrowhead).
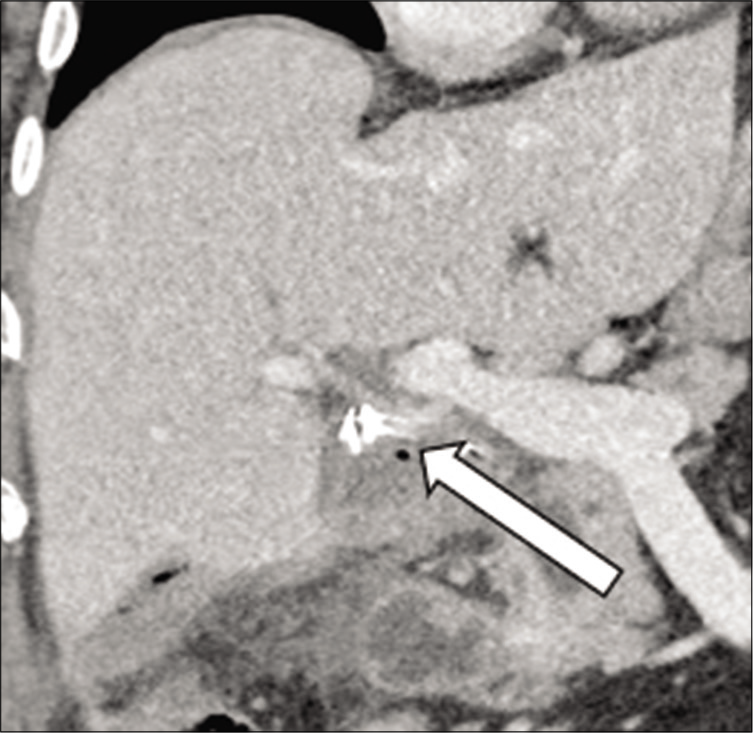
- 71-year-old male with a recent cholecystectomy complicated by multiple abdominal abscesses who presents with abdominal pain. Coronal contrast-enhanced CT of the Abdomen and pelvis demonstrates a subtle aneurysmal dilatation of the proper hepatic artery (arrow) concerning for a mycotic aneurysm.
A visceral angiogram was performed, which demonstrated aneurysmal dilation at the distal portion of the proper hepatic artery measuring approximately 1.5 cm [Figure 4]. A collaborative decision was made to conservatively treat the aneurysm with medical management due to the high risk of inducing liver failure with coil embolization. The patient was later discharged on oral antibiotics.
- 71 year old male with a recent cholecystectomy complicated by multiple abdominal abscesses who presents with worsening abdominal pain. Angiogram of the hepatic artery demonstrating an aneurysm at the distal hepatic artery (arrow) with no evidence of active bleeding.
He returned to the ED approximately a week later with worsening of his symptoms, and a repeat CT A/P demonstrated a new fluid collection within the porta hepatis with interval enlargement of the pseudoaneurysm [Figure 5]. The patient acutely decompensated and suffered cardiac arrest requiring multiple rounds of cardiopulmonary resuscitation. It was presumed that there was active bleeding from the known hepatic artery aneurysm. Bleeding was presumed secondary to a rupture of the known mycotic aneurysm which required massive transfusion protocol with approximately 10 units of PRBC and vasopressors. The patient was immediately taken to the interventional radiology suite.
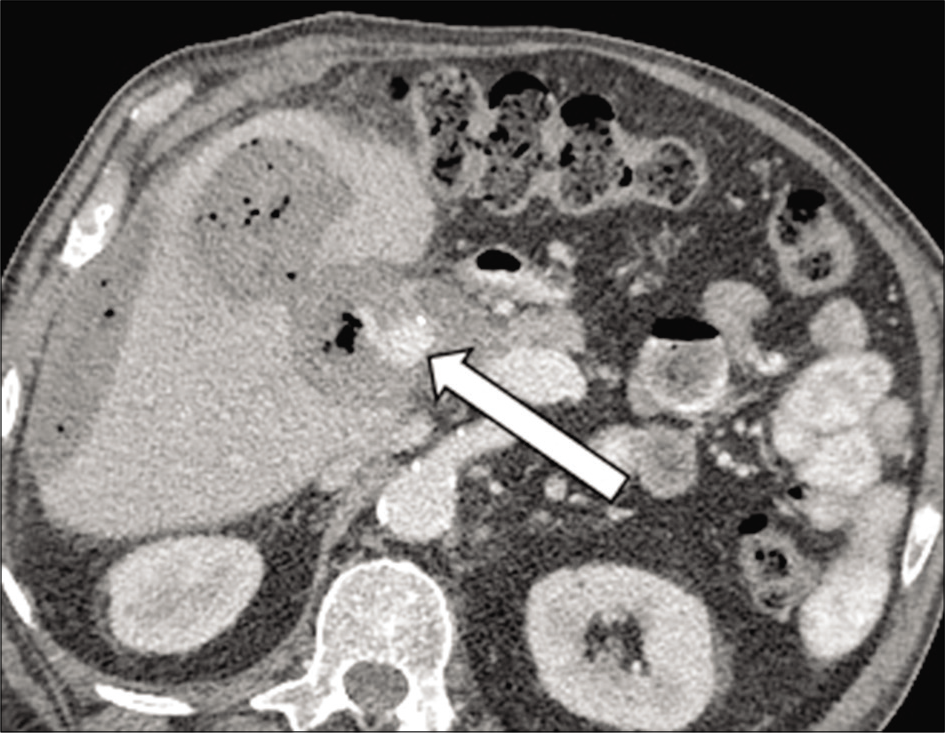
- 71 year old male with a known mycotic pseudoaneurysm of the hepatic artery presents with worsening abdominal pain. Axial contrast-enhanced image of the abdomen demonstrates interval growth of the mycotic aneurysm (arrow).
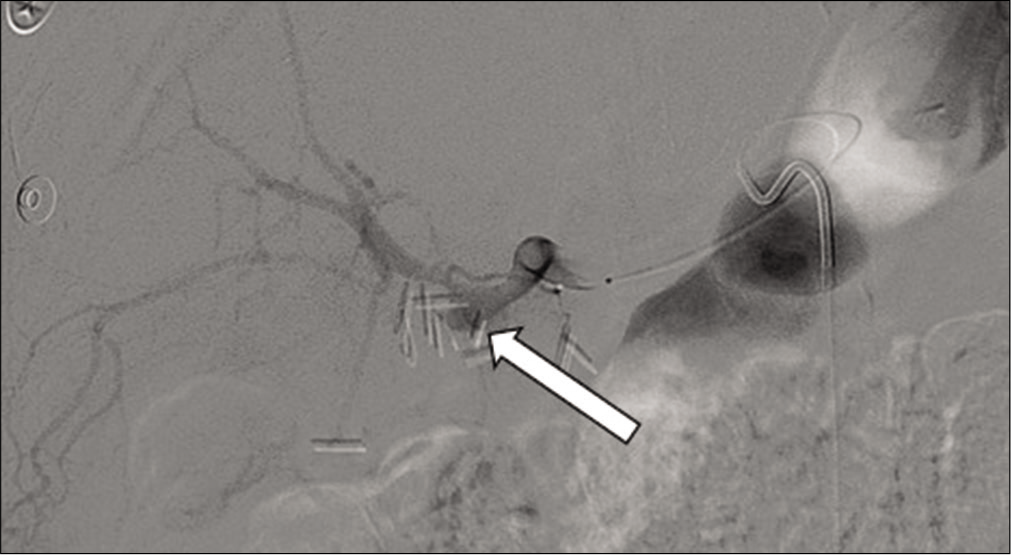
The celiac artery was cannulated with an SOS catheter. An angiogram was performed demonstrating frank extravasation just after the origin of the proper hepatic artery [Figure 6]. No distal flow into the liver parenchyma was identified past the rupture. A microcatheter and microwire were used to obtain access past the gastroduodenal artery (GDA) into the stump of the proper hepatic artery, which was then embolized with coils up to the confluence of the proper hepatic artery and GDA branches. Despite the placement of multiple coils, there was continued visualization of active hemorrhage [Figure 7]. The continued hemorrhage was presumed to be from evolving DIC a transfusion-related coagulopathy.
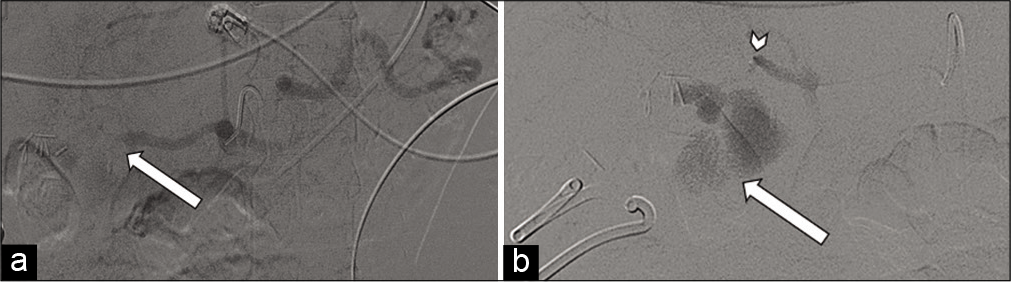
- 71-year-old male with a known mycotic pseudoaneurysm of the hepatic artery who recently coded on multiple vasopressors. (a) Angiogram at the celiac artery demonstrates no distal flow to the hepatic artery with large active hemorrhage (arrow). (b) Microcatheter was advanced to the distal hepatic artery (arrowhead), with angiogram demonstrating massive active hemorrhage (arrow).
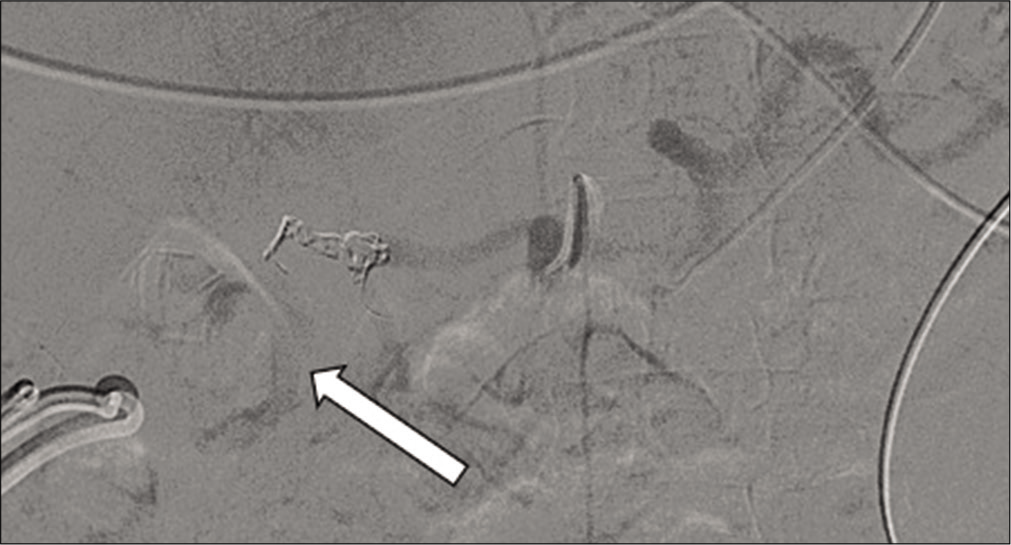
- 71-year-old male with a known mycotic pseudoaneurysm of the hepatic artery who recently coded on multiple vasopressors. Angiogram demonstrating multiple coils deployed into the gastroduodenal and proximal proper hepatic artery. Despite the placement of the coils, there is still active contrast extravasation from the aneurysm (arrow).
Given lack of other suitable embolic agents available at the time, at a regional community hospital, a decision was made to inject thrombin into the coil mass. The microcatheter was advanced into the proximal coils, positioning was verified with an injection of contrast, and subsequently, 1000 units of reconstituted thrombin were injected. The microcatheter was then removed, the 5 French catheters was aspirated and flushed, and a repeat angiogram through the 5 French catheters demonstrated successful embolization [Figure 8]. Unfortunately, due to multiorgan failure and persistent shock-like physiology from bacteremia, the patient was pronounced dead 3 days after the procedure.
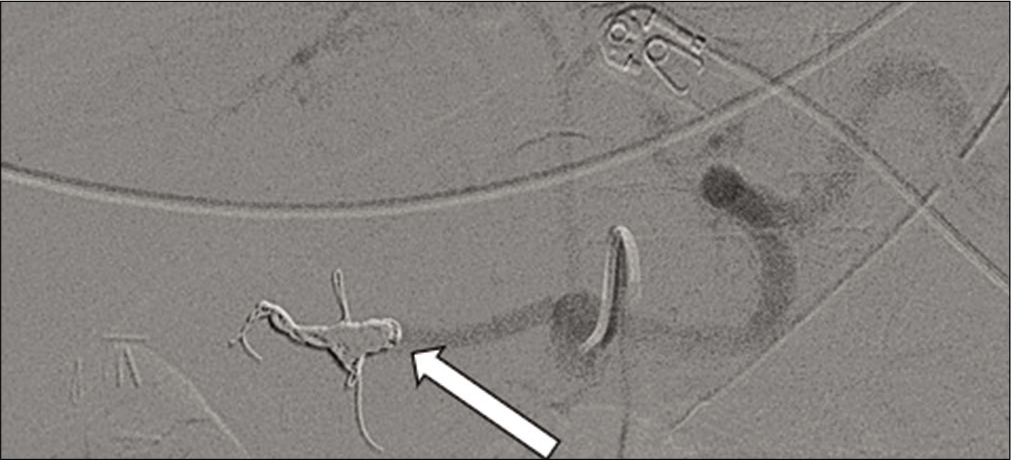
- 71-year-old male with a known mycotic pseudoaneurysm of the hepatic artery who recently coded on multiple vasopressors. After the direct administration of thrombin through the catheter, an angiogram of the celiac artery demonstrates successful embolization and no further extravasation (arrow).
DISCUSSION
Mycotic aneurysms, commonly referred to as infected aneurysms, are rare in their frequency. However, they can be life-threatening with a high incidence of arterial rupture and recurrence even after treatment. The term “mycotic,” which usually means fungus, was coined by physician Sir William Osler in 1885 when he described the gross pathological appearance of two small saccular aortic aneurysms and not the underlying pathological organism responsible for its development.[3,4]
Mycotic aneurysms normally arise from bacterial invasion into the arterial wall through hematogenous spread usually in immunocompromised patients such as those with diabetes mellitus, liver cirrhosis, and acquired immune deficiency syndrome or with patient switch arterial luminal defects such as atherosclerotic plaque, ulceration, and preexisting aneurysm. Common locations for the development of mycotic aneurysms include the thoracic and abdominal aorta, abdominal visceral arteries, lower extremity arteries, and intracranial arteries. In the case of our patient, the presence of the abscess within the porta hepatis allowed for the proliferation of the mycotic aneurysm.
Aneurysms of the hepatic artery are uncommon. They tend to be saccular and multiple. Causes typically include infection, arteriosclerosis, trauma, and vasculitis. Post-liver transplantation patients can have pseudoaneurysm formation of the hepatic artery and its branch vessels, these usually occur within the 1st month of transplantation. A vasculitis such as polyarteritis nodosa has been reported to result in the formation of aneurysms.[5] Pseudoaneurysm formation can occur after trauma to the hepatic artery; furthermore, there have even been cases of pseudoaneurysm formation trauma from a percutaneous biopsy.[6]
Diagnostic testing
Before definitive treatment, most patients with known or suspected mycotic aneurysms undergo essential diagnostic testing, including complete blood count, comprehensive metabolic panel, gram stain, blood cultures, and sensitivities, as well as contrast-enhanced CT or magnetic resonance imaging. The two most essential components in the treatment of mycotic aneurysm involve culture-specific antibiotic therapy and physical treatment of the pseudoaneurysm either with surgical excision or with endovascular coiling depending on the patient’s clinical condition.[7]
Treatment
Surgical intervention is the first-line treatment in patients who are sta ble once antibiotic therapy which is initiated. Surgical options such as excision and ligation can be performed in smaller distal arteries where the chance of end- organ ischemia is low. Excision with reconstruction is another option when there is the involvement of larger arteries such as the thoracic aorta or suprarenal aorta. The reconstruction can be performed immediately but can result in recurrent infection or breakdown of the anastomoses. Interval reconstruction can be performed as well but with the risk of end-organ ischemia.[3] Due to the hemodynamic instability of our patient, he was not a candidate for immediate surgical intervention and immediate endovascular treatment was needed.
Endovascular treatment is a viable option when surgical treatment is contraindicated or unavailable. The two main available endovascular therapies involve the placement of endovascular stents in the case of an intact aneurysm or coil embolization. Endovascular stent placement is a suitable option in situations involving high-risk patients where surgery is prohibitive due to high mortality risk or during emergent cases involving the rupture of a mycotic aneurysm. Coil embolization can be performed as a temporizing measure until surgical debridement can be performed in more controlled circumstances or in situations where the likelihood of end-organ ischemia is considered low, such as with smaller visceral organ branches.[3] In the case of our patient, the placement of an endovascular stent was not feasible due to the acuity of the patient’s condition and lack of availability of stents at the regional community hospital.
Thrombin
Thrombin acts within the last step of the coagulation cascade by converting fibrinogen into fibrin to ultimately form a clot. Thrombin also assists in causing vasoconstriction and platelet activation. Thrombin was initially used clinically in the early 1900s as an emulsion on incision sites to promote healing.[7] Human thrombin together with fibrin was used in burn injuries during World War II. However, due to the risk of transmission of viral hepatitis, bovine and recombinant thrombin formulations were developed.[8]
Thrombin is only approved by the Food and Drug Administration for topical application. The most common formulations of thrombin involve a solution or reconstituted powder. Thrombin is commonly used in conjunction with fibrin-based products or hemostatic sponges. Thrombin is available in three topical formulations: Bovine plasma- derived (b-thrombin), human plasma-derived (h-thrombin), and recombinant (r-thrombin).[8,9] Even though thrombin is typically used for topical formulations due to the potential risk of intravascular thrombus formation, off label uses for percutaneous injections in the treatment of superficial angiography access site pseudoaneurysms has been documented and has demonstrated a high success rate upward of 95%. In our review of the literature for Cather directed endovascular use of thrombin, only 28 cases were reported from 1987.[8]
CONCLUSION
While the complications of cholecystectomy are well known, some of the adverse outcomes may result in complex and life-threatening complications, often requiring a multidisciplinary approach in treatment such as in the case of our patient which required medical, surgical, and interventional collaboration.
The treatment of our patient’s ruptured aneurysm required the use of thrombin, an agent which is rarely used endovascularly as a means of hemostasis, allowing for hemodynamic stability. Post-surgical complications after cholecystectomy and other similar procedures may necessitate the use of endovascular intervention to aid in the patient’s care, often in highly complex and life-threatening conditions. Depending on the clinical situation and availability of resources, the use of novel or atypical agents such as thrombin may be the difference between life or death for patients.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Complications of laparoscopic cholecystectomy: Our experience from a retrospective analysis. Open Access Maced J Med Sci. 2016;4:641-6.
- [CrossRef] [PubMed] [Google Scholar]
- Infected aneurysm: Current management. Ann Vasc Dis. 2010;3:7-15.
- [CrossRef] [PubMed] [Google Scholar]
- The gulstonian lectures, on malignant endocarditis. Br Med J. 1885;1:467-70.
- [CrossRef] [Google Scholar]
- Ruptured hepatic aneurysm as first presenting symptom of polyarteritis nodosa. Oxf Med Case Reports. 2018;2018:omx100.
- [CrossRef] [PubMed] [Google Scholar]
- Delayed fatal hemorrhage from pseudoaneurysm of the hepatic artery after percutaneous liver biopsy. Am J Gastroenterol. 2001;96:233-27.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular and Interventional Radiology, the Requisites Maryland Heights: Mosby Inc; 2004.
- [Google Scholar]
- Catheter-directed endovascular application of thrombin: Report of 3 cases and review of the literature. Clin Imaging. 2017;42:96-105.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical use of topical thrombin as a surgical hemostat. Biologics. 2008;2:593-9.
- [CrossRef] [PubMed] [Google Scholar]






