Translate this page into:
Multiple Artery Aneurysms: Unusual Presentation of IgG4 Vasculopathy

*Corresponding author: Anurag Yadav, Department of Computed Tomography and Magnetic Resonance Imaging, Sir Ganga Ram Hospital, New Delhi, India. anu_nalin@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Yadav A, Godasu G, Buxi TB, Sheth S. Multiple artery aneurysms: Unusual presentation of IgG4 vasculopathy. J Clin Imaging Sci 2021;11:17.
Abstract
Immunoglobulin G4 (IgG4)-related disease is a chronic systemic disease. It is characterized by inflammatory fibrosis and high serum IgG4 levels. IgG4-positive plasma cells infiltrate target organs in this disease. It may involve the pancreas, biliary tract, lacrimal glands, salivary glands, orbits, thyroid, kidneys, lymph nodes, or retroperitoneum. It may present as vasculitis with involvement of large to medium sized vessels such as the aorta, the common iliac, carotid, and coronary arteries. We present a case of 55-year-old male patient who presented with shortness of breath on exertion and atypical chest pain. On CT angiography, a giant coronary artery aneurysm involving the left anterior descending artery, multiple visceral and intercostal artery aneurysms, and nodular paravertebral soft-tissue thickening secondary to IgG4 vasculopathy.
Keywords
Angiography
Coronary artery aneurysm
Immunoglobulin G4
Vasculitis
INTRODUCTION
Immunoglobulin G4 (IgG4) was described as a multisystemic disease in 2003, when extrapancreatic involvement was noted in a case of autoimmune pancreatitis.[1] The disease is seen to affect nearly every organ system, including the vascular system. It is referred to by various terms such as IgG4-related sclerosing disease, IgG4-related systemic disease, IgG4-related autoimmune disease, and systemic IgG4-related plasmacytic syndrome.[2] The exact pathogenesis of the disease is ambiguous with features of autoimmune and allergic disorders. It primarily affects men in the 5th to 7th decade. Clinical symptoms are usually mild, with mildly elevated C-reactive protein (CRP) levels. The presence of >50 IgG4 plasma cells per high-power microscopic field is considered highly specific for IgG4-related disease. A ratio of IgG4-positive to IgG-positive plasma cells >40% helps in differentiating it from the other inflammatory conditions. Storiform fibrosis and obliterative phlebitis are other hallmark findings on histopathology.
CASE REPORT
A 55-year-old hypertensive man presented with complaints of shortness of breath on exertion and atypical chest pain to the emergency department. There was a history of fenestrated endovascular aneurysm repair of the abdominal aorta in 2014, stents in the superior mesenteric artery and bilateral renal arteries, injection of a ruptured aneurysm of gastroduodenal artery with thrombin, glue, and coils in 2017, and cholecystectomy in 2018. Pre-operative evaluation had documented presence of multiple visceral artery aneurysms, but no involvement of the visceral organs was seen. Further investigation was not done at that point of time to determine the etiology.
A prospective ECG-gated CT coronary angiography was performed on a 128-slice Philips Ingenuity CT after intravenous injection of 80 ml of non-ionic contrast media, for the evaluation of the exertional dyspnea and atypical chest pain. It showed a giant partially thrombosed aneurysm of the proximal – mid-left anterior descending (LAD) artery [Figure 1] which was more clearly depicted in the volume rendered image [Figure 2], a partially thrombosed aneurysm of the proximal part of 8th right intercostal artery [Figure 4]. Nodular paravertebral soft-tissue thickening adjacent to the proximal parts of 6th-10th intercostal spaces was noted [Figure 4]. CT abdominal angiography was also performed the next day, which revealed multiple aneurysms of the celiac axis, the common hepatic artery [Figure 5], inferior pancreaticoduodenal artery, and the jejunal branch of superior mesenteric artery [Figure 6]. Abnormal wall thickening or dissection of these involved vessels was not seen. CT angiography of the brain and neck vessels was normal. His serum IgG4 levels were elevated (440 mg/ dL), CRP levels were high, and levels of anti-c ANCA and anti-p ANCA titers were normal. The patient underwent conventional angiography which showed aneurysmal dilatation of LAD with contrast blush [Figure 3], followed by surgical resection of the aneurysm of LAD artery. The distal LAD was perfused by a reverse saphenous vein graft acting as a conduit from the ascending aorta. Histopathology of the resected specimen revealed chronic inflammation in the adventitia, diffuse fibrosis in media and intima with a fresh thrombus on intimal surface. The patient also underwent repair of the common hepatic artery aneurysm a month later. Post-operative periods were uneventful. Keeping in view the elevated levels of serum IgG4, the patient was put on corticosteroids therapy. The patient remained symptom-free ever since. There was a decrease in the level of CRP and the serum IgG4 (210 mg/dL) at 5-month follow-up. Imaging follow-up of the patient was not done.
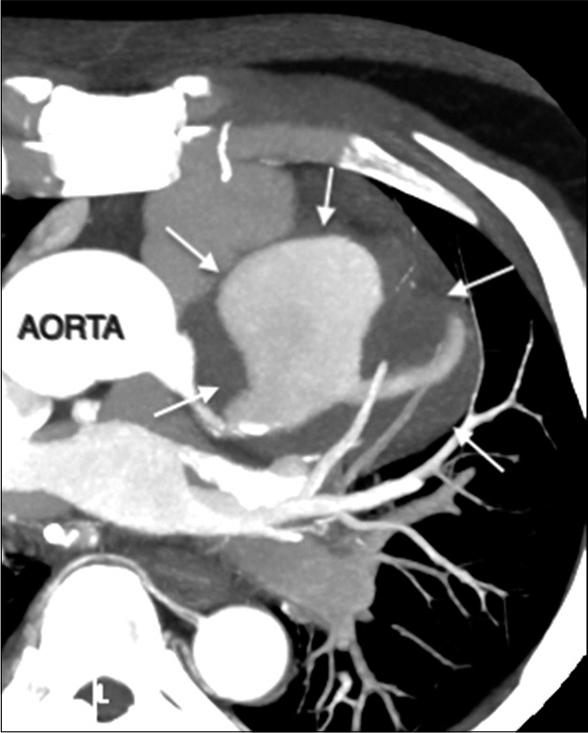
- A 55-year-old male with confirmed IgG4 vasculopathy. Axial maximum intensity projection (MIP) image of CT coronary angiography shows partially thrombosed aneurysm of the left anterior descending artery (arrows).
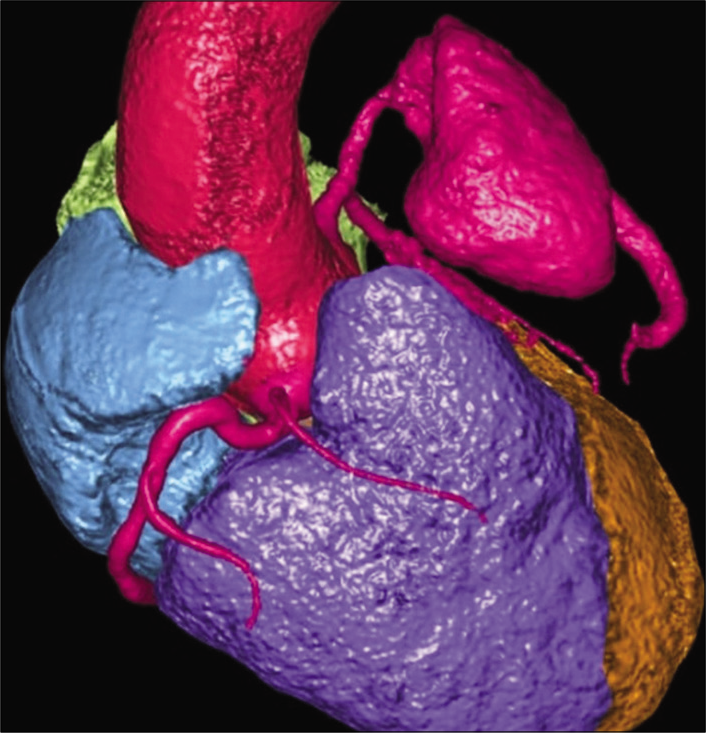
- A 55-year-old male with confirmed IgG4 vasculopathy. Volume rendered image shows the aneurysm of the left anterior descending artery (in pink).
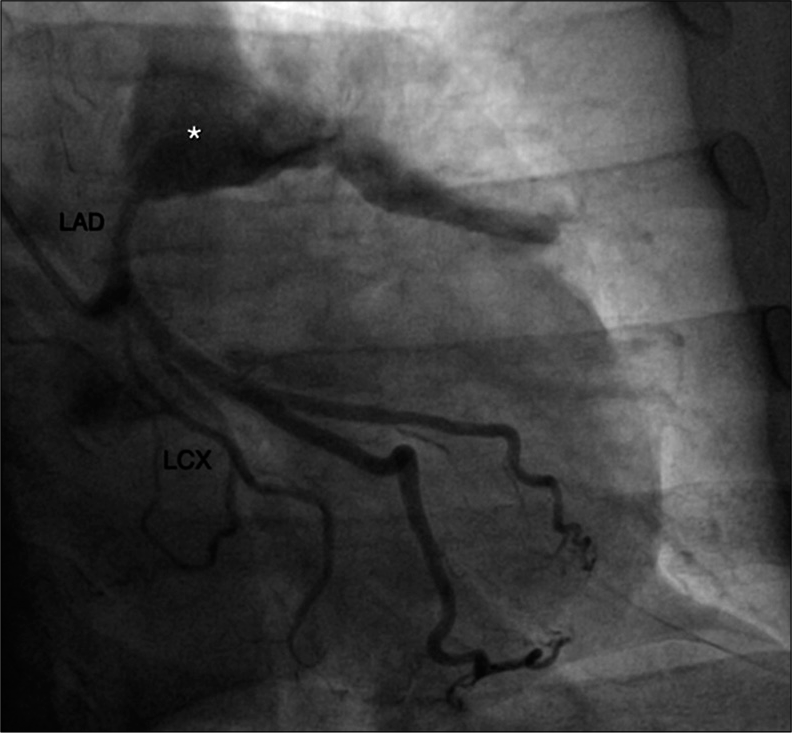
- A 55-year-old male with confirmed IgG4 vasculopathy. The right anterior oblique caudal view of conventional angiography shows aneurysmal dilatation of the left anterior descending artery with contrast blush (*).
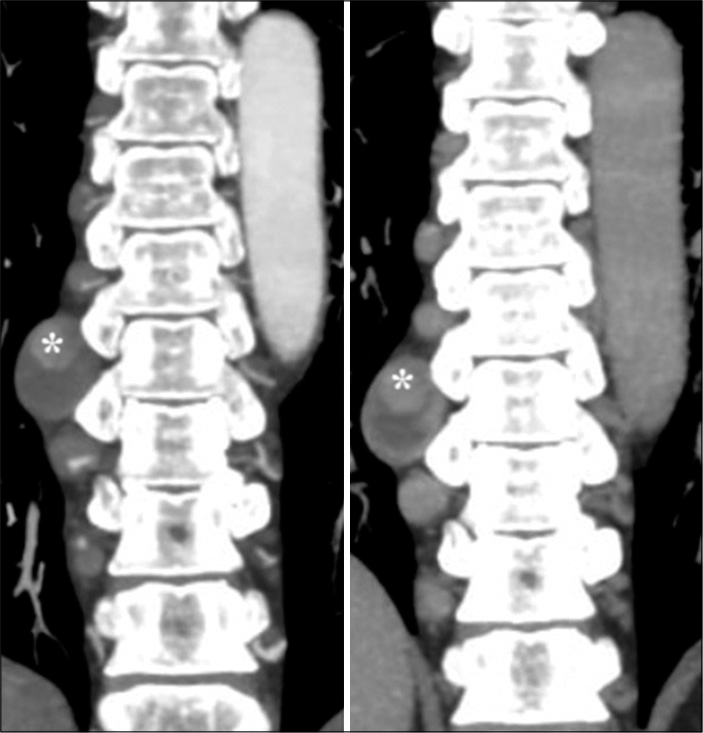
- A 55-year-old male with confirmed IgG4 vasculopathy. Coronal maximum intensity projection images of CT abdominal angiography in arterial (left) and venous (right) phases show partially thrombosed aneurysm in the proximal part of 8th right intercostal artery (*). Enhancing nodular paravertebral soft-tissue thickening is noted adjacent to the proximal right intercostal arteries above and below this level.
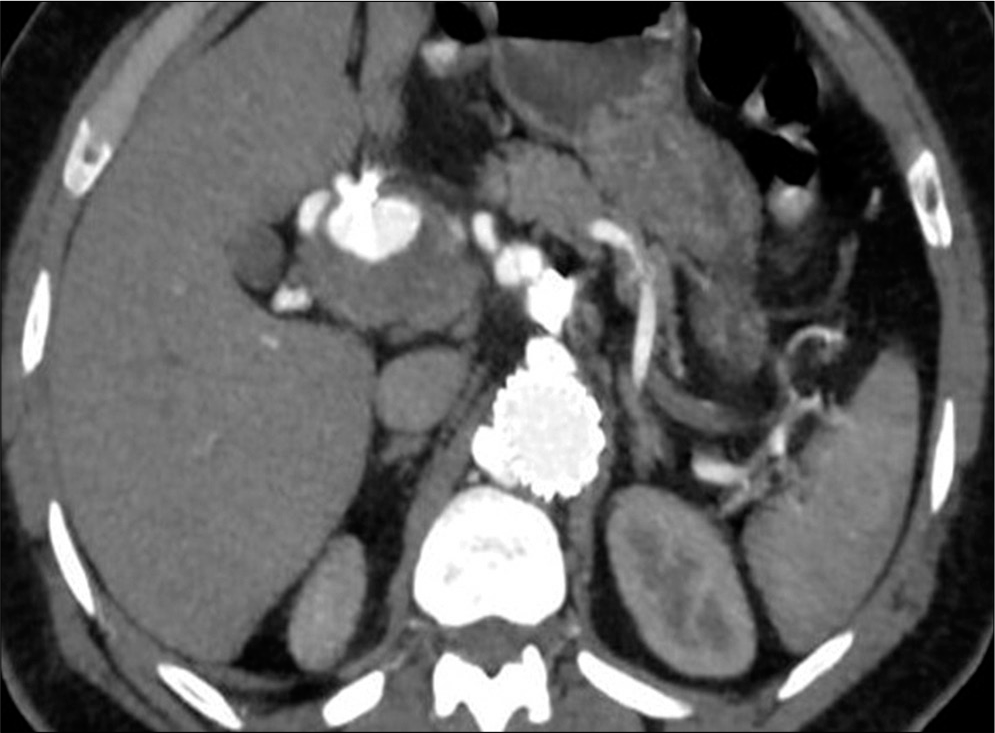
- A 55-year-old male with confirmed IgG4 vasculopathy. Axial image of CT abdominal angiography in the arterial phase shows a partially thrombosed aneurysm in relation to the common hepatic artery. Artifacts are seen in relation to the aneurysm (post-injection in the gastroduodenal artery). The aortic stent graft is also seen in situ.
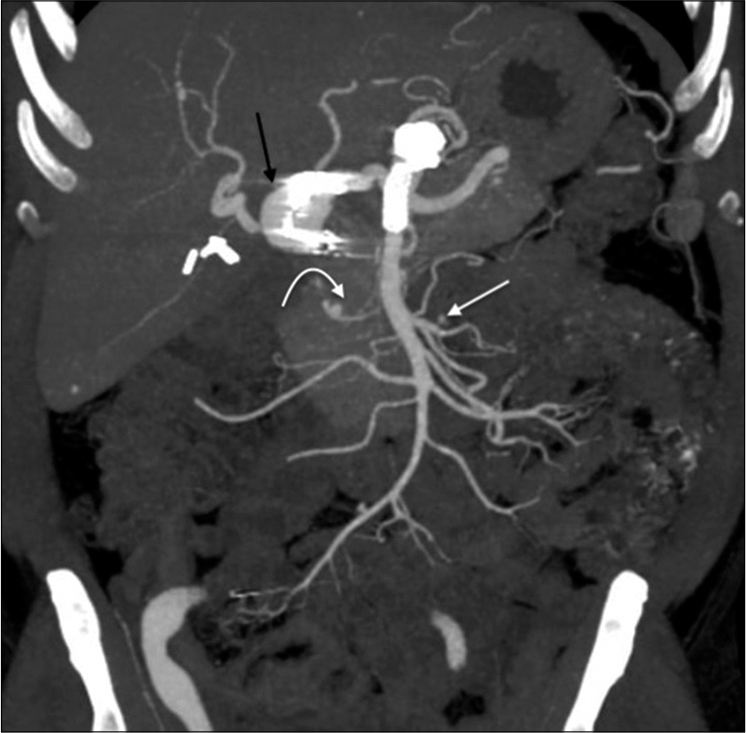
- A 55-year-old male with confirmed IgG4 vasculopathy. Coronal maximum intensity projection image of CT abdominal angiography shows multiple visceral artery aneurysms – in relation to the common hepatic artery (black arrow), inferior pancreaticoduodenal artery (white curved arrow), and the jejunal branch of superior mesenteric artery (white arrow). Artifacts are noted in relation to the gastroduodenal artery, aorta, and the superior mesenteric artery – representing stent grafts and post-procedural changes. Cholecystectomy clips are also seen.
DISCUSSION
IgG4-related disease is a chronic systemic disease that affects multiple organs in the body either synchronously or metachronously. Its diagnosis is subject to the fulfillment of certain diagnostic criteria [Table 1]. IgG4 vasculitis has three hallmark histopathological features – lymphoblastic infiltration, obliterative phlebitis, and storiform fibrosis.
| 1. Diffuse or localized swelling or masses in one or more organs on clinical examination | |
|---|---|
| 2. Elevated serum IgG4 concentrations (≥135 mg/dL) | |
| 3. Marked lymphoplasmacytic infiltration and storiform fibrosis, as well as organ infiltration by IgG4-positive plasma cells | |
| Definite | 1+2+3 |
| Probable | 1+3 |
| Possible | 1+2 |
Lymphoblastic cellular infiltration and associated sclerosing inflammation primarily cause severe thickening of the adventitia, as opposed to the involvement of intima or media in other vasculitides. The vessel involvement can present as homogenous diffuse arterial wall thickening or nonstenotic masses with irregular margins without dilatation. [3] Storiform fibrosis is characterized by collagen fibers in a radial distribution and weaves through the tissue. Small vascular lesions may also be seen as pseudotumor formation when arterial walls are thickened due to lymphosclerosing infiltration but there is no associated occlusion. This pseudotumor formation in coronary vasculature is specific for IgG4 disease and is described as “pigs-in-a-blanket” sign.
IgG4 vasculopathy most commonly affects the aorta and presents as aortic wall thickening with or without the formation of aneurysms. Aortitis and periaortitis are found in nearly 41% of cases of IgG4-related disease, and nearly 80% of these cases show involvement of multiple vessels. The order of the involvement of the arteries is the iliac arteries (35%), infrarenal aorta (33%), thoracic aorta and its first branches (8% each), and suprarenal abdominal aorta (6%) and its first branches (5%).[4] The presentation of vasculitis of mediumsized vessels as aneurysms, as seen in our case, is quite rare. In peripheral arteries such as femoral/popliteal arteries, saccular aneurysms with insignificant arterial wall thickening are noted.[5] It can affect the cerebral vessels and present as intracranial aneurysms.[6] The presence of multifocal aneurysms involving the coronary and visceral arteries, maybe seen in atherosclerosis, Kawasaki disease, IgG4-related disease, other inflammatory conditions such as Behcet’s disease, Takayasu arteritis, giant cell arteritis, and connective tissue disorders such as Marfan’s syndrome and Ehlers-Danlos syndrome.[7]
Atherosclerotic coronary artery aneurysms are commonly seen in males and usually associated with hypertension, dyslipidemia, and cigarette smoking. Moderate-to-severe atherosclerotic changes in the aorta help in the diagnosis.[8] Kawasaki disease is usually seen in children less than 5 years of age presenting with classic symptoms of fever, rash, oral mucositis, conjunctivitis, lymphadenopathy, and skin desquamation.[8] Behcet’s disease affects young males with clinical symptoms of oral, genital ulcers, and uveitis and usually associated deep venous thrombosis and pulmonary artery aneurysms.[9] Takayasu arteritis affects females, is predominantly a large vessel vasculitis involving aorta and its first-order branches with concentric mural thickening and abnormal wall enhancement. Coronary vessels involvement is seen at the ostia and proximal segments.[9] Giant cell arteritis, just like Takayasu arteritis, affects females but frequently involves the temporal arteries and the thoracic aorta. It is not associated with coronary artery aneurysms.[3]
The presence of paravertebral soft-tissue thickening in IgG4-related disease is rare with an incidence of approximately 2%. It can present as homogenously enhancing band-like, nodular or mixed lesions with a preponderance on the right side.[10] Our case showed nodular paravertebral thickening. It should be differentiated from lymphoma by excluding other coexistent features such as lymphadenopathy and splenomegaly.
Our case, documenting concomitant involvement of coronary, intercostal, and multiple visceral arteries as aneurysms, with nodular paravertebral soft-tissue thickening is quite a rare presentation of IgG4-related disease.
CONCLUSION
IgG4-related disease predominantly involving the coronary arteries is uncommon. In the presence of concomitant aneurysms in coronary artery, intercostal artery, and multiple visceral arteries along with nodular paravertebral soft-tissue thickening, IgG4 vasculopathy should be considered in the differential diagnosis of other vasculopathies.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- IgG4-related disease from head to toe. Radiographics. 2015;35:2007-25.
- [CrossRef] [PubMed] [Google Scholar]
- Recommendations for the nomenclature of IgG4-related disease and its individual organ system manifestations. Arthritis Rheum. 2012;64:3061-7.
- [CrossRef] [PubMed] [Google Scholar]
- IgG4-related cardiovascular disease from the aorta to the coronary arteries: Multidetector CT and PET/ CT. Radiographics. 2018;38:1934-48.
- [CrossRef] [PubMed] [Google Scholar]
- Characteristics of immunoglobulin G4-related aortitis/periaortitis and periarteritis on fluorodeoxyglucose positron emission tomography/computed tomography co-registered with contrast-enhanced computed tomography. EJNMMI Res. 2017;7:20.
- [CrossRef] [PubMed] [Google Scholar]
- Coronary artery aneurysm combined with other multiple aneurysms at multiple locations: A case report and systematic review. Medicine (Baltimore). 2017;96:e9230.
- [CrossRef] [PubMed] [Google Scholar]
- Spectrum of coronary artery aneurysms: From the radiologic pathology archives. Radiographics. 2018;38:11-36.
- [CrossRef] [PubMed] [Google Scholar]
- Coronary artery vasculitis: Assessment with cardiac multi-detector computed tomography. Int J Cardiovasc Imaging. 2015;31(Suppl 1):59-67.
- [CrossRef] [PubMed] [Google Scholar]
- CT findings of thoracic paravertebral lesions in IgG4-related disease. AJR Am J Roentgenol. 2019;213:W99-104.
- [CrossRef] [PubMed] [Google Scholar]






