Translate this page into:
Multiparametric Magnetic Resonance Imaging of Recurrent Prostate Cancer
Address for correspondence: Dr. Daniel Corey Oppenheimer, Department of Imaging Sciences, University of Rochester Medical Center, 601 Elmwood Ave, Rochester, NY 14642, USA. E-mail: Daniel_Oppenheimer@URMC.Rochester.edu
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Multiparametric magnetic resonance (MR) imaging of the prostate combines both morphological and functional MR techniques by utilizing small field of view T1-weighted, T2-weighted, diffusion-weighted imaging, dynamic contrast-enhanced imaging, and MR spectroscopy to accurately detect, localize, and stage primary and recurrent prostate cancer. Localizing the site of recurrence in patients with rising prostate-specific antigen following treatment affects decision making regarding treatment and can be accomplished with multiparametric prostate MR. Several different treatment options are available for prostate cancer including radical prostatectomy, external beam radiation therapy, brachytherapy, androgen deprivation therapy, or a number of focal therapy techniques. The findings of recurrent prostate cancer can be different depending on the treatment the patient has received, and the radiologist must be able to recognize the variety of imaging findings seen with this common disease. This review article will detail the findings of recurrent prostate cancer on multiparametric MR and describe common posttreatment changes which may create challenges to accurate interpretation.
Keywords
Biochemical recurrence
diffusion weighted imaging
dynamic contrast enhanced imaging
multiparametric prostate magnetic resonance

INTRODUCTION
Prostate cancer is the most common malignancy affecting males in the United States and the second most common cause of cancer death.[1] Patients with well-differentiated localized Gleason score 6 prostate cancer tend to have a good prognosis, whereas patients with Gleason score 8 or greater, extracapsular extension and/or distant metastasis have a worse prognosis. Gleason score 7 prostate cancer has a variable prognosis and intermediate risk of recurrence.[2] A variety of treatment options exists for prostate cancer including radical prostatectomy, external beam radiation therapy (EBRT), brachytherapy, androgen deprivation therapy, focal ablation therapy, or combinations thereof.[3]
Prostate magnetic resonance (MR) imaging has evolved and significantly improved since its introduction in the 1980s. Multiparametric MR of the prostate is the current state-of-the-art in prostate imaging because it has the ability to distinguish post-treatment changes from cancer recurrence. This technique involves combining the morphological information obtained from small field of view T1-weighted (T1W) and T2-weighted (T2W) images in axial, coronal, and sagittal planes with the functional information achieved with diffusion-weighted imaging (DWI), dynamic contrast-enhanced (DCE) imaging, and MR spectroscopy.[4] In addition to its role in localizing and staging prostate cancer, the development of MRI-guided targeted biopsy and MR-ultrasound fusion guided biopsy has provided an alternative to the current standard of transrectal ultrasonography-guided systematic biopsy.[56]
RECURRENCE FOLLOWING PROSTATECTOMY
Radical prostatectomy has been performed for over 100 years and remains the most common treatment option for organ-confined prostate cancer.[7] Radical prostatectomy involves surgical removal of the entire prostate, seminal vesicles, portions of the vasa deferentia, and the creation of a vesicourethral anastomosis. The neurovascular bundles are preserved whenever possible to maintain urinary continence and erectile function.
Rising serum prostate-specific antigen (PSA) following radical prostatectomy is known as biochemical recurrence, and usually precedes clinical evidence of disease progression, sometimes by many years.[8] Recurrent cancer is most frequently located near the vesicourethral anastomosis or less commonly in the seminal vesicle bed, posterior bladder wall, and anterior rectum.[9] Because of tumor neovascularity and increased permeability of the endothelial barrier, prostate cancer recurrence following radical prostatectomy is most reliably identified on DCE imaging as an area rapid contrast enhancement after the intravenous administration of gadolinium-based contrast agent [Figure 1].[1011]
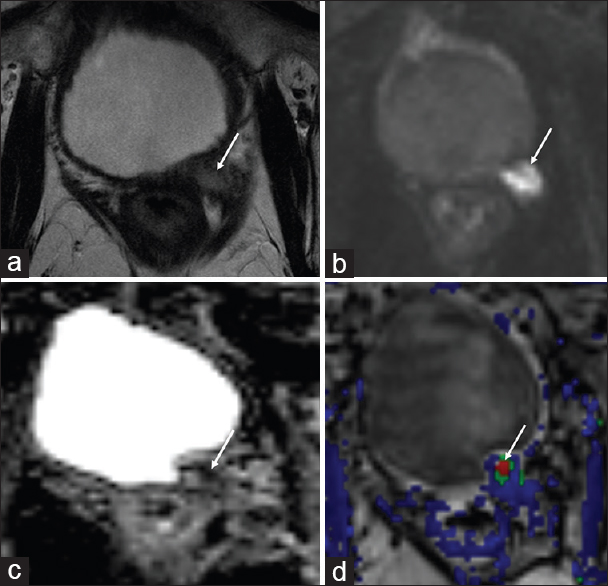
- 78-year-old male with a history of Gleason 7 prostate cancer status postradical prostatectomy in 1998 with localized recurrence in the left prostatectomy bed. (a) Axial T2-weighted, (b) diffusion weighted imaging, (c) apparent diffusion coefficient and (d) dynamic contrast enhanced images demonstrate a T2 intermediate signal nodule in the left prostatectomy bed with restricted diffusion and rapid contrast enhancement (arrows). The patient was advised to undergo pelvic irradiation but declined. He was briefly treated with androgen deprivation therapy, however subsequently declined further treatment
The presence of postoperative fibrosis may mimic recurrent tumor following radical prostatectomy, particularly if the fibrosis has a nodular morphology. DCE imaging can help distinguish fibrosis from recurrent tumor because fibrosis typically demonstrates delayed contrast enhancement, whereas prostate cancer usually has rapid contrast uptake.[12] Fibrosis is also usually lower in signal intensity compared to muscle on T1W and T2W images, whereas recurrent tumor tends to be mildly T2 hyperintense or isointense relative to muscle.
Retained seminal vesicles are observed in a significant minority of patients following prostatectomy. Distinguishing retained seminal vesicles from recurrent tumor can be challenging, especially if the normal convoluted tubular appearance and T2 hyperintensity of the seminal vesicles is lost. Hypointense signal on T1W and T2W images may be observed with fibrosis in the remnant seminal vesicles, but can also be seen with recurrent tumor.[13] Therefore, in cases where retained seminal vesicles do not maintain their normal morphology or signal intensity, careful evaluation of DWI and DCE images is critical. Retained normal seminal vesicles will not restrict diffusion, nor will they show rapid contrast enhancement [Figure 2], whereas recurrent tumor will show restricted diffusion and rapid enhancement on DCE imaging.[14]
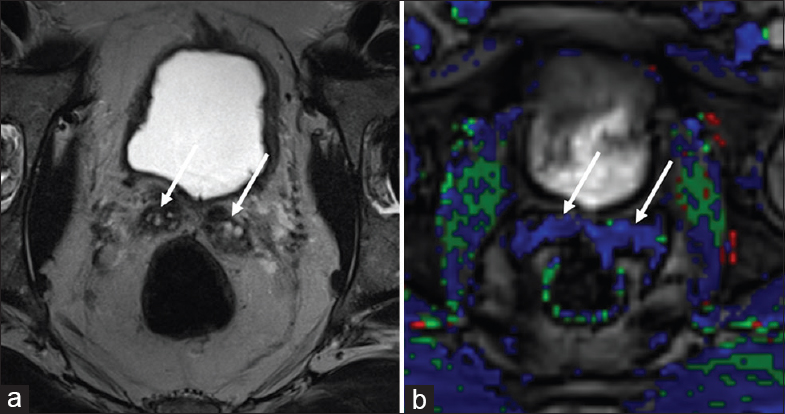
- 70-year-old male with a history of Gleason 6 prostate cancer status postprostatectomy 5 years previously with normal magnetic resonance appearance of retained seminal vesicles. (a) Axial T2-weighted and (b) dynamic contrast enhanced images through the prostatectomy bed show no residual prostate tissue status postprostatectomy. Normal T2 hyperintensity is maintained in retained seminal vesicles bilaterally, without rapid contrast enhancement to suggest recurrent tumor (arrows).
Contrast enhancement inferior to the vesicourethral anastomosis and anterior to the proximal urethra is a frequently identified normal finding in male patients who have not had prostate surgery. This observation is thought to represent physiological periurethral vascularity adjacent to the urogenital diaphragm and should not be misinterpreted as recurrent tumor following prostatectomy.[15]
MR spectroscopy has the potential to detect prostate cancer recurrence after radical prostatectomy, but may be limited by poor spatial resolution, sensitivity to field inhomogeneity, and susceptibility artifact from surgical clips in the anastomotic area.[16] In addition, MR spectroscopy is time-consuming and decreases patient throughput. Therefore, while MR spectroscopy combined with DCE and DWI can improve detection of local recurrence, some authors have suggested that MR spectroscopy is not necessary in this setting.[13]
RECURRENCE FOLLOWING EXTERNAL BEAM RADIATION THERAPY
EBRT is a noninvasive treatment option for localized and locally advanced prostate cancer. Recurrent prostate cancer after EBRT tends to occur at the site of the primary tumor.[17] Radiation to the prostate induces gland atrophy and fibrosis and causes diffusely decreased T2 signal.[18] The homogeneously decreased T2 signal observed in the prostate after EBRT results in obscuration of the normal zonal anatomy on T2W images and may impair the ability to detect recurrent tumor, which is typically T2 hypointense.[19] However, the fibrotic changes and decreased microvasculature associated with atrophic tissue following radiation tend to make the neovascularity of recurrent tumor more conspicuous relative to the remainder background atrophic tissue. Therefore, DCE imaging has excellent ability to detect recurrent tumor following EBRT and is seen as an area of rapid contrast enhancement [Figures 3 and 4].[20]
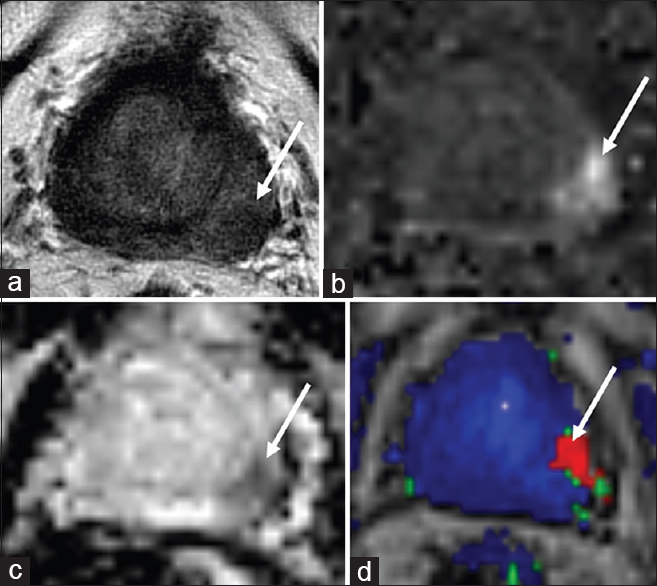
- 86-year-old male with a history of Gleason score 6 prostate cancer status postexternal beam radiation therapy in 2008 with localized recurrence in the left base peripheral zone. (a) Axial T2-weighted, (b) diffusion weighted imaging, (c) apparent diffusion coefficient and (d) dynamic contrast enhanced images demonstrate a focal T2 hypointense circumscribed nodule in the left base peripheral zone with restricted diffusion and rapid contrast enhancement (arrows). Biopsy confirmed high grade focally recurrent prostate cancer with perineural invasion. The patient was subsequently treated with high dose rate brachytherapy.
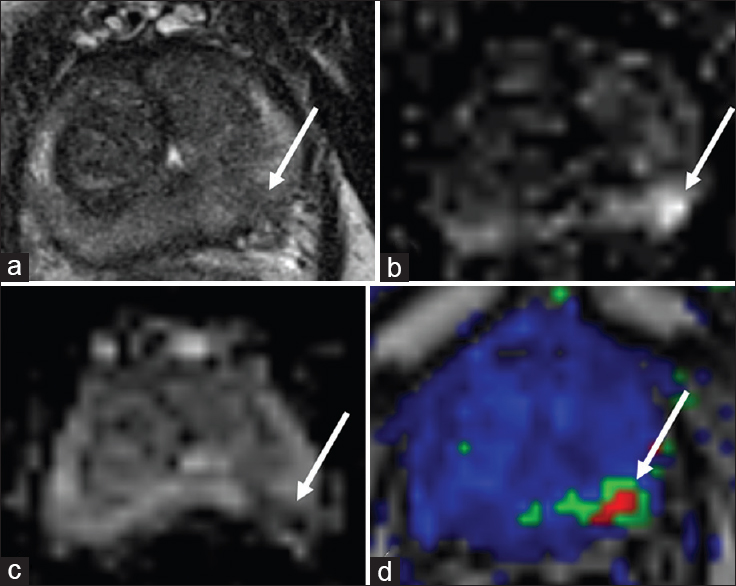
- 65-year-old male with a history of Gleason 6 prostate cancer status post with external beam radiation therapy 3 years previously with rising prostate-specific antigen and a suspicious lesion in the left mid-peripheral zone, biopsy pending. (a) Axial T2-weighted, (b) diffusion weighted imaging, (c) apparent diffusion coefficient, and (d) dynamic contrast enhanced images demonstrate an ill-defined T2 hypointense nodule in the left base peripheral zone with restricted diffusion and rapid contrast enhancement (arrows).
While it has been previously shown that combining T2W, DWI, DCE, and MR spectroscopy increases the sensitivity of detecting prostate cancer recurrence following radiation therapy,[21] there is emerging evidence to suggest that DCE can be omitted from the protocol and still detect cancer recurrence with high sensitivity.[22] Donati et al.[22] found that the combination of T2W and DWI achieved the highest diagnostic accuracy and inter-reader agreement in the detection of prostate cancer after radiation therapy. The addition of DCE to T2W and DWI did not significantly improve the detection of recurrent prostate cancer in their cohort. They conclude that in this clinical context, DCE imaging can be omitted from the protocol without sacrificing diagnostic performance.
Performing a noncontrast prostate MR examination has several potential advantages, including eliminating the risks and costs associated with intravenous administration of gadolinium-based contrast agents in addition to shorter scan times, which may increase patient throughput. In addition, DCE is limited in patients who have recently undergone EBRT because the prostate tissue develops an inflammatory reaction which can result in increased perfusion and blood volume. For this reason, prostate MR with DCE imaging should be performed at least 3 months following radiation therapy. Since DWI has shown excellent ability to detect and localize recurrent prostate cancer following EBRT,[2123] a noncontrast prostate MR examination may be performed at the request of the referring physician soon after completing EBRT and still maintain a high degree of accuracy.
RECURRENCE FOLLOWING BRACHYTHERAPY
Prostate brachytherapy involves implanting either a permanent low dose rate (LDR) or temporary high dose rate (HDR) radiation source within or around the prostate in an effort to cause cancer cell death. Although LDR prostate brachytherapy has been the gold standard for many years and is generally of low morbidity, HDR prostate brachytherapy possesses the advantage of flexibility of dosimetry by modulating the source dwell time and position.[24]
As with EBRT, prostate cancer recurrence following brachytherapy tends to occur at the same site as the original tumor. LDR and HDR brachytherapy both cause diffuse hypointense T2 signal in the prostate gland and loss of the normal zonal anatomy, which limits morphologic imaging sequences in the detection of recurrence.[18] DWI is also suboptimal following LDR brachytherapy because the metallic seed implants may create susceptibility artifacts and image distortion. Similarly, MR spectroscopy is limited by magnetic field inhomogeneities, susceptibility artifacts, and decreased signal to noise ratio related to brachytherapy seeds. Therefore, DCE is a critical sequence in the multiparametric prostate MR exam in order to detect recurrence following LDR permanent brachytherapy and is characterized by an area of rapid contrast enhancement and early washout [Figures 5 and 6].[1319]
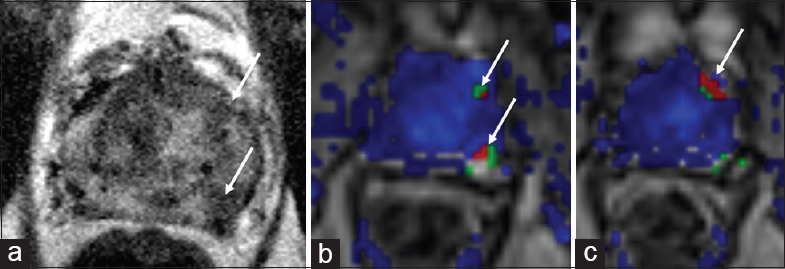
- 77-year-old male with a history of Gleason 6 prostate adenocarcinoma treated with low dose rate brachytherapy in 2005 with rising prostate-specific antigen and suspicious lesions in the left mid-peripheral zone. (a)_Axial T2-weighted and (b and c) dynamic contrast enhanced images demonstrate two circumscribed T2 hypointense nodules in the left mid peripheral zone with rapid contrast enhancement (arrows). Biopsy was not pursued as the patient did not desire salvage therapy and was deemed to be a poor candidate due to comorbidities.
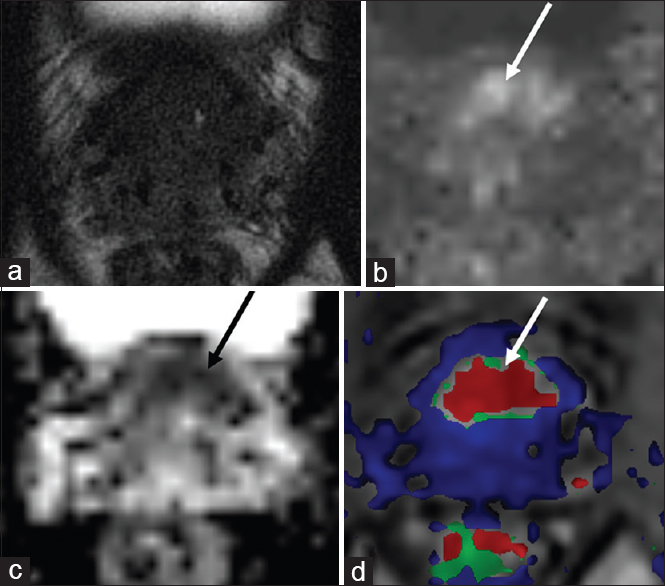
- 72-year-old male with a history of Gleason score 6 prostate cancer status post low dose rate brachytherapy in 2003 with rising prostate-specific antigen 11 years following treatment and findings consistent with recurrence in the anterior base transition zone. (a) Axial T2-weighted, (b) diffusion weighted imaging, (c) apparent diffusion coefficient, and (d) dynamic contrast enhanced images demonstrate a large area of restricted diffusion in the anterior base transition zone with associated rapid contrast enhancement (arrows). No discrete signal abnormality is seen in this region on T2-weighted images, but the zonal anatomy is obscured due to postradiation changes.
In contrast, temporary HDR prostate brachytherapy is not affected by susceptibility artifacts because no metallic material is retained. Data are limited related to multiparametric prostate MR following temporary HDR prostate brachytherapy. However, a recent study by Tamada et al. evaluated the specificity and sensitivity of locally recurrent prostate cancer after HDR brachytherapy.[25] As expected, the sensitivity of T2W imaging in their study was low because diffuse hypointense T2 signal in the prostate after radiation therapy decreases the inherent contrast between recurrent tumor and surrounding benign tissues. However, hypervascular recurrent cancer related to tumor neovascularity was conspicuous against the hypoenhancing fibrotic adjacent tissues after HDR brachytherapy, similar to recurrent tumor after other forms of radiation treatment. Importantly, also DWI showed high sensitivity for detecting recurrent prostate cancer after HDR brachytherapy because it is not prone to the susceptibility artifacts and signal distortion which degrade these sequences in patients following LDR brachytherapy.[26]
RECURRENCE FOLLOWING FOCAL ABLATION THERAPY
As small volume prostate cancers are detected earlier through PSA screening and multiparametric prostate MR, focal ablation therapy for prostate cancer is gaining popularity as a minimally invasive alternative to radical prostatectomy. Focal therapy techniques include laser ablation, radiofrequency ablation, cryotherapy, high-intensity focused ultrasound (HIFU), irreversible electroporation, and photodynamic therapy. Posttherapy changes that may be identified include hypointense T2 signal foci, loss of zonal anatomy, thickening of the prostatic capsule, and periprostatic fibrosis and scarring.[11]
The current evidence for utilizing multiparametric prostate MR to detect recurrence following focal therapy is limited. Recurrence after focal ablation appears to be most accurately detected on DCE imaging as a region of rapid contrast wash-in and wash out [Figure 7].[26] Lack of enhancement on DCE performed soon after treatment is suggestive of technical success, but follow-up MR 6 months after therapy is generally recommended to confirm that the tumor has been adequately treated.[27] However, reactive enhancing regions of prostate tissue related to the treatment may be difficult to distinguish from residual viable tumor, particularly at the margins of a treated lesion.[27] The typical features of recurrence seen on T2W images (i.e., focal hypointense nodule) may be absent following focal ablation therapy, which can also confound the interpretation.[28]
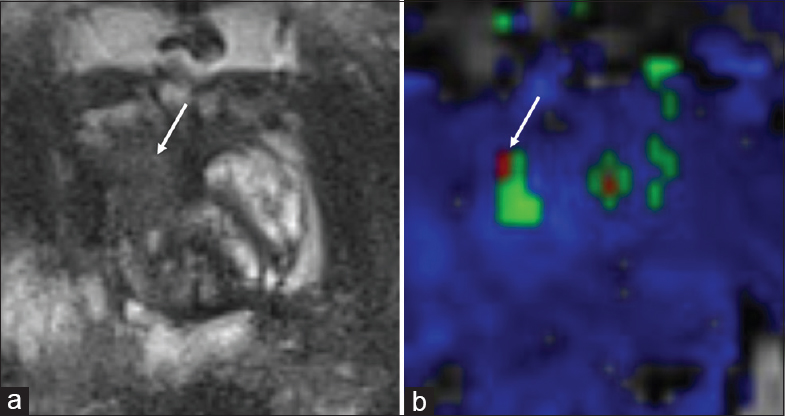
- 73-year-old male with a history of Gleason 7 prostate cancer treated with focal laser ablation therapy with rising prostate-specific antigen and findings suspicious for recurrence in the right anterior mid-peripheral zone. (a) Axial T2-weighted and (b) dynamic contrast enhanced images demonstrate a region of T2 hypointense signal in the right anterior mid-peripheral zone consistent with posttreatment changes (arrow), but within this region there is a small focus of rapid contrast wash-in and wash out (arrow), suspicious for recurrent tumor.
Although data are limited, evidence suggests that DCE is more sensitive but less specific than T2W and DWI for predicting local tumor progression after HIFU.[29] The use of MR spectroscopy to detect recurrence following focal therapy has not been studied sufficiently to impact clinical decision making and is not currently recommended in this setting.
CONCLUSION
This review article details the findings of recurrent prostate cancer on multiparametric prostate MR following radical prostatectomy, EBRT, brachytherapy, and focal ablation therapy. Important pitfalls and posttreatment changes that may mimic recurrence were also described. The use of multiparametric MRI in the detection, localization, and staging of primary and recurrent prostate cancer continues to improve, and a solid grasp of the above material will aid radiologists in their effort to provide accurate imaging interpretations for referring physicians to positively impact patient care.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/18/181494
REFERENCES
- American Cancer Society. Cancer Facts and Figures 2016. Atlanta: American Cancer Society; 2016.
- 2015. PI-RADS Prostate Imaging and Reporting and Data System: Version.2. American College of Radiology. Available from: http://www.acr.org/~/media/ACR/Documents/PDF/QualitySafety/Resources/PIRADS/PIRADS%20V2.pdf
- Systematic review: Comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann Intern Med. 2008;148:435-48.
- [Google Scholar]
- A clinically relevant approach to imaging prostate cancer: Review. AJR Am J Roentgenol. 2011;196(3 Suppl):S1-10.
- [Google Scholar]
- Magnetic resonance imaging guided prostate biopsy in men with repeat negative biopsies and increased prostate specific antigen. J Urol. 2010;183:520-7.
- [Google Scholar]
- MRI-ultrasound fusion for guidance of targeted prostate biopsy. Curr Opin Urol. 2013;23:43-50.
- [Google Scholar]
- The changing face of low-risk prostate cancer: Trends in clinical presentation and primary management. J Clin Oncol. 2004;22:2141-9.
- [Google Scholar]
- Management of biochemically recurrent prostate cancer after local therapy: Evolving standards of care and new directions. Clin Adv Hematol Oncol. 2013;11:14-23.
- [Google Scholar]
- Endorectal magnetic resonance imaging at 1.5 Tesla to assess local recurrence following radical prostatectomy using T2-weighted and contrast-enhanced imaging. Eur Radiol. 2009;19:761-9.
- [Google Scholar]
- Role of dynamic contrast-enhanced magnetic resonance (MR) imaging and proton MR spectroscopic imaging in the detection of local recurrence after radical prostatectomy for prostate cancer. Eur Urol. 2008;54:589-600.
- [Google Scholar]
- The normal post-surgical anatomy of the male pelvis following radical prostatectomy as assessed by magnetic resonance imaging. Eur Radiol. 2008;18:1281-91.
- [Google Scholar]
- Retained seminal vesicles after radical prostatectomy: Frequency, MRI characteristics, and clinical relevance. AJR Am J Roentgenol. 2006;186:539-46.
- [Google Scholar]
- Pitfalls in Interpreting mp-MRI of the prostate: A pictorial review with pathologic correlation. Insights Imaging. 2015;6:611-30.
- [Google Scholar]
- Detection of local recurrent prostate cancer after radical prostatectomy in terms of salvage radiotherapy using dynamic contrast enhanced-MRI without endorectal coil. Radiat Oncol. 2012;7:185.
- [Google Scholar]
- Multiparametric MRI for recurrent prostate cancer post radical prostatectomy and postradiation therapy. Biomed Res Int 2014 2014 316272
- [Google Scholar]
- Clinically significant prostate cancer local recurrence after radiation therapy occurs at the site of primary tumor: Magnetic resonance imaging and step-section pathology evidence. Int J Radiat Oncol Biol Phys. 2007;69:62-9.
- [Google Scholar]
- Brachytherapy for prostate cancer: Endorectal MR imaging of local treatment-related changes. Radiology. 2001;219:817-21.
- [Google Scholar]
- Imaging of prostate cancer local recurrences: Why and how? Eur Radiol. 2010;20:1254-66.
- [Google Scholar]
- Accuracy of multiparametric magnetic resonance imaging in detecting recurrent prostate cancer after radiotherapy. BJU Int. 2010;106:991-7.
- [Google Scholar]
- Comparative sensitivities of functional MRI sequences in detection of local recurrence of prostate carcinoma after radical prostatectomy or external-beam radiotherapy. AJR Am J Roentgenol. 2013;200:W361-8.
- [Google Scholar]
- Multiparametric prostate MR imaging with T2-weighted, diffusion-weighted, and dynamic contrast-enhanced sequences: Are all pulse sequences necessary to detect locally recurrent prostate cancer after radiation therapy? Radiology. 2013;268:440-50.
- [Google Scholar]
- Incremental value of diffusion weighted and dynamic contrast enhanced MRI in the detection of locally recurrent prostate cancer after radiation treatment: Preliminary results. Eur Radiol. 2011;21:1970-8.
- [Google Scholar]
- Low-dose-rate or high-dose-rate brachytherapy in treatment of prostate cancer - Between options. J Contemp Brachytherapy. 2013;5:33-41.
- [Google Scholar]
- Locally recurrent prostate cancer after high-dose-rate brachytherapy: The value of diffusion-weighted imaging, dynamic contrast-enhanced MRI, and T2-weighted imaging in localizing tumors. AJR Am J Roentgenol. 2011;197:408-14.
- [Google Scholar]
- Dynamic contrast-enhanced MRI of prostate cancer at 3 T: A study of pharmacokinetic parameters. AJR Am J Roentgenol. 2007;189:849.
- [Google Scholar]
- The role of magnetic resonance imaging (MRI) in focal therapy for prostate cancer: Recommendations from a consensus panel. BJU Int. 2014;113:218-27.
- [Google Scholar]
- Prostate carcinoma: MR imaging findings after cryosurgery. Radiology. 1996;198:807-11.
- [Google Scholar]
- MRI techniques for prediction of local tumor progression after high-intensity focused ultrasonic ablation of prostate cancer. AJR Am J Roentgenol. 2008;190:1180-6.
- [Google Scholar]






