Translate this page into:
Management of Bleeding Duodenal Varices with Combined TIPS Decompression and Trans-TIPS Transvenous Obliteration Utilizing 3% Sodium Tetradecyl Sulfate Foam Sclerosis
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Objectives:
Endoscopic experience in the management of duodenal varices (DVs) is limited and challenging given the anatomic constraints and limited experience. The endovascular management of DVs is not yet established and the controversy of whether to manage them by decompression with a transjugular intrahepatic portosystemic shunt (TIPS) or by transvenous obliteration is unresolved. In the literature, the 6–12 month rebleeding rate of DVs after TIPS is 21-37% and after transvenous obliteration is 13%. The purpose of the study is to evaluate the clinical outcome of combined TIPS decompression and transvenous obliteration/sclerosis.
Materials and Methods:
This is a retrospective study (case series) of two institutions, evaluating patients who underwent TIPS and/or transvenous obliteration/sclerosis for bleeding DVs (from January 2009 to June 2013). TIPS was performed according to a standard procedure using covered stents. Transvenous obliteration (variceal sclerosis) from the systemic and/or portal venous circulation was performed utilizing 3% sodium tetradecyl sulfate foam. Transvenous obliteration was commonly augmented with coils and/or vascular plugs. Technical (technical success of establishing TIPS and completely obliterating the DVs) and clinical outcomes (rebleeding rate and survival) were evaluated.
Results:
Five patients with liver cirrhosis presenting with bleeding DVs were included in the study with all eventually (and coincidentally) receiving TIPS and transvenous obliteration. Two of the five patients underwent concomitant TIPS and transvenous obliteration in the same procedural setting. However, three patients underwent transvenous obliteration due to bleeding despite a patent TIPS that had been previously placed. The average time from TIPS placement to transvenous obliteration was 125 days (range: 3-324 days). After having both procedures, there was no rebleeding in the patients during a mean follow-up period of 22 months (6–50 months). Coils and/or metallic vascular plugs were used to augment the sclerosant obliteration in four of five patients.
Conclusion:
The combination of TIPS decompression and foam sclerosant transvenous obliteration appears to be effective in preventing rebleeding in this limited case series and compares favorably with the existing evidence for either approach [TIPS or balloon-occluded retrograde transvenous obliteration (BRTO)] alone.
Keywords
Balloon-occluded retrograde transvenous obliteration
bleeding
duodenal varices
vascular plugs
INTRODUCTION

Duodenal varices (DVs) are dilated meso-portal varicosities and/or are dilated portosystemic collaterals in the submucosa of the duodenal wall.[1] DVs represent 17% of ectopic varices.[23] Moreover, DVs have a fourfold risk of bleeding when compared to esophageal varices[4] and can have a mortality rate of up to 40%.[1567] The experience with medical management and endoscopic management (mostly direct endoscopic-guided sclerosant injection) of DVs is limited and found to be usually not as effective as they are for esophageal varices, due to anatomical and hemodynamic reasons.[1257] From an endovascular standpoint, the controversy of whether to manage these varices by decompression with a transjugular intrahepatic portosystemic shunt (TIPS) versus transvenous obliteration is unresolved.[1] Unfortunately, the number of reported cases of DVs in the literature are small and the experience is scant precluding a definite consensus for the ideal approach to management. The rebleed rate of DVs after TIPS is 21–37% and after transvenous obliteration is 13%.[1]
The aim of this study is to describe the techniques and review the clinical outcomes of combined TIPS decompression and transvenous sclerosis utilizing 3% sodium tetradecyl sulfate (STS) foam of DVs.
MATERIALS AND METHODS
Study design
This is a retrospective study of consecutive adult patients undergoing TIPSs and/or transvenous obliteration (sclerosis) for DVs from January 2009 to June 2013 at two academic centers. Patients suffering from bleeding DVs and managed by means other than TIPS and/or transvenous obliteration were excluded from the study. Institutional review board approval was obtained.
Description and classification of duodenal varices
DVs were categorized by contrast-enhanced computer tomography (CT) and endoscopy to the duodenal bulb (D1), second (D2), third (D3), and fourth (D4) portions of the duodenum. The actual bleeding site of DVs was also noted based on the above four sites (D1, D2, D3, and D4). The portal and portosystemic hemodynamics (and thus, the hemodynamic-based management) were classified according to the hemodynamic classification of ectopic varices of Saad and coworkers [Table 1].[1]

TIPS and transvenous obliteration techniques
The TIPSs were placed with the traditional right internal jugular approach utilizing a Colapinto needle (Cook Corp., Bloomington, IN, USA). An 8 mm or 10 mm Viatorr stent (Gore and Associates, Flagstaff, AZ, USA) was deployed to bridge the right portal vein to the right hepatic vein. Transvenous obliteration of the DVs was performed utilizing 3% sodium tetradecyl sulfate (STS) (AngioDynamics, Queensbury, NJ, USA) foam sclerosant. The composition of the foam sclerosant was 1 part lipiodol (Lipiodol UltraFluide; Guerbet, Cedex, France), 2 parts 3% STS, and 3 parts air. All transvenous obliterations were performed via the TIPS in a balloon-occluded antegrade transvenous obliteration (BATO) approach. Occasionally, a concomitant gonadal vein (systemic) balloon-occluded retrograde transvenous obliteration (BRTO) approach was performed.[1] Balloon occlusion during transvenous obliteration was commonly, but not necessarily, performed using a 10-mm or 11-mm-diameter balloon occlusion catheters (Cook Flow Catheter; Cook Corp., Bloomington, IN, USA and Python Catheter; Applied Medical, Santa Margareta, CA, USA, respectively). The remaining patients (without balloon occlusion) underwent catheter-directed sclerosis interlaced with 0.035-inch coils (Cook Corp.) and/or Amplatzer vascular plugs (AVP) (AGA Medical Corp., Plymouth, MN, USA) instead of balloon occlusion with balloon occlusion catheters. This was because there were no micro-balloons available to navigate the tortuous collaterals/varices to balloon occlude them. The coils and vascular plugs were used (interlaced with the foam sclerosant) to maximize the sclerosant dwell time.[1]
RESULTS
Five patients were included in the study had undergone DVs sclerosis and/or TIPS and all five had both procedures (no patients had undergone only one of the two procedures). Two additional patients were found with bleeding DVs requiring blood transfusions and leading to hemodynamic instability. However, they were not treated with TIPS or transvenous sclerosis. These two excluded patients had chronic extrahepatic portal vein thrombosis and were treated with antegrade and retrograde recanalization and stent placement of the mesenteric and portal veins. The description of the five cases is presented in Tables 2 and 3. The demographics of the five patients and the description and hemodynamic classification of the DVs are presented in Table 2. The technical details and clinical outcomes are presented in Table 3. The clinical outcomes which were the rebleed rate (from any source) and the patient survival were noted.


Patient 1
Patient 1 is a 28-year-old female who presented post right hepatic lobe transplant done 4 months earlier and had recurrent bleeding episodes requiring repeated blood transfusion with at least one episode of hemodynamic instability [Table 2]. Her Child–Pugh score classification was “A”. The cause of her apparent recurrent post-transplant portal hypertension was a small-for-size (undersized) graft [Table 2]. During endoscopy, the DVs were noted in the second (D2) and third (D3) portions of the duodenum, but the source of bleeding was localized to the varices in the second portion of the duodenum [Table 2]. A TIPS was performed despite the lack of a pre-TIPS portosystemic gradient (PSG; zero mmHg) [Table 3]. The lack of gradient was due to significant portosystemic shunting involving the DVs and the portal vein was widely patent (Saad ectopic classification: Type 3A) [Tables 1 and 2]. The transvenous obliteration was performed concomitantly to specifically manage (obliterate) the DVs and occlude the portosystemic shunting that was hemodynamically competing with the newly established TIPS [Table 3]. The transvenous obliteration was performed via the TIPS (portal or BATO approach). No balloons were used; however, metallic coils and vascular plugs were used to establish stasis and maximize the sclerosant dwell time [Table 3]. In retrospect, some of the vascular plugs and coils were deposited in the submucosa of the duodenum. In the following 50.4 months, there was no clinical evidence of bleeding and the TIPS was patent; however, routine follow-up endoscopy demonstrated erosion of the vascular plugs 103 days after the combined TIPS and transvenous obliteration [Table 3]. No significant bleeding was noted by endoscopy. The vascular plugs were removed without bleeding.
Patient 2
Patient 2 is a 48-year-old male with alcohol-related cirrhosis, who had endoscopically worsening DVs over a 3-month period after BRTO for bleeding gastric varices. The gastric varices had bled despite a patent TIPS established approximately 7.5 months earlier. The overall time lapse between the TIPS and the duodenal transvenous obliteration was 324 days [Table 2]. Due to the size of the varices, the progressive growth of the varices over 3 months, and the overlying erythematous mucosa, the varices were considered high risk with potential for impending bleeding [Table 2]. The patient had an insignificant (<50% diameter reduction) main portal vein stenosis as a result of partial portal vein thrombosis. The portal vein was balloon-dilated with an 18 mm balloon to balloon-macerate the partial thrombus in an attempt to optimize portal hemodynamics; this was successful. His Child–Pugh score at the time of the transvenous obliteration was classified as “B”. The transvenous obliteration was performed via the pre-established TIPS (established 324 days prior) in a portal/BATO approach manner utilizing a balloon occlusion catheter (Cook Flow Catheter, Cook Corp.) and coils. The metallic coils and vascular plugs were used to establish stasis and maximize the sclerosant dwell time, and they were intentionally placed in collaterals far away from the submucosal varices [Figure 1 and Table 3] to avoid the iatrogenic erosion encountered in patient 1. During the procedure, venography demonstrated porto-portal collaterals (early cavernous transformation) and a single portosystemic [varices to inferior vena cava (IVC)] collateral. However, there was no significant portosystemic shunting through that portosystemic collateral [Figure 1]. The patent portal (<50% diameter reduction) and the significant porto-portal shunting in the presence of an insignificant portosystemic collateral places the DVs as category 2A [Table 1]. In the following 30.4 months, there was no bleeding from the gastric or duodenal varices [Table 3].
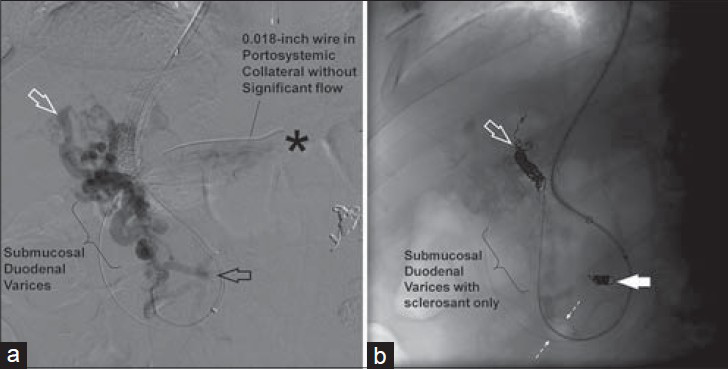
- Procedural images for patient 2 [Table 2]. (a) Digitally subtracted angiogram of the mesenteric to intrahepatic portal collateral with the 5-French catheter in the mesenteric side of the collateral. Essentially, this is a partly cavernose portal vein with partial portal vein thrombosis (not shown). The bracket points to where the meso-portal collaterals (duodenal varices) run through the duodenal wall. Above the bracket is the hepatic hilum and below it is extraduodenal. This segment of the collaterals (delineated by the bracket) should not have the “hardware” (in the form of coils or metallic vascular plugs) deployed within it as it is a high risk for mucosal erosion into the lumen of the duodenum. The 0.018-inch guide wire is in a portosystemic collateral that exhibits negligible, if any, portosystemic shunting. The asterisk denotes where the contrast (with powerful injections) empties into the inferior vena cava (systemic circulation). The hollow black arrow points to a portal communicator and the hollow white arrow points to the intrahepatic portal outflow of the duodenal varices. b) Fluoroscopic image of the coil augmented trans-TIPS balloon-occluded antegrade transvenous obliteration (BATO). There are coils (b: Solid white arrow), the portal communicator (a: Hollow black arrow), and coils (b: Hollow white arrow) in part of the intrahepatic portal outflow (a: Hollow white arrow). An air-filled 10 mm balloon (b: Between dashed arrows) is seen occluding the main portal contributor to the duodenal varices. Sclerosant (double density) is seen filing and spanning the duodenal varices between the balloon and the two coil nest. The deployed coils are clear of the submucosal/intramural portion of the porto-portal collaterals. Duodenal varices (bracket).
Patient 3
Patient 3 is a 48-year-old male, with hepatitis C related cirrhosis, who had several episodes of bleeding with instability and chronic blood transfusion requirements [Table 2]. The exact source of bleeding was not determined and he underwent at least two arterial mesenteric angiograms in an attempt to identify the bleeding source. No active arterial bleeding sources were identified; however, in retrospect, the portal venous phase of the angiograms did demonstrate a tuft of venous structures (varices) in the right upper quadrant of the abdomen. CT and endoscopy identified the DVs to be in second (D2) and third (D3) portions of the duodenum and endoscopy localized the bleeding in D2 section [Table 2]. The patient underwent a TIPS procedure in an attempt to decompress the varices. The initial pre-TIPS PSG was 33 mmHg and post TIPS, the PSG went down to 9 mmHg [Table 3]. Despite a hemodynamically successful TIPS with a PSG of 9 mmHg, the patient continued to bleed due to which he became unstable and required blood transfusions. His Child–Pugh score at the time of the transvenous obliteration was classified as “B”. Three days after the TIPS, he underwent a trans-TIPS BATO combined with a transcaval BRTO approach. No balloon occlusion was used; however, coils used in both the BRTO and BATO approaches were interlaced with the sclerosant foam. The coils were intentionally placed in collaterals at a distance from the submucosal varices to avoid the iatrogenic erosion encountered in patient 1. The patent portal vein and the significant portosystemic shunting place the DVs as category 3A [Table 1]. In the following 13.9 months, there was no bleeding from the DVs [Table 3].
Patient 4
Patient 4 is a 37-year-old male with cryptogenic cirrhosis, who had chronic intermittent bleeding and required frequent blood transfusions [Table 2]. CT identified the DVs to be in second (D2) and third (D3) sections of the duodenum and endoscopy localized the bleeding in D2 [Table 2]. The patient underwent a TIPS procedure in an attempt to decompress the varices. The initial pre-TIPS PSG was 14 mmHg and post TIPS, the PSG went down to 2 mmHg [Table 3]. Despite a hemodynamically successful TIPS, with a PSG of 2 mmHg, the patient continued to bleed requiring blood transfusions. His Child–Pugh score at the time of the transvenous obliteration was classified as “B”. Forty-nine days after the TIPS, he underwent a trans-TIPS BATO utilizing balloon occlusion. Coils were not used [Table 3]. The patent portal vein and the significant portosystemic shunting place the DVs as category 3A [Table 1]. In the following 9.5 months, there was no bleeding from the DVs [Table 3].
Patient 5
Patient 5 is a 50-year-old female with cirrhosis-related primary sclerosing cholangitis who presented with acute portal vein thrombosis, upper gastrointestinal tract bleeding, and hemodynamic instability [Table 2]. Her Child–Pugh score at the time of the transvenous obliteration was classified as “B”. CT confirmed the portal vein thrombosis and identified the bleeding DVs to be in the second (D2) portion of the duodenum [Table 2]. The patient underwent a (8 mm Viatorr stent dilated to 6 mm) TIPS procedure and portal vein thrombolysis. The thrombolysis (transcatheter fibrinolysis) was performed utilizing 8 mg of tissue plasminogen activator (tPA) power-pulse Possis AngioJet. Both the TIPS and portal vein thrombolysis were technically and hemodynamically successful. The initial PSG was 17 mmHg and post TIPS and thrombolysis, the PSG went down to 4 mmHg [Table 3]. Due to the initially thrombosed (occluded) portal vein and the significant portosystemic shunting encountered during the procedure, the DVs are categorized as 3B [Table 1]. The patient underwent a transvenous sclerosis with a balloon from a trans-TIPS approach (BATO) aided with coils that were placed away from the submucosal varices. In the following 6 months, there was no bleeding from the DVs [Table 3].
DISCUSSION
Majority of DVs are simply due to generalized portal hypertension; however, not uncommonly (more than 30%), they are associated or accentuated by focal pathology that localizes the varices to the duodenum.[1] Based on these two etiologies, there are two etiologic types: Oncotic or non-occlusive type (due to generalized portal hypertension) and occlusive type (due to mesenteric and/or portal vein thrombosis) [Table 1].[1] The most common locations of DVs are in the first and second portions of the duodenum, although they can also be seen in rare cases in the third and fourth portions of the duodenum.[1] In the five patients in the study, all patients had DVs in the second portion and three patients had additional extension of the DVs to the third portion of the duodenum. However, the site of bleeding in all five patients was identified in the second portion of the duodenum [Table 3].
DVs are thin-walled porto-portal and/or portosystemic retroperitoneal collaterals.[1] The portal venous suppliers of DVs include the pancreaticoduodenal veins, cystic branches from the superior mesenteric veins, gastroduodenal vein (s), and pyloric vein.[18] The systemic venous draining veins of DVs are commonly the gonadal veins (especially the right gonadal vein) and the capsular renal veins which then drain into the IVC.[18] The left gonadal vein can be involved in DVs that are in the third and fourth portions of the duodenum. Direct venous drainage into the IVC or right renal vein can also be observed.[1] These are consistent with the findings of this study, where 60% (n = 3/5) of patients had DVs drained by the right gonadal vein and 40% (n = 2/5) of patients had DVs drained directly into the IVC [Table 2].
The standard protocol for management of DVs is yet to be established.[56] Medical management includes fluid resuscitation and the administration of systemic vasopressin and octreotide.[24] Endoscopic-guided management includes injection of thrombin, N-butylcyanoacrylate, histoacryl, banding, and sclerotherapy.[14] Long-term hemostasis is difficult to achieve by medical and endoscopic means alone and bleeding after endoscopic management is common.[17] Surgical management includes resection and/or ligation of varices and duodenectomy.[579] The morbidity and mortality associated with surgical management of DVs is probably high in cirrhotic patients.[1]
The standard endovascular management of DVs is not established.[56] It is difficult to ascertain the ideal management strategy for DVs since the literature consists of case reports and small case series.[4] Moreover, there are no standards of reporting and the small case series are compounded by numerous anatomical, pathological, and clinical variables that are inconsistently reported.[1] Due to the varying degrees and possibilities of etiology, a classification system [Table 1] has been proposed by Saad et al.,[1] which helps characterize, categorize, and triage the management of ectopic varices. Based on this classification, a management protocol has been proposed that is directed toward addressing the cause of the DVs primarily with or without secondary management with obliteration/sclerosis. Occlusive types require recanalization of the portal thrombosis (as was the case in patient 5) or portosystemic decompression proximal to the occlusion. Non-occlusive types [Table 1: Type A, patients 1–4], as in this study, can be treated with portal decompression (with or without obliteration) utilizing a TIPS, particularly DVs that exhibit portosystemic shunting [Table 1: Types 2A and 3A].[1] Table 4 demonstrates the distribution of the seven DV cases (including the two excluded patients) encountered by the two institutions participating in this study, according to the hemodynamic classification of ectopic varices by Saad.[110]

TIPS is the most commonly described surgical or endovascular decompressive procedure in recent literature.[1] Traditionally, surgically placed shunts were described in the management of DVs.[134571011] However, TIPS has a high rebleed rate of up to 21–37% DVs in cases of ectopic varices.[134] This is likely due to the fact that 20-25% of ectopic mesenteric variceal bleeds occur in the presence of a PSG of less than 12 mmHg.[11] Moreover, more than half (57%) of post-TIPS rebleeding episodes occur with a PSG less than 12 mmHg.[11] This is highly consistent with the findings of this study where three patients had undergone prior TIPS procedures (mean time lapse between TIPS and subsequent transvenous obliteration was 125 days, range: 3–324 days) which had failed (continued bleeding) requiring subsequent transvenous obliteration. Moreover, in the presence of a patent portal vein (patients 1–4), there was progression of DVs growth/development and/or DVs bleeding occurred at a mean PSG of 3.8 mmHg (range: 0–9 mmHg) [Table 3].
Transvenous obliteration or sclerosis can be with or without balloon-assisted occlusion.[1] The primary sclerosant described has been ethanolamine oleate with the adjunctive sclerosants being 50% glucose and absolute ethanol.[123] The approach can be BRTO,[12379] BATO,[18] or both.[19] Sclerosis from the systemic venous side (BRTO) is usually successful in less than 50% of cases. However, more than half of the cases require portal venous side access (BATO) either as an adjunct or as an alternative to BRTO.[37] There are two cases of failed BRTO (one rescued by TIPS) and three additional cases of BRTO procedures requiring adjunctive BATO access.[19] In 16 cases of transvenous obliteration of DVs in three studies evaluated by Saad et al., the 1-year Kaplan–Meier DVs rebleed rate was 13%.[1411]
The current study describes the novel approach and impressive results of combining TIPS decompression and trans-TIPS transvenous obliteration of DVs. There have been no post-procedural hemorrhagic episodes in a mean follow-up period of 22 months (6–50 months). However, as this experience shows [Table 3: Patient 1], if coil embolization is performed, there is a risk of coils (or other metallic embolic material that is used such as AVP) eroding into the lumen of the duodenum. DVs have thin walls and thin overlying mucosa; thus, when embolizing with metallic material, one should try to embolize the portosystemic and/or porto-portal collaterals that are not submucosal. However, if coil embolization is performed in the extra-luminal collaterals/varices, the actual submucosal varices are not “obliterated” or sclerosed and new collateral formation may occur with recurrence of bleeding.[1] As a result, we do not advocate only coil embolization in the setting of TIPS, but prefer transvenous obliteration. The only exception is coiling of adjacent collaterals (that are not submucosal) to help create blood flow stasis and maximize sclerosant dwell time to optimize variceal obliteration [Figure 1]. This is particularly true when small occlusion balloon cannot be advanced to locations that are ideal for variceal blood flow stasis as was the case in patients 2 and 3 [Table 3]. Moreover, soft titanium coils are preferred over stiffer first-generation stainless steel coils in an attempt to reduce the risk of duodenal mucosal erosions.[1]
Limitations
This study is a retrospective audit of two institutions for patients with DVs undergoing TIPS and/or transvenous obliteration. The sample size is small owing to the rarity of the condition. However, decisive conclusions cannot be made due to the small sample size. Moreover, a denominator of exactly how many bleeding DVs were encountered (regardless of management) by the two institutions in the study period is difficult to obtain and would probably be inaccurate because of the obscurity of the diagnosis which may be overlooked as a source of bleeding.
CONCLUSION
In conclusion, combining portal decompression utilizing TIPS and transvenous obliteration utilizing STS foam sclerosant appears to be safe and effective in controlling duodenal variceal bleeding. Metallic embolization material (coils and/or AVP) should be used sparingly and, when used, should be deployed in duodenal collaterals that are not submucosal.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/67/145903
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Ectopic varices: Anatomical classification, hemodynamic classification, and hemodynamic-based management. Tech Vasc Interv Radiol. 2013;16:158-75.
- [Google Scholar]
- should balloon-occluded retrograde transvenous obliteration be the first-line interventional radiologic treatment for bleeding duodenal varices. A case report and review of the literature? Acta Radiol. 2008;49:32-6.
- [Google Scholar]
- The efficacy of balloon-occluded retrograde transvenous obliteration on small intestinal variceal bleeding. Surgery. 2010;148:145-50.
- [Google Scholar]
- Bleeding ectopic varices in cirrhosis: The role of transjugular intrahepatic portosystemic stent shunts. Aliment Pharmacol Ther. 2008;28:294-303.
- [Google Scholar]
- Ectopic varices rupture in the gastroduodenal anastomosis successfully treated with N-butyl-2-cyanoacrylate injection. Acta Med Okayama. 2007;61:361-5.
- [Google Scholar]
- Roentgenographic demonstration of unusual extra-esophageal varices. Am J Roentgenol Radium Ther Nucl Med. 1968;103:281-90.
- [Google Scholar]
- Successful management of duodenal varices by balloon-occluded retrograde transvenous obliteration. J Nippon Med Sch. 2008;75:36-40.
- [Google Scholar]
- Combined balloon-occluded embolization for treatment of concurrent duodenal, gastric, and esophageal varices: A case report. Gastrointest Endosc. 2001;53:665-8.
- [Google Scholar]
- Successful therapy of bleeding duodenal varices by TIPS after failure of sclerotherapy. Am J Gastroenterol. 1998;93:272-4.
- [Google Scholar]
- Combined transmesenteric and transhepatic recanalization of chronic portal and mesenteric vein occlusion to treat bleeding duodenal varices. J Vasc Interv Radiol. 2014;25:1295-9.
- [Google Scholar]
- Bleeding ectopic varices--treatment with transjugular intrahepatic porto-systemic shunt (TIPS) and embolisation. J Hepatol. 2004;41:560-6.
- [Google Scholar]






