Translate this page into:
Malignant Pleural and Peritoneal Mesothelioma Consequential to Brief Indirect Asbestos Exposure
Address for correspondence: Dr. Supreethi Kohli, Tower 5, Flat No. 1403, Sushant Estate, Sector 52, Gurgaon - 122 002, Haryana, India. E-mail: supreethikohli@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
This report highlights that pleural and peritoneal mesothelioma can occur without direct asbestos exposure as was seen in our young patient. The patient had indirect exposure for as short as 3 months as a child, 15 years earlier, when she was residing with her miner father in the district of Jharia, Jharkhand, which is an asbestos-rich mining area in eastern India. The patient presented with chest pain and breathlessness. Chest X-ray showed opaque right hemithorax. Typical contrast- computed tomography (CECT) enhanced radiological features included nodular, soft-tissue attenuation and homogenously enhancing rind-like mass causing scalloping of the underlying lung and liver. Similar lesions were also found involving the pelvis. Diagnosis of malignant mesothelioma was confirmed on lung biopsy. Under-reporting of exposure is usual because it is unrecognized by both patients and investigators.
Keywords
Asbestos
contrast-enhanced computed tomography
mesothelioma
pleural
INTRODUCTION

Mesothelioma is a malignancy of the mesothelium, which is a serosal membrane covering that protects internal organs of the body. Malignant mesotheliomas mainly affect the pleural mesothelium (50-60%), but can also affect the peritoneum (20-30%), pericardium, or even the tunica vaginalis testis of a male patient or tunica serosa uteri of a female patient. Malignant peritoneal mesothelioma is a rare cancer with poor patient survival. The incidence of peritoneal mesothelioma worldwide is approximately one case in a population of 4-5 million,[1] with an incidence of all mesotheliomas being 1 in 1 million.[1] However, the incidence rates in industrialized countries are greater and range between 0.5 and 3 cases per million in men and between 0.2 and 2 cases per million in women.[1]
In approximately 50% of peritoneal mesothelioma cases, there is a history of asbestos exposure. It appears that the risk of mesothelioma and the risk of subsequent mortality correlate with cumulative exposure to asbestos fibers and also the type of fiber. Asbestos has been implicated as the main carcinogen.[12]
Typical radiological features coupled with a high level of suspicion mainly of a history of asbestos exposure can indicate this rare but fatal disease. Final diagnosis has to be confirmed by pathognomonic pathological features.
CASE REPORT
A 26-year-old female patient, resident of Bihar, India presented with a 4-month history of insidious, gradually progressive, dull aching chest pain. Patient also complained of progressive breathlessness during this 4-month period. No history of constitutional features or fever or cough was present. Patient was a nonsmoker and housewife without a history of exposure to any dust/fumes. However, patient revealed during her childhood she had resided in Jharia, a mining area in Bihar, where her father worked in the mines. On general physical examination, her vitals were stable. Generalized puffiness over face was noted, with no associated peri-orbital swelling. Swelling and dilatation of veins over chest wall and abdomen were seen, suggesting superior vena (SVC) cava obstruction. Increased volume of right hemi thorax with decreased movements, dullness on percussion, and breath sounds of decreased intensity were observed over the right side.
Chest X-ray poster anterior (PA) view revealed opaque right hemi thorax with areas of radiolucency in the right mid zone. Right costophrenic angle and right hemi diaphragm silhouette was obliterated by soft-tissue opacity. No evidence of any calcification or bone destruction was noted. Mild tracheal and mediastinal shift to the right side was observed. The left side was clear [Figure 1]. Contrast-enhanced computerized tomography (CT) scanning revealed pleural-based soft-tissue attenuation mass lesion completely encasing the right lung (rind-like appearance) with resultant consolidation collapse of the right lung. SVC obstruction was noted. Contiguous rind-like soft-tissue attenuation mass lesion was seen to cross the diaphragm causing scalloping of the liver surface. Right axillary lymphadenopathy was seen. No pleural effusion or significant mediastinal lymphadenopathy was seen. No pleural calcification was noted [Figures 2 and 3]. Chest wall showed loss of extra pleural fat planes at several locations; however, no definite invasion of intercostal muscles was seen [Figure 4]. Multiple liver parenchymal hypo densities were also seen, suggestive of metastasis.
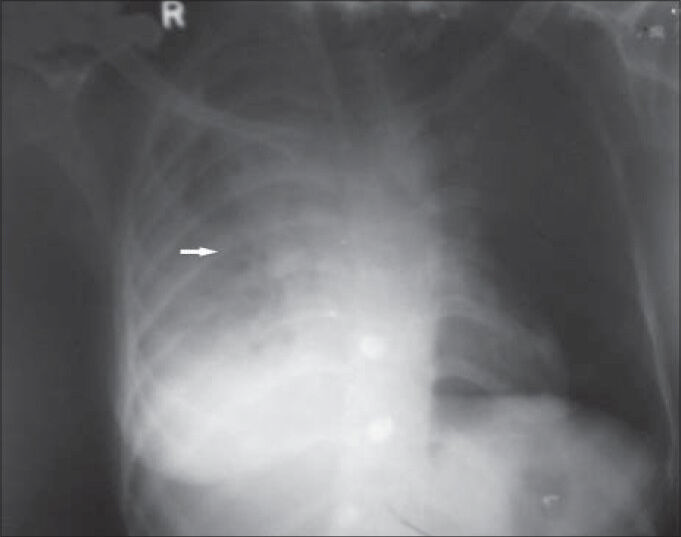
- 26-year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. Chest X-ray poster anterior view reveals opaque right hemithorax with areas of radiolucencies in the right mid zone (horizontal arrow). Right costophrenic angle and right hemi diaphragm silhouette is obliterated by soft-tissue opacity. There is no evidence of any calcification or bone destruction. Right side shows mild tracheal and mediastinal shift. Left lung field and costophrenic angle on the left side are clear.
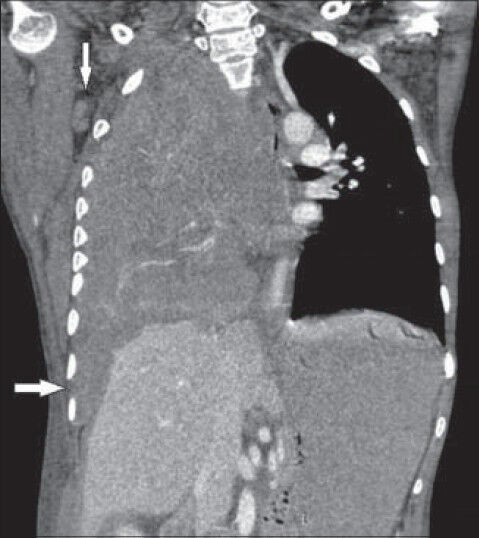
- 26-year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. Post-contrast CT coronal view shows contiguous rind-like soft-tissue attenuation pleural mass lesion encasing right hemithorax and scalloping of liver (horizontal arrow). Right side shows axillary lymphadenopathy (vertical arrow).
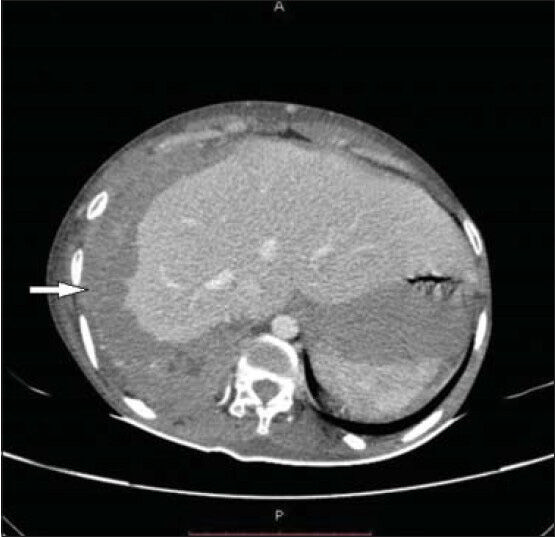
- 26 year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. Post-contrast CT scan axial view shows pleural-based mass lesion (horizontal arrow) causing scalloping of the liver parenchyma with loss of extra pleural fat planes along the right lateral chest wall. No rib destruction is noted.
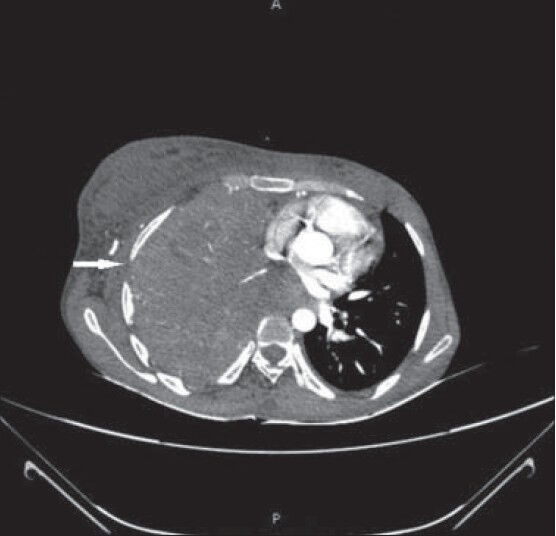
- 26-year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. Post-contrast CT scan axial view shows soft-tissue attenuation pleural mass lesion completely encasing the right lung which shows consolidation collapse. Right lateral chest wall shows loss of extra pleural fat plane (horizontal arrow). However, no definite invasion of intercostal muscles is seen.
CT sections from the pelvis showed non-contiguous, homogenously enhancing soft-tissue attenuation mass lesion in the pelvis, abutting the small and large bowel loops without causing any obstruction. Ascites was also noted [Figure 5].
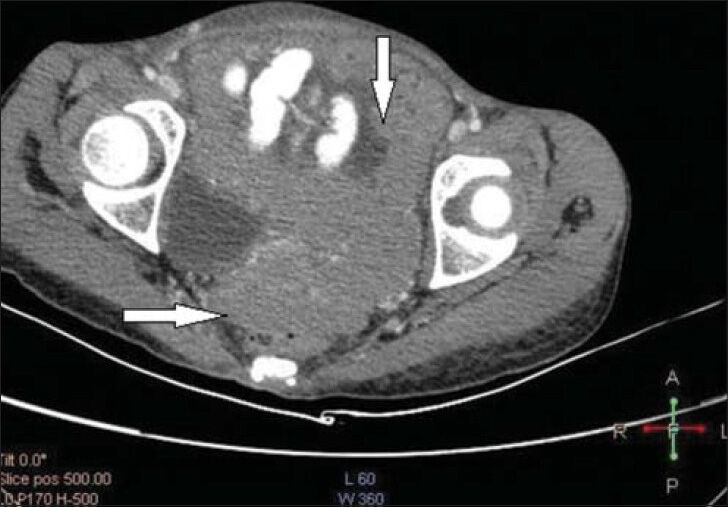
- 26-year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. Post-contrast axial CT sections from the pelvis show homogenously enhancing soft-tissue attenuation mass lesion (horizontal and vertical arrows) in the pelvis, abutting the small and large bowel loops without causing any obstruction. Ascites is also noted.
Hematoxylin and eosin staining of the lung biopsy sample showed malignant cells in acinar pattern and infiltrating in a linear fashion [Figure 6], which suggested the possibility of mesothelioma or adenocarcinoma. Immunohistochemistry was done which showed the tissue was positive for calretinin and Wilms’ tumor protein (WT1) and negative for carcinoembryonic antigen (CEA) cluster of differentiation molecule (CD15), anti-endomysial antibody (EMA), and thyroid transcription factor 1 (TTF1).
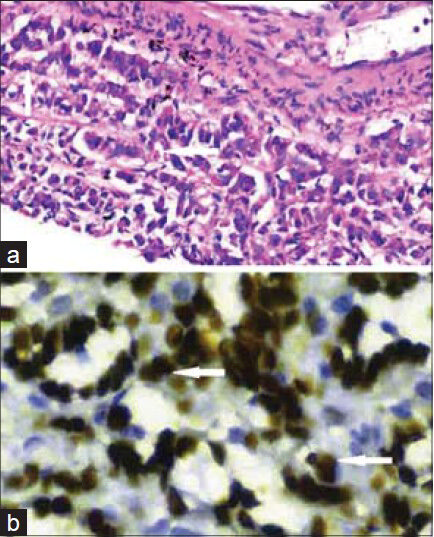
- 26-year-old female with chest pain and breathlessness diagnosed with pleural and peritoneal mesothelioma. (a) Histopathology slide of the biopsy specimen. Hematoxylin and eosin staining of the lung biopsy sample (×400) shows malignant epithelial cells in acinar pattern (vertical arrow) infiltrating the lung tissue in a linear fashion (horizontal arrow). (b) Biopsy sample stained for Wilms’ tumor protein shows nuclei positive (horizontal arrows) for the protein (×1000), which confirms the diagnosis of mesothelioma.
Based on lung biopsy sample and immunohistochemistry, diagnosis of malignant mesothelioma was made. The patient was referred to a medical oncologist. She was prescribed Cisplatin and Pemetrexed. The patient did not comply with the treatment or come for follow-up.
DISCUSSION
Peritoneal or pleural mesothelioma is a rare but fatal disease with increasing number of cases being reported possibly secondary to the incidence of asbestos exposure.
Although asbestos is considered the primary etiologic factor for this tumor, some published series also report a small number of subjects without known exposure to asbestos.[3] Under-reporting of exposure may partially account for this, since exposure in a variety of settings is sometimes unrecognized by both patients and investigators. In our case, the patient had indirect exposure for as less as 3 months, about 15 years earlier when the patient was residing with her miner father in the district of Jharia, Jharkhand, which is an asbestos-rich mining area in eastern India.
Occupational asbestos exposure in women is relatively uncommon. Domestic exposure via contaminated clothes of an asbestos-exposed member of the family has also been documented to be a high risk factor.[3] Having a father or spouse employed in an asbestos-related occupation was found to increase the risk of mesothelioma by a factor of 10.[3]
Mesothelioma may arise in “genetically predisposed” host with autosomal transmitted gene in concert with asbestos exposure.[45] Hammer and associates reported familial mesothelioma in two families.[4] Martensson and colleagues described malignant mesothelioma in two pairs of siblings, i.e. a brother and sister and identical twin brothers.[5]
Possible protective effect of vegetables or some vegetable constituents on mesothelioma development has been studied. A hospital-based case-control study was conducted to determine the association of malignant mesothelioma and consumption of carotenoid containing fruits and vegetables. The study suggested that provitamin A or beta-carotene may decrease the risk of mesothelioma.[6]
Mesothelioma is rare in pediatric population or in young adults. Grundy and Miller[7] reviewed 42,597 death certificates of children under 15 years of age. Of a total of 31 cases of mesothelioma identified, only 13 could be confirmed. Brenner et al.,[8] reported on seven cases of this tumor seen at Memorial Sloan-Kettering Cancer Center in New York City. Six of these patients had pleural involvement.
CT is the mainstay of diagnosis of this lesion. Common manifestations include pleural effusion, circumferential and nodular pleural thickening, interlobar fissural thickening, and pleural calcification. The tumor may encase the hemithorax, giving a rind-like appearance. There is also frequent contraction of the affected hemithorax with associated ipsilateral mediastinal shift, narrowed intercostal spaces, and elevation of the ipsilateral hemidiaphragm.
Chest wall involvement is seen as obliteration of extra pleural fat planes, invasion of intercostal muscles, displacement of ribs, or bone destruction. Tumoral extension into vascular structures and mediastinal organs including the heart, esophagus, and trachea may occur. There is obliteration of surrounding fat planes and presence of a soft-tissue mass that surrounds more than 50% of the circumference of a vascular structure. Malignant pleural mesothelioma (MPM) may invade the pericardium as nodular pericardial thickening or pericardial effusion. Transdiaphragmatic extension of MPM is suggested by a soft-tissue mass that encases the hemidiaphragm.
Two peritoneal mesothelioma subtypes have been described dependent on CT findings. The “dry” type of mesothelioma is associated with multiple small masses or a single dominant localized mass on CT with little or no ascites. In the “wet” type of mesothelioma, CT reveals widespread small nodules with ascites present, but no dominant mass.
Pulmonary metastasis is seen as nodules/mass or military nodules. Chest CT may also rarely demonstrate extra-thoracic spread of MPM (direct hepatic invasion, retroperitoneal extension, retrocrural adenopathy).
The role of magnetic resonance imaging (MRI) is better evaluation of chest wall and assessing diaphragmatic involvement. Positron emission tomography/computed tomography (PET/CT) scan is helpful in evaluating extra thoracic and nodal metastasis. 18F-fluorodeoxyglucose (18F-FDG) PET/CT has greater sensitivity than CECT in identifying local extent, lymph nodes, and metastasis. 18F-FDG PET/CT whole-body explorations are useful to monitor the follow-up and evaluate the metabolic response to chemo- and radiotherapy.[9]
Due to its insidious onset and slowly progressive features, the patient usually presents in the terminal stages where only palliative therapy can be offered. However, a number of therapeutic regimens have been used to improve prognosis and currently debulking surgery is followed by hyper-thermic intraperitoneal chemotherapy (HIPEC). The current median survival is around 10 months and relative 5-year survival is approximately 16%.[10]
CONCLUSION
Malignant mesothelioma involving the pleura or peritoneum is a rare but fatal disease. Widespread use of asbestos-containing products in the developing world ensures that this highly fatal disease will continue to represent a serious public health problem worldwide.
Despite the fact that most cases of malignant mesothelioma are secondary to the inhalation of asbestos fibers, a few patients present with no known source of asbestos exposure. Some exposures fail to be recognized and are therefore not reported. Like in our case, even small amount of exposure and indirect exposure through other asbestos-exposed family members can be associated with development of malignant mesothelioma. Our case was unusual due to the young age of the patient and limited indirect asbestos exposure. A short period of indirect exposure during childhood is often ignored by the patient and in several cases not known to the patient, leading to under-reporting of cases.
Hence, typical radiological and pathological features may help confirm the diagnosis of mesothelioma if the index of suspicion is high for the disease.
ACKNOWLEDGMENTS
The authors would like to acknowledge the contribution of Dr. Anand Kumar Verma, Associate Professor, Department of Pathology and Dr. Deepti Gothi, Associate Professor, Department of Pulmonary Medicine, Post Graduate Institute of Medical Science and Research, and Employee, State Insurance Hospital, Basaidarapur, New Delhi, India in preparing this manuscript.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/35/135662
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Update on the molecular biology of malignant mesothelioma. Cancer. 2007;109:1454-61.
- [Google Scholar]
- Malignant mesothelioma in two pairs of siblings; is there a hereditary predisposing factor. Eur J Resp Dis. 1984;65:179-84.
- [Google Scholar]
- Dietary intake and the risk of malignant mesothelioma. Br J Cancer. 1996;73:1122-5.
- [Google Scholar]
- Malignant mesothelioma in childhood. Report of 13 cases. Cancer a hereditary predisposing factor? Eur J Respir Dis. 1984;65:179-84.
- [Google Scholar]
- Malignant mesothelioma in children: Report of seven cases and review of the literature. Med Pediatr Oncol. 1981;9:367-73.
- [Google Scholar]
- 18F-FDG PET/CT in suspected recurrences of epithelial malignant pleural mesothelioma in asbestos-fibers-exposed patients (comparison to standard diagnostic follow-up) Clin Imaging. 2013;37:1098-103.
- [Google Scholar]
- Analysis of factors associated with outcome in patients with malignant peritoneal mesothelioma undergoing surgical debulking and intraperitoneal chemotherapy. J Clin Oncol. 2003;21:4560-7.
- [Google Scholar]






