Translate this page into:
Giant Cerebellar Ganglioglioma Mimicking a Pilocytic Astrocytoma

*Correspondence author: Nguyen Minh Duc, Department of Radiology, Pham Ngoc Thach University of Medicine, Ho Chi Minh City, Vietnam. bsnguyenminhduc@pnt.edu.vn
-
Received: ,
Accepted: ,
How to cite this article: Tuan TA, Duc NM. Giant cerebellar ganglioglioma mimicking a pilocytic astrocytoma. J Clin Imaging Sci 2021;11:3.
Abstract
A benign brain tumor, called a ganglioglioma (GG), can sometimes be found in the cerebrum. We describe an unusual pediatric case of GG in the cerebellum in this article. An 11-year-old male patient had a headache and epilepsy disorder. The head magnetic resonance imaging results revealed a giant cyst with an enhancing mural nodule in the right cerebellar hemisphere that flattened the fourth ventricle. Pilocytic astrocytoma was the provisional diagnosis based on clinical and imaging details. After radical mass eradication, a GG was demonstrated through histopathological analysis. Even though GG is an uncommon tumor, it should be included in the differential diagnosis for a cerebellar mass with both cystic and solid components in children.
Keywords
Ganglioglioma
Pilocytic astrocytoma
Cerebellum
Posterior fossa tumor
Children
INTRODUCTION
Ganglioglioma (GG), a benign and rare tumor of the central nervous system (CNS), is typically found supratentorially in the temporal lobe.[1-3] Infratentorial GG, especially cerebellar GG, is quite unusual. Histopathologically, GG consists of both glial and ganglion cells.[1-3] Although GG can occur during all stages of life, it is primarily observed in children.[4] Seizure and epilepsy are the most frequent symptoms presented by patients with GG.[1,3,4] We describe an unusual case of cerebellar GG in this paper, which presented radiologically as a pilocytic astrocytoma.
CASE PRESENTATION
An 11-year-old male, suffering from headache and seizure, was admitted to the Department of Neurosurgery, Children’s Hospital 2. The patient’s medical profile was typical, and no neurological defects were observed during clinical assessment. Blood test measurements were within acceptable ranges. The patient was referred for head magnetic resonance imaging (MRI) with a gadolinium-based contrast agent. On T2-weighted [Figure 1] and fluid-attenuated inversion recovery [Figure 2] images, a giant heterogeneous mass, composed of both cystic and solid components, was revealed in the right cerebellar hemisphere (77 × 49 × 53 mm). No signs of vasogenic edema, calcification, or bleeding associated with the lesion were detected. The mean apparent diffusion coefficient (ADC) values obtained from diffusion-weighted imaging (DWI) for the cystic and solid components of the tumor and for the parenchyma were 2.52, 0.77, and 0.63 × 10-3 mm2/s, respectively [Figure 3].
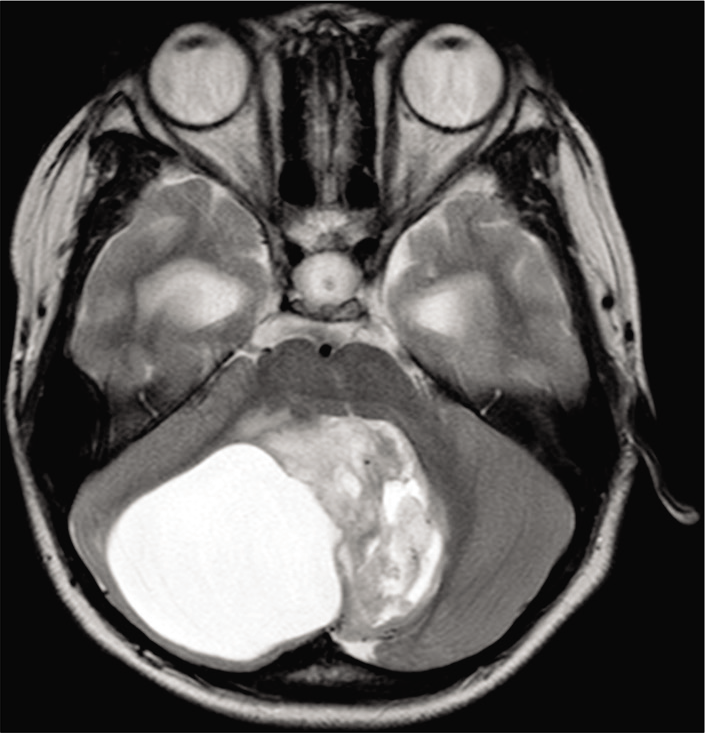
- A heterogeneous mass, consisting of both solid and cystic components, located in the right cerebellar hemisphere, on axial T2-weighted imaging.
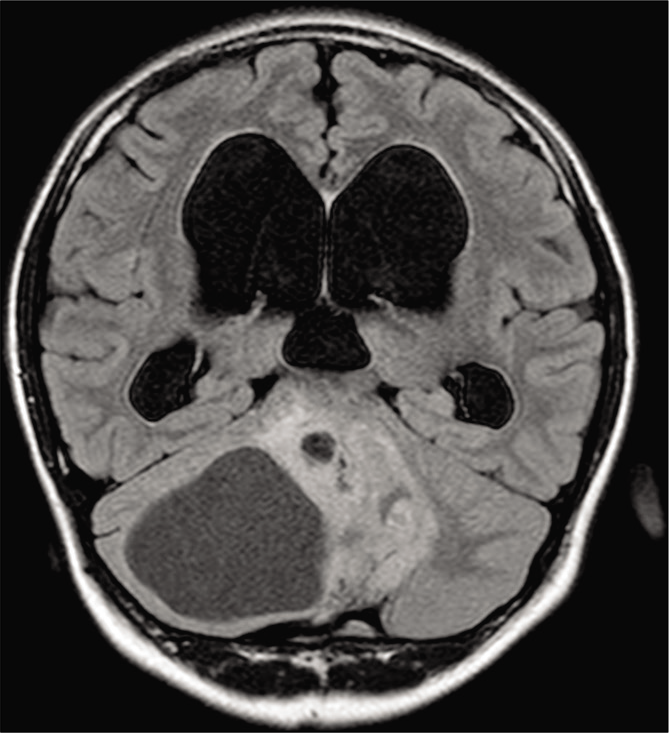
- The solid portion of the tumor appears hyperintense, whereas the cystic portion of the tumor appears hypointense on coronal FLAIR (fluid-attenuated inversion recovery) imaging.
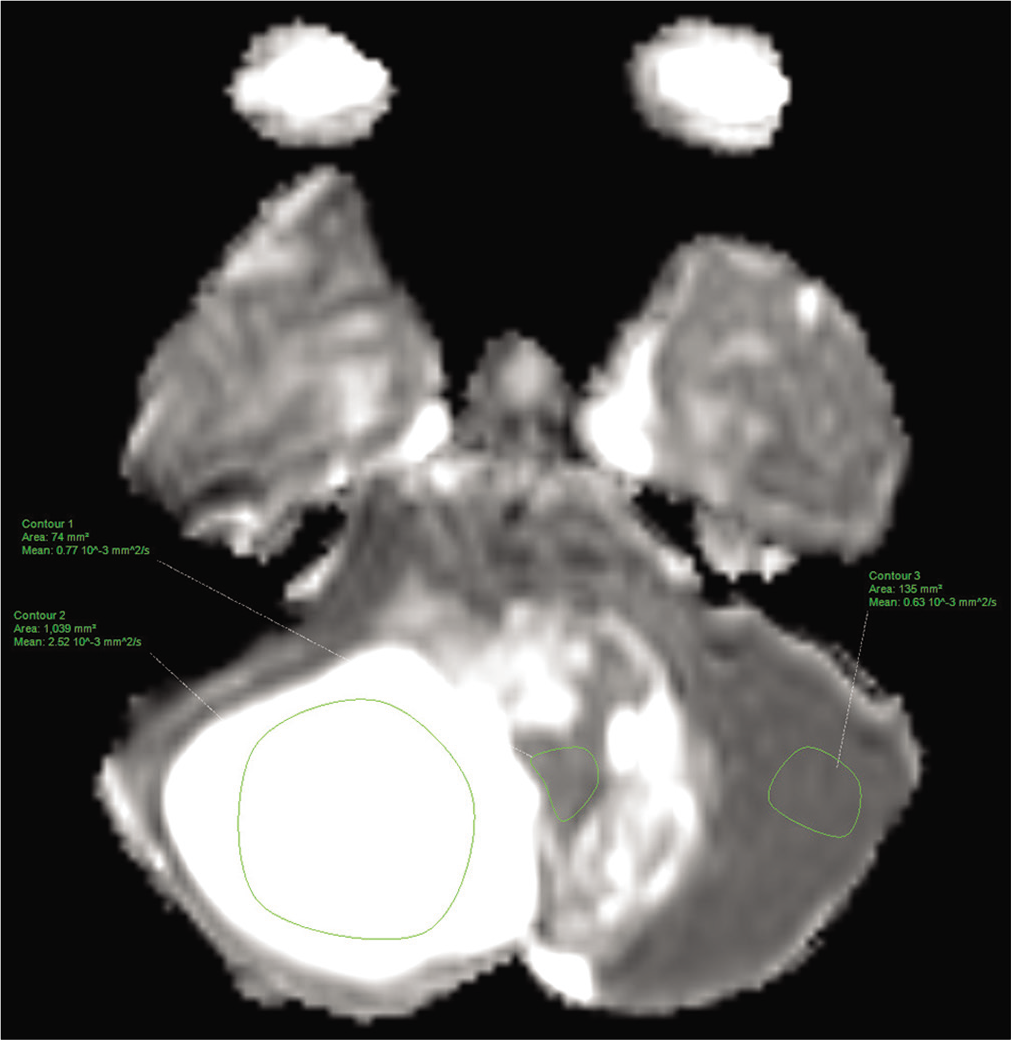
- Axial apparent diffusion coefficient of the cystic and solid components of the tumor and of the parenchyma.
The cerebral blood flow values recorded for the solid portion of the tumor and the parenchyma, determined by three-dimensional arterial spin labeling, were 72.33 and 22.98, respectively [Figure 4]. The fractional anisotropy values, which were derived from diffusion tensor imaging (DTI), for the solid segment of the tumor and the parenchyma, were 0.440 and 0.543, respectively [Figure 5]. The Choline/N-acetylaspartate ratio for the solid portion of the tumor, as measured by spectroscopy, was 1.96 [Figure 6]. The relative enhancement (%), peak enhancement, peak relative enhancement (%), time to peak (s), wash-in rate (s-1), wash-out rate (s-1), and area under the curve values determine for the solid part of the tumor compared with that of the parenchyma, as calculated from the axial T1-perfusion map [Figure 7], were 103.68 versus 1.21, 1304.82 versus 25.39, 107.26 versus 1.54, 125.28 versus 93.96, 61.29 versus 5.22, 5.94 versus 5.71, and 188,480 versus 167.23, respectively. The tentative diagnosis based on these clinical and radiological details was pilocytic astrocytoma. After radical neurosurgery, the histopathological examination of the tumor tissues was entirely compatible with traditional GG [Figure 8]. The patient was uneventfully discharged 9 days post-operation.
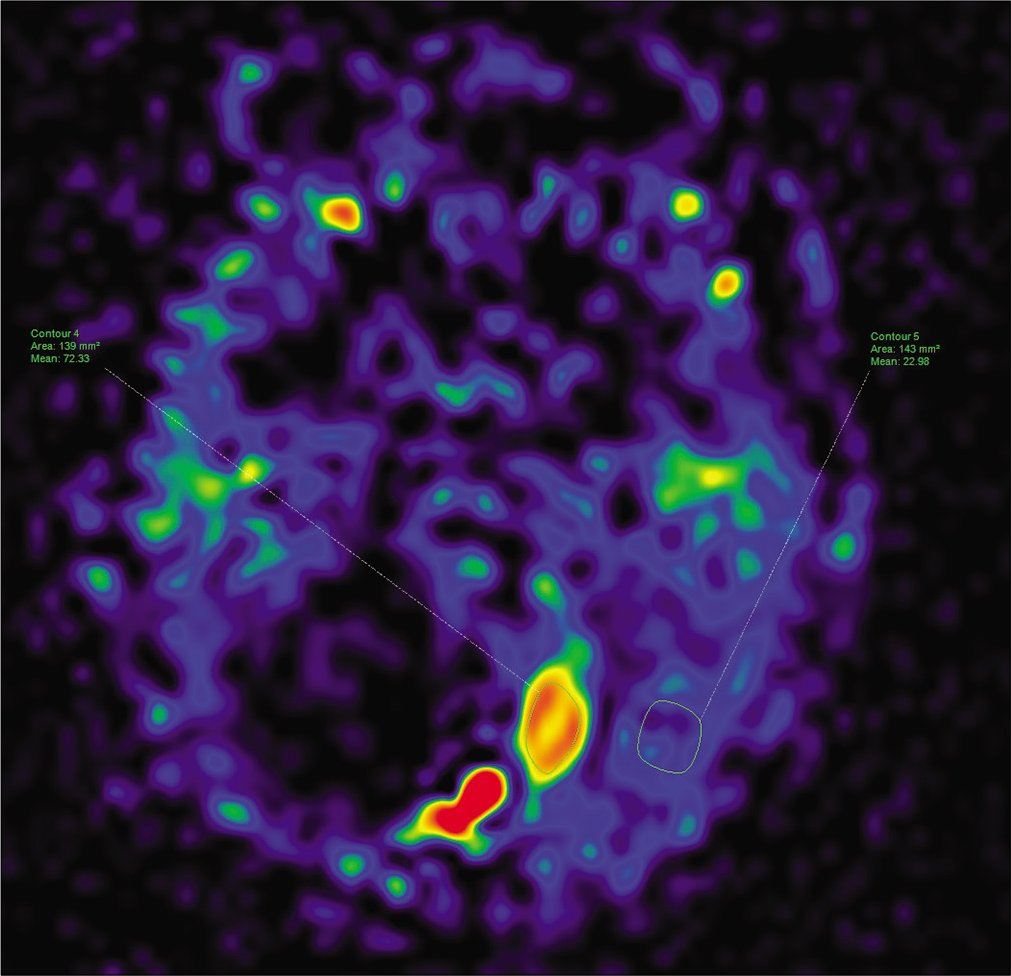
- The cerebral blood flow values of the solid tumoral component and the parenchyma on 3-dimensional arterial spin labeling.
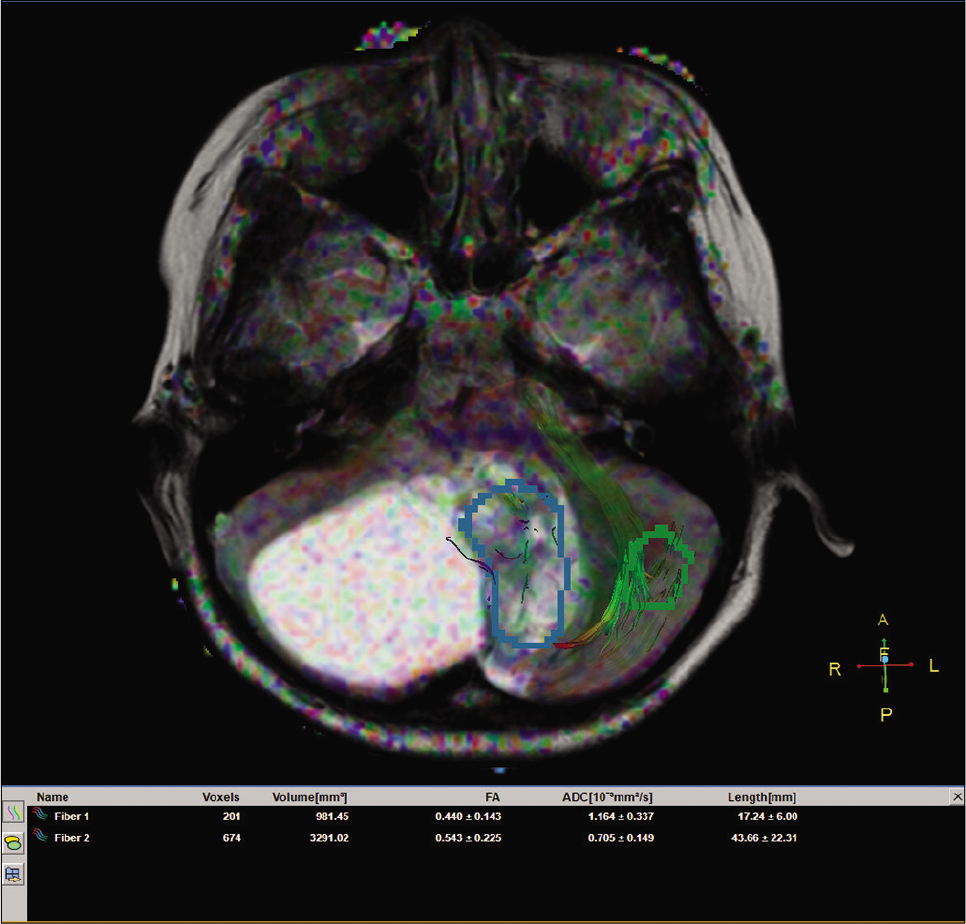
- The fractional anisotropy values for the solid portion of the tumor and the parenchyma on diffusion tensor imaging.
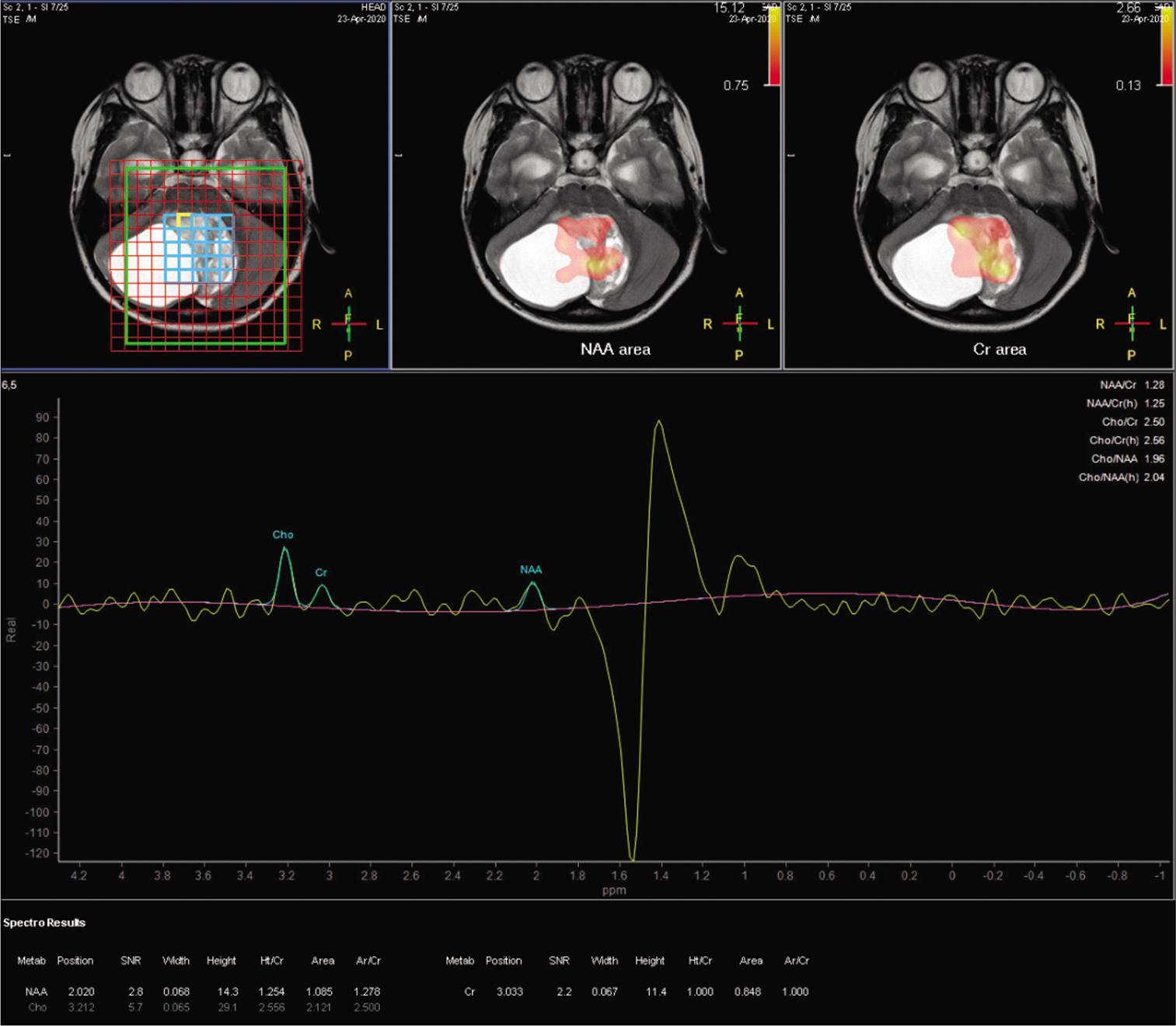
- The metabolic parameters determined for the solid tumor component on MRI spectroscopy.
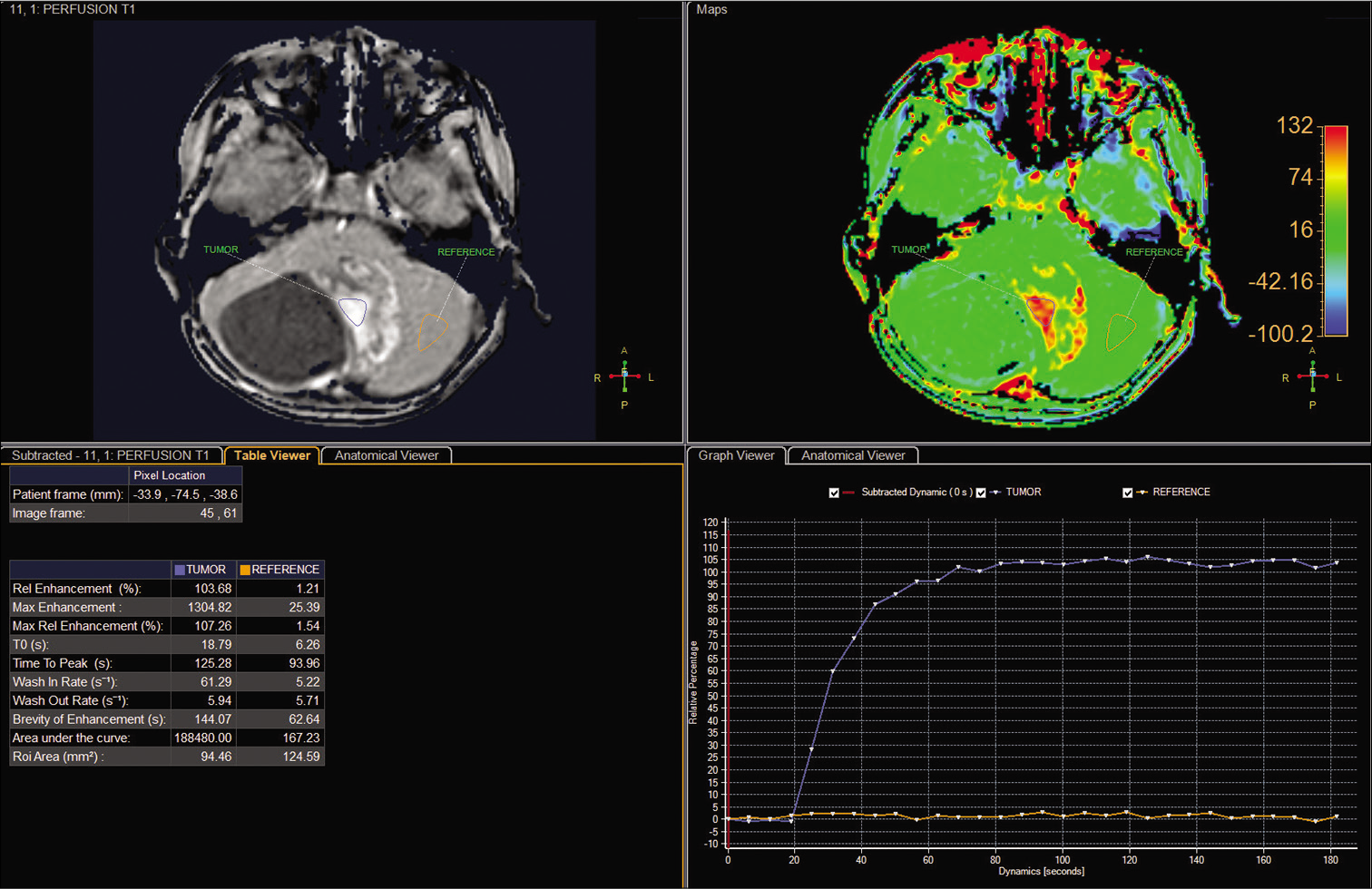
- The perfusion parameters for the solid portion of the tumor and the parenchyma on the semiquantitative T1-perfusion map.
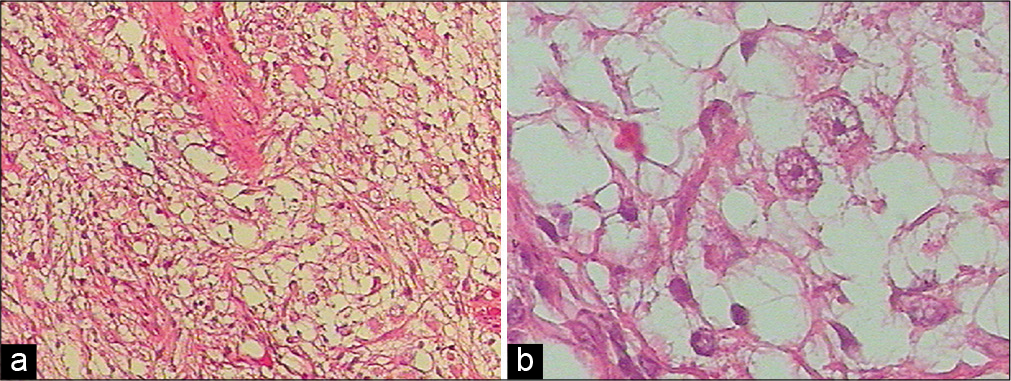
- Histopathological findings for excised sample tissue revealed ganglion cells with prominent nucleoli and the diffuse proliferation of astrocytes, without necrosis (hematoxylin and eosin, × 100 (a), × 400(b)).
DISCUSSION
Epidemiologically, Loretz first used the term “ganglioglioma” in 1870,[5] and Perkins further popularized the term in 1926.[6] GG is a rare, intra-axial tumor of the CNS that accounts for between 0.4% and 1.3% of all intracranial neoplasms.[7,8] GG presents with a peak age of incidence between 10 and 20 years and without gender preponderance. GG is typically located in the supratentorial region, with the largest portion arising in the temporal lobe.[7-9] Infratentorial GG is very unusual and is most commonly derived from the brainstem.[10] Thus, the occurrence of GG in the cerebellum is exceedingly rare. To the best of our knowledge, the present case may represent the first case report of a cerebellar GG that was diagnosed with the assistance of multiple advanced imaging sequences, including DWI, three-dimensional arterial spin labeling, DTI, spectroscopy, and T1-perfusion.
Clinically, supratentorial GG causes epileptic seizures, whereas infratentorial GG results in focal neurological deficits due to a local mass effect and symptoms related to increased intracranial pressure, such as headache, nausea, and vomiting.[1,3,4,8-10] However, no clear clinical symptoms exist that can suggest cerebellar GG and differentiate GG from other, more frequent cerebellar neoplasms that occur in children.[1,3,4]
Radiologically, the morphological imaging of GG typically reveals a mural nodule-enhancing cyst, without adjacent edematous parenchyma. Calcifications inside the mass have been reported in 6%–30% of cases.[7-13] On MRI, GG often presents with low signal intensity on T1-weighted images and high signal intensity on T2-weighted images.[7-13] Unfortunately, these imaging characteristics do not contribute to the differentiation of GG from other posterior fossa brain tumors, especially pilocytic astrocytoma, because the identification of a cyst with an enhancing mural nodule is indicative of but not unique to GG.[7-13] In the present case, no tumoral ossification or parenchymal edema was identified. Despite the utilization of innovative imaging sequences, a misdiagnosis of pilocytic astrocytoma was established preoperatively.
A mixed histological composition of mature ganglion cells and glial cells, at various proportions and degrees of differentiation, was specifically defined in 1930 by Courville as GG.[14] The atypical appearance of ganglion cells in the sample tissue is necessary to support a GG diagnosis.[1,3,7,14,15] Typical GG is considered a grade I tumor, according to the World Health Organization classification guidelines.[15] Although GG is an indolent neoplasm, it possesses the capacity for malignant transformation.[16] A few cases of anaplastic GG have been described that were classified as Grade III.[17] Malignant degeneration of GG is extraordinarily uncommon and is often associated with the glial components of the mass, with an approximate incidence of 6%.[18] GG is unlikely to present subarachnoid and leptomeningeal spread.[19]
Therapeutically, gross-total resection is the first-line treatment option for GG. GG is always well-circumscribed, and radical surgery tends to result in an effective prognosis, with a 5-year survival rate greater than 90%.[1,20] However, the complete resection of cerebellar GG or GG located in the brainstem is challenging due to the surrounding eloquent structures.[10,21] In these cases, partial removal should be considered to diminish the risks of neurological damage. Since GG has the potential to become malignant and relapse is a risk, radiotherapy is suggested. However, whether adjuvant radiotherapy for GG can enhance GG outcomes remains a point of ongoing debate.[22,23]
CONCLUSION
Cerebellar GG is exceedingly uncommon. Clinically, the symptoms associated with GG are not specific and include seizure, epilepsy, and headache. Radiologically, the manifestation of GG and pilocytic astrocytoma may overlap. Radical neurosurgery, the optimal treatment of choice, results in the best prognosis but may not be applicable to all cases due to adjacent eloquent structures. The use of adjuvant radiotherapy for GG remains controversial.
Acknowledgment
We would like to express our gratitude to Dr. Mai Tan Lien Bang, Dr. Dang Do Thanh Can, Dr. Huynh Quang Huy, Prof. Pham Minh Thong, Mrs. Dang Thi Bich Ngoc, and Mr. Nguyen Chanh Thi for their assistance and technical support in completing this research.
Declaration of patient consent
Institutional Review Board permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Gangliogliomas: Pathological and clinical correlation. J Neurosurg. 1981;54:58-63.
- [CrossRef] [PubMed] [Google Scholar]
- Central nervous system gangliogliomas. Part 1: Pathology. J Neurosurg. 1993;79:859-66.
- [CrossRef] [PubMed] [Google Scholar]
- Central nervous system gangliogliomas. Part 2: Clinical outcome. J Neurosurg. 1993;79:867-73.
- [CrossRef] [PubMed] [Google Scholar]
- Gangliogliomas in children. A clinicopathological study. Childs Brain. 1978;4:339-46.
- [CrossRef] [Google Scholar]
- From the archives of the AFIP: Superficial gliomas: Radiologic-pathologic correlation, Armed forces institute of pathology. Radiographics. 2001;21:1533-56.
- [CrossRef] [PubMed] [Google Scholar]
- Posterior cranial fossa gangliogliomas. Skull Base. 2007;17:253-64.
- [CrossRef] [PubMed] [Google Scholar]
- Intracerebral ganglioglioma: Clinical and radiological study of eleven surgically treated cases with follow-up. Neurosurg Rev. 2001;24:114-8.
- [CrossRef] [PubMed] [Google Scholar]
- Ganglioglioma of brain stem and cervicomedullary junction: A 50 years review of literature. J Clin Neurosci. 2017;44:34-46.
- [CrossRef] [PubMed] [Google Scholar]
- Cerebellar ganglioglioma. J Korean Neurosurg Soc. 2008;43:165-8.
- [CrossRef] [PubMed] [Google Scholar]
- Atypical presentation of a pediatric cerebellar ganglioglioma. Pediatr Neurosurg. 2018;53:43-8.
- [CrossRef] [PubMed] [Google Scholar]
- Ganglioglioma: Tumor of the central nervous system; review of the literature and report of two cases. Arch Neurpsychiatry. 1930;24:439-91.
- [CrossRef] [Google Scholar]
- The 2016 World Health Organization classification of tumors of the central nervous system: A summary. Acta Neuropathol. 2016;131:803-20.
- [CrossRef] [PubMed] [Google Scholar]
- Tumor recurrence and malignant progression of gangliogliomas. Cancer. 2008;113:3355-63.
- [CrossRef] [PubMed] [Google Scholar]
- Anaplastic ganglioglioma in children. J Neurooncol. 2009;92:157-63.
- [CrossRef] [PubMed] [Google Scholar]
- Malignant transformation of a gangliocytoma/ganglioglioma into a glioblastoma multiforme: A molecular genetic analysis. Case report. J Neurosurg. 2001;95:138-42.
- [CrossRef] [PubMed] [Google Scholar]
- Ganglioglioma with leptomeningeal and subarachnoid spread: Results of CT, MR, and PET imaging. AJR Am J Roentgenol. 1992;159:391-3.
- [CrossRef] [PubMed] [Google Scholar]
- Intracranial ganglioglioma: Preoperative characteristics and oncologic outcome after surgery. J Neurooncol. 2002;59:173-83.
- [CrossRef] [PubMed] [Google Scholar]
- Pediatric infratentorial gangliogliomas: A retrospective series. J Neurosurg. 2007;107(Suppl 4):286-91.
- [CrossRef] [PubMed] [Google Scholar]
- Gangliogliomas: Experience with 34 patients and review of the literature. Am J Clin Oncol. 1998;21:557-64.
- [CrossRef] [PubMed] [Google Scholar]
- Radiation therapy and malignant degeneration of benign supratentorial gangliogliomas. Neurosurgery. 1998;42:1038-43.
- [CrossRef] [PubMed] [Google Scholar]







