Translate this page into:
Fast Magnetic Resonance Enterography Protocol for the Evaluation of Patients with Crohn’s Disease: A Pilot Study

*Corresponding author: Giuseppe Cicero, MD, Department of Biomedical Sciences and Morphological and Functional Imaging, University of Messina Via Consolare Valeria, 198100 Messina, Italy. gcicero@unime.it
-
Received: ,
Accepted: ,
How to cite this article: Cicero G, Mondello S, Wichmann JL, Albrecht MH, Vogl TJ, Cavallaro M, et al. Fast Magnetic Resonance Enterography protocol for the evaluation of patients with Crohn’s disease: A pilot study. J Clin Imaging Sci 2020;10:25.
Abstract
Objective:
Magnetic resonance enterography has achieved an increasingly importance in the evaluation of patients with Crohn’s disease, although it is limited by high costs and prolonged scanning times. The aim of our work was to design a “fast” abbreviated MRE protocol and to compare it with the standard one.
Materials and Methods:
A single-center retrospective study was performed on 73 patients with Crohn’s disease who underwent MRE with standard protocol over a 7-month period. The images of the standard protocol were separated from those included in the proposed abbreviated one and independently evaluated by two radiologists with different years of experience in MRE imaging. Statistical analysis was performed with the Cohen kappa (κ) value, used to assess the agreement in case of categorical variables, the Lin’s concordance correlation coefficient and Bland–Altman plot, in assessing the degree of agreement between numerical measurements, while the non- parametric Mann–Whitney U-test was used in comparing the evaluation times of the two protocols.
Results:
The intraobserver evaluation showed a perfect agreement between the two protocols for presence, number and extension of lesions, abdominal complications, and excellent/perfect in identification of active inflammation.The interobserver reproducibility was excellent for overall presence and number of lesions, for the presence and number of lesions in any abdominal quadrant, inflammation, intestinal and extraintestinal complications, and lesions extension.
Conclusion:
The proposed protocol achieves comparable performance with standard MRE. Furthermore, it would carry potential benefits in terms of patient’s comfort, time, and health-care costs savings.
Keywords
Inflammatory bowel diseases
Crohn’s disease
Magnetic resonance imaging
INTRODUCTION
Crohn’s disease (CD) is an invalidating chronic inflammatory bowel disorder that commonly affects young adults.[1]
It is characterized by a deregulated immune response against the commensal gut microbiota resulting into intestinal symptoms, such as abdominal pain and bloody diarrhea, and extraintestinal manifestations.[2]
The diagnosis is based on a combination of clinical characteristics, serological tests, and endoscopic and radiologic evaluation. In the latter context, various imaging techniques have been used for diagnosis and monitoring in patients with CD.
Earlier studies were focused on barium follow-through (BaF) performance, while during the past years, an increasingly interest has been shown for small intestine contrast- enhanced ultrasonography (SICUS), computed tomography enterography (CTE), and magnetic resonance enterography (MRE).[3-5]
However, while other imaging modalities involve radiation exposure or require technical skills and operator’s experience in interpretation, MRE is a comprehensive, radiation-free examination with a diagnostic accuracy equivalent to CTE.[6]
In particular, MRE has shown a significant correlation with the CD endoscopic index of severity and it is suitable for evaluating intestinal abnormalities and extraintestinal complications.[7-9] However, high costs and prolonged scan times limit its utility in routine scanning of patients with CD.
To date, the international consensus recommends to obtain a series of images based on T2-weighted scans (with and without fat saturation), diffusion-weighted imaging (DWI), and T1-weighted acquisitions performed before and after the intravenous administration of contrast medium. Although complementary to each other, these sequences may show overlapping and redundant findings, with a total scan time that can last up to 45 min.[10-12]
Moreover, intravenous injection of contrast medium is contraindicated in subjects with severe renal impairment and may expose the patient to the risk of an allergic reaction.
The aim of this single-center, retrospective study is to evaluate diagnostic performance and reliability of a fast abbreviated and contrast-free MRE protocol in comparison with the common standardized one.
MATERIALS AND METHODS
Population
This single-center retrospective study evaluated 94 patients with Crohn’s disease who underwent MRE at our institution between August 2016 and March 2017.
The inclusion criteria consisted of adult age (≥18 years old) and proven diagnosis of CD, based on endoscopy with deep mucosal biopsy or histologic analysis of the bowel resection specimen.
Inwards from the initial population, 21 patients were excluded from the retrospective evaluation. Exclusion criteria were related to avoided intravenous (i.v.) contrast medium injection due to allergic diathesis (4 patients); artifacts by patient’s movement during contrast-enhanced phases (3); low patient’s acceptance in ingesting polyethylene glycol (PEG) with insufficient filling of the bowel lumen (4); suspended examination due to claustrophobic event (3), defecation urge (3), allergic reaction after gadolinium administration (1), nausea and vomiting (2), and burning sensation in lumbar region (1).
The final study cohort consisted of 73 patients [Figure 1], whose characteristics are displayed in Table 1.
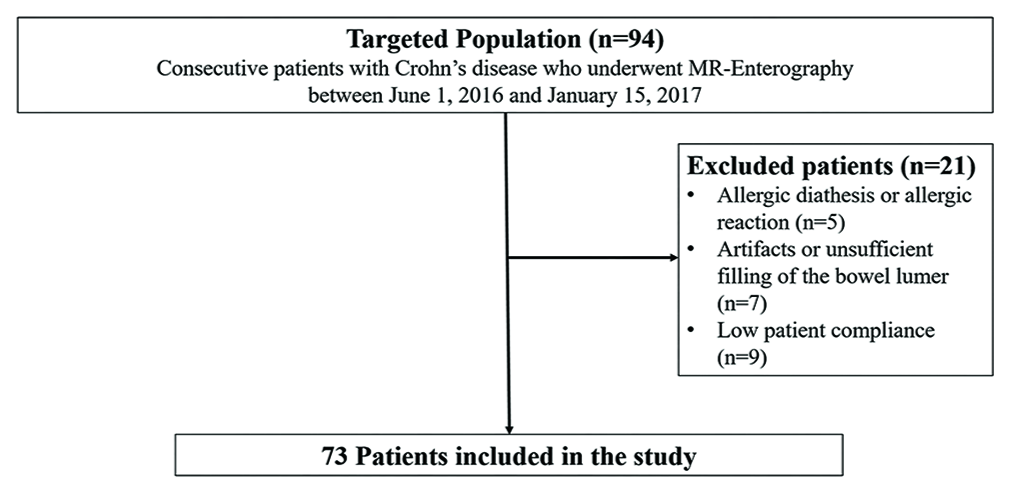
- Flowchart of the study population.
| Patients (n=73) | |
|---|---|
| Range | 18–65 |
| Gender, n(%) | |
| Female | 27 (37) |
| Male | 46 (63) |
| Examination indication, n(%) | |
| Recently diagnosed CE | 19 (26) |
| CD reevaluation | 54 (74) |
| First examination, n(%) | |
| Yes | 22 (30) |
| Follow-up | 51 (70) |
Study procedures
Our institutional review board approved the study and waived the requirement for informed consent.
All patients were asked to fast for at least 6 h before begin the examination. Bowel distension was obtained with the ingestion of a water solution of polyethylene glycol, a biphasic oral contrast agent. In particular, a total dose of 1500– 1800 mL was provided to the patients. The oral ingestion started approximately 45 min before the beginning of the scan, and split in three doses administered every 15 min.
A standardized MRE protocol was performed on a 1.5T MR scanner (Achieva, Philips, Best, The Netherlands), with gradients of 33 mT/m/s and a 16-channel phased array coil placed to cover the abdomen. After standard localizer image, axial and coronal single shot (SSH) T2-weighted turbo spin-echo sequences were acquired, followed by axial and coronal balanced steady-state free precession (bSSFP), axial and coronal spectral fat pre-saturation with inversion recovery (SPAIR), axial DWI at b-values of 0-400-800 mm2/s, axial pre-contrast ultrafast 3D T1-weighted gradient-echo fat-suppressed, and ultrafast axial 3D T1-weighted gradient- echo fat-suppressed images obtained after injection of gadoterate meglumine (Dotarem) at a dose of 0.2 ml/kg body weight, followed by a bolus of 30 mL of normal saline. Post-contrast images were acquired during arterial (45–55 s), venous (70 s), and delayed phases (7 min).
Image analysis
All MRE images were anonymized for patient data and all investigators were blinded to patient clinical information. Two radiologists, blinded to case review, were asked to duplicate a simulated “fast” MRE protocol by extrapolating from every MRE dataset three types of sequences: Coronal SSH T2 weighted, axial SPAIR T2 weighted, and axial DWI [Figure 2]. As result, each patient was anonymously presented twice in two different datasets, one including the standard protocol and one including the abbreviated “fast” MRE protocol.
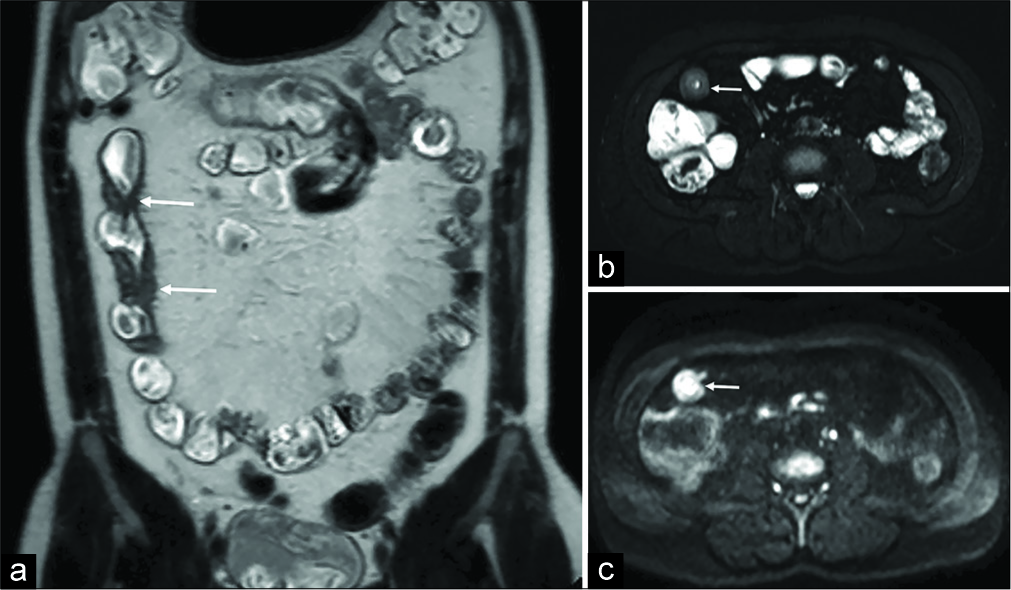
- Example of the proposed “fast” protocol in a 40-year- old male patient with confirmed Crohn’s disease. While coronal T2-weighted SSH images (a) yields in recognizing and measuring the bowel wall thickening (arrows), axial SPAIR T2-weighted (b) and DWI (c) sequences are suitable in identifying signs of active inflammation and/or intestinal complications.
Both datasets were randomized through a random number generator software and independently evaluated by two radiologists with 12 and 2 years of experience in MRE imaging. In particular, “fast” MRE dataset was first evaluated and, 58 days after a washout period, full MRE dataset was observed. The reviewers were blinded to the patients’ clinical data.
The standard full MRE protocol was reviewed by a panel of trained abdominal radiologists (3 in total, with more than 5 years of experience in body/abdominal CT/MRI, respectively) to reach consensus when discrepancies between the two readers were observed. In all the cases evaluated, the panel was in full agreement with the senior radiologist. All MRE findings were recorded in structured data forms. The data forms included the following items: Presence and number of small-bowel lesions; the affected abdominal quadrant (upper right quadrant, URQ; upper left quadrant, ULQ; lower right quadrant, LRQ; and lower left quadrant, LLQ); maximal mural bowel thickness, measured in millimeters (mm) through electronic calipers on axial or coronal SSH T2-weighted images; length of the affected loop, measured on the sequences and orientation that the observers considered to be the most accurate; presence of active inflammation; and presence of intestinal and extraintestinal complications.
The presence of active inflammation was determined according to the parameters currently described in the literature for each kind of sequence. In particular, wall thickness >3 mm measured on axial or coronal T2-weighted images (with and without fat saturation); submucosal edema and mural T2 hyperintensity; presence of ulcers; transmural or layered enhancement during arterial and portal venous phase; restricted diffusion of the thickened bowel walls (similar or superior than lymph nodes or spleen); engorgement of vasa recta (“comb sign”); mesenteric edema; abdominal reactive lymph nodes; and presence of phlegmons, abscesses, sinus tracts, or fistulas.
Statistical analysis
Exploratory analysis was carried out to determine the distribution of the data. Continuous variables are presented as mean±SD or median (interquartile range). Distributions of categorical variables were presented as frequencies and percentages. The Cohen’s kappa (κ) was calculated to assess the agreement in case of categorical variables. Cohen’s kappa coefficient indicated reproducibility of interpretations: κ > 0.81 is excellent, 0.80 > κ > 0.61 is good, 0.60 > κ > 0.41 is moderate, 0.40 < κ < 0.00 is average, and κ < 0.00 is poor.[1] The kappa coefficients were recorded with their confidence intervals (CI). A contingency table was constructed to calculate the sensitivity and specificity. Full MRE protocol findings were used as reference criteria. Lin’s concordance correlation coefficient (CCC)[2,3] and Bland–Altman plot[4] were used to assess the degree of agreement between numerical measurements. According to Lin’s CCC, κc > 0.99 was considered to represent an almost perfect agreement, κc = 0.99–0.95 a substantial agreement, and κc = 0.95–0.90 a fair agreement, whereas κc < 0.90 was considered to represent a poor agreement. Comparisons of evaluation time between the two protocols were performed with the nonparametric Mann–Whitney U-test. A two-tailed P < 0.05 was considered statistically significant. Statistical analysis was performed using R software version 3.3.3 (R Foundation for Statistical Computing, Vienna, Austria) and Stata version 13.0 (College Station, TX).[13-16]
RESULTS
Agreement between protocols
There was a perfect agreement (100%) between the two protocols for what concerned the presence and number of lesions either when considering them overall or in singular abdominal quadrants. Perfect agreement was also seen for the detection of intestinal complications. Excellent agreement was found for the identification of inflammation and extraintestinal complications, with a kappa value of 0.94 (95% CI 0.8–21.00) and 0.96 (95% CI 0.89–1.0), respectively.
For the assessment of lesion extensions, there was an almost perfect concordance (κc=0.999) with a negligible mean difference (limits of agreement −12.193 mm–12.993 mm) between the two sets of measurements [Figure 3].
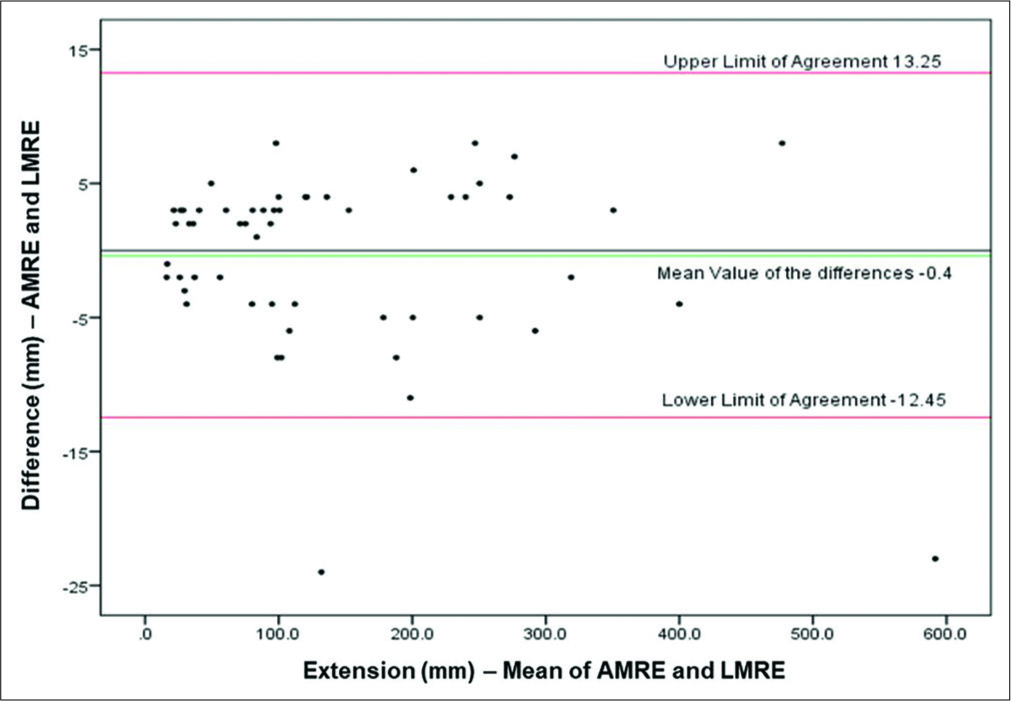
- Bland–Altman analysis for the comparison between AMRE and LMRE. Bland–Altman diagram showing the plot of the difference between the lesion extension (mm) in two AMRE and LMRE against the mean of the pair. Red lines show 95% limits of agreement and the green line shows the mean value of the differences. The black line is the zero line used to assess the discrepancy of the observed mean difference from zero.
Agreement between readers
The interobserver reproducibility between the senior and junior physicians was excellent for the overall presence and number of lesions as well as for the presence and number of lesions in any quadrants. Interobserver reproducibility between the two physicians was excellent for detecting inflammation and intestinal and extraintestinal complications. A concordance coefficient of 0.997 (95% CI 0.995–0.999) between the readers was obtained when considering the lesion extension. The Bland–Altman analysis showed that the difference against the mean in this study did not vary in any systematic way over the range of lesion extension [Figure 4].
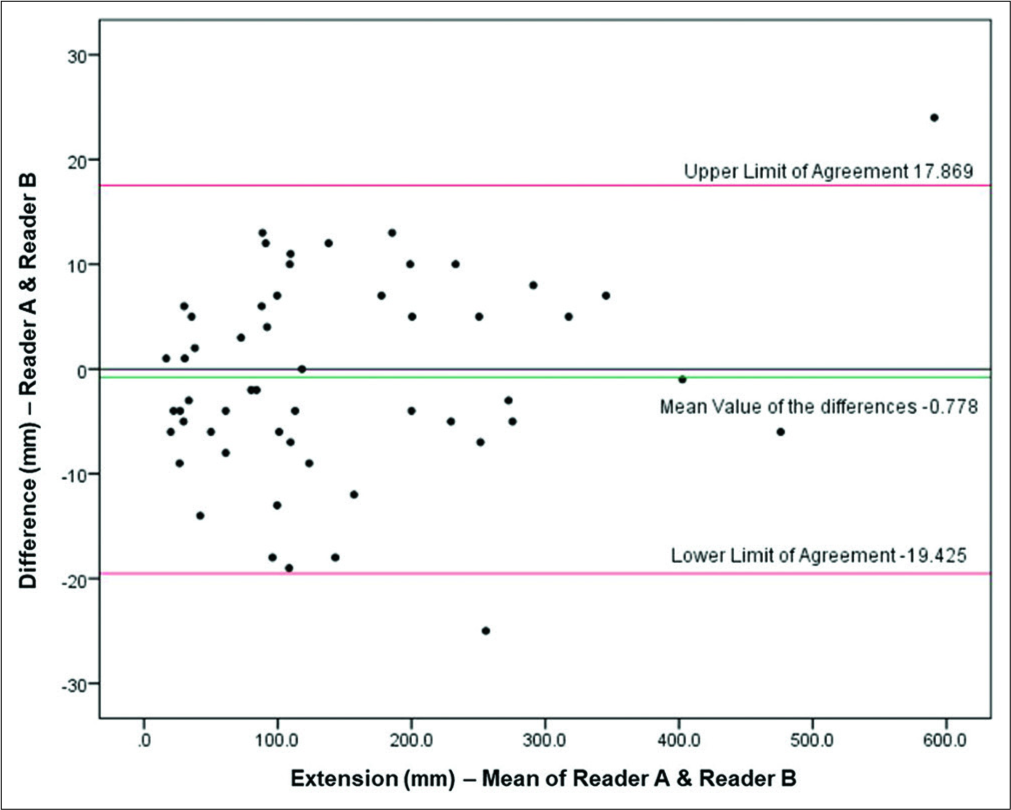
- Interobserver agreement for lesion extension using AMRE protocol. Interobserver diagrams do not show a systematic variability. Red lines show 95% limits of agreement, and the green line shows the mean value of the differences. The black line is the zero line used to assess the discrepancy of the observed mean difference from zero.
Time involved in MRE evaluation
We also examined the time differences between abbreviated MRE protocol and full MRE study. Evaluation time was significantly decreased using the abbreviated MRE protocol compared with full MRE study (median 11 vs. 19 min, p<0.0001), hence, leading to time savings per patient of approximately 35% [Figure 5].
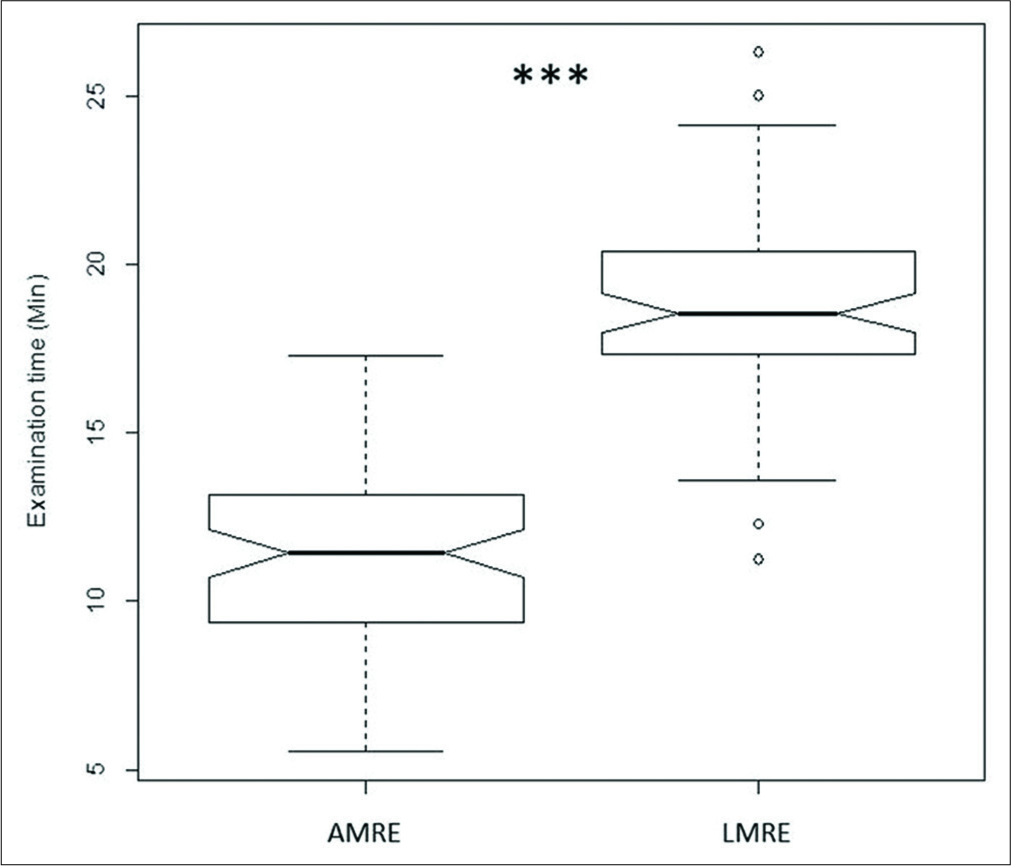
- Notched box-and-whisker plots demonstrating time difference using abbreviated MRE protocol versus full MRE study. Examination time was lower using abbreviated MRE protocol (***P < 0.0001). The black horizontal line in each box represents the median, with the boxes representing the interquartile range. Significant differences are indicated with *** (P < 0.0001) (Mann– Whitney U-test). Evaluation time was significantly decreased using the abbreviated MRE protocol compared with full MRE study (median 11 vs. 19 min), with a time saving of approximately 35% per patient. AMRE: Abbreviated magnetic resonance enterography protocol; LMRE: Long magnetic resonance enterography protocol.
DISCUSSION
MRE is increasingly welcome among clinicians, due to its comprehensive evaluation of bowel loops in CD as well as different intestinal pathologic conditions.[4,17-19]
However, typical limitations of MRE consist in relatively high cost, low spatial resolution, susceptibility to artifacts, and long acquisition times.[3,20]
Considering all the sequences that are routinely included in a standard protocol, MRE scanning time may range from 25 to 45 min.[21,22] The information obtained with all these sequences, though complementary to each other, can sometimes be overlapping and redundant.
The aim of this work was to develop an abbreviated MRE protocol capable to provide equivalent information to the conventional one at the same time improving feasibility at the same time.
The sequences chosen for the fast protocol are those that we consider necessary and indispensable in providing at least sufficient information about the assessment of the disease: Coronal T2-weighted SSH images allow identification and assessment of pathologic bowel thickening, while axial SPAIR T2-weighted and DWI sequences may state the presence of active inflammation and complications related to CD.
According to our suggested protocol, the assessment of active inflammation should rely only on DWI and T2-weighted sequences.
In fact, on the basis of previous published works, DWI could replace the i.v. administration of gadolinium due to their broad equivalence in terms of radiological findings, allowing the recruitment of a larger number of patients (e.g., with renal failure or during pregnancy) and avoiding the risk of an allergic reaction.[17] In fact, while a direct relation between the degree of enhancing and inflammation was already established, the qualitative assessment of inflammation degree is currently based on contrast enhancement patterns, which can be reliably reproduced using DWI.[20]
In fact, inflamed bowel walls are typically hyperintense in DWI, due to the decreased Brownian motion of water molecules consequent to the increased cellular number and formation of lymphoid aggregate.[18]
Several articles already showed encouraging results in DWI evaluation, demonstrating a high degree value with endoscopic findings and an excellent correlation with contrast-enhanced MRE.[21-24]
According to Dohan et al., DWI used in combination with T2-weighted images showed a sensitivity of 93% and a specificity of 67% which was equivalent to T1-weighted contrast-enhanced images.[17]
Nevertheless, Choi et al. demonstrated that non-enhanced MRE (ne-MRE) has a sensitivity of 92.9% and summary specificity of 91% (95% CI, 79.7%–96.3%), higher than contrast-enhanced MRE (ce-MRE).[25]
Furthermore, Quaia et al. noted that ne-MRE versus ce-MRE did not differ in terms of diagnostic accuracy in the active disease, resulting in a uselessness of intravenous contrast agent injection, in assessing disease activity in CD except for patients with equivocal mural T2 hyperintensity. They also found a general agreement in the visualization of sinus tracts, fistulas, and abscesses between ne-MRE and ce-MRE.[26]
Other studies have also shown a high accuracy of DWI in identifying CD complications (i.e. abscesses, enteric fistulas, and neoplasms) with a high degree of sensitivity and specificity.[17,27]
In our work, the evaluation of DWI hyperintensity of the pathologic bowel walls was based on a three-grade classification proposed by Seo et al., through a comparison with the lymph nodes and the spleen.[24]
On the other hand, DWI may be affected by artifacts, consequent to the inadequate bowel distension, presence of intestinal gas or surgical clips, and it showed low accuracy in differentiating inflammation from fibrosis.[24] Moreover, up to now, there is no evidence of an ADC cutoff useful in discriminating active from non-active pattern of CD.[17]
A similar study recently conducted by Rimola et al. suggested to use only T2-weighted sequences as the first step in evaluating CD patients, stating that DWI cannot improve the accuracy of MRE.[28] Nevertheless, the authors concluded that the administration of contrast medium could be avoided in most of the cases and be performed only when uncertain relieves occur at DWI, as a “problem-solving” technique.
While comparing the two protocols, our data showed a perfect concordance for the presence and number of lesions in any abdominal quadrant, in lesion extensions and thickness, and in abdominal complications assessment. High agreement was also found in recognizing active inflammation.
Agreement between readers was excellent for the overall presence and number of lesions as well as for the evaluation of inflammation and intestinal and extraintestinal complications.
Abbreviated MRE also demonstrates a significant time sparing in the evaluation of the images and, on the basis of our regional price list, could allow a cost reduction of 52.5%.
However, our work presents some limitations.
The first is the retrospective evaluation of data on a relatively small population and the lack of a disease-free control group.
Furthermore, this work was based on a subjective radiological evaluation, as an initial step of a wider study, which should include correlation with endoscopy.
Comparing abbreviated and standard protocols, we noticed a not precise correlation between DWI and enhanced scans. Some artifacts may affect DWI evaluation in case of inadequate distention of the bowel with occurrence of false- positive or false-negative results.[18]
CONCLUSION
Our work is focused on the proposal of a fast MRE protocol for intestinal CD assessment that could be more easily performable in uncompliant patients, providing at the same time useful information for clinicians. Our data demonstrate a high concordance between short and standard protocol with an almost complete overlapping of relieves and the advantage of decreased health-care costs as well as acquisition and referring times.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Epidemiology and clinical course of Crohn's disease: Results from observational studies. World J Gastroenterol. 2012;18:1723-31.
- [CrossRef] [PubMed] [Google Scholar]
- Crohn's disease in Italy: A critical review of the literature using different data sources. Dig Liver Dis. 2017;49:459-66.
- [CrossRef] [PubMed] [Google Scholar]
- Colonic inflammation in pediatric inflammatory bowel disease: Detection with magnetic resonance enterography. Pediatr Radiol. 2017;47:850-9.
- [CrossRef] [PubMed] [Google Scholar]
- Utility of MR enterography and ultrasound for the investigation of small bowel Crohn's disease. J Magn Reson Imaging. 2017;45:1573-88.
- [CrossRef] [PubMed] [Google Scholar]
- Small bowel imaging: Computed tomography enterography, magnetic resonance enterography, angiography, and nuclear medicine. Gastrointest Endosc Clin N Am. 2017;27:133-52.
- [CrossRef] [PubMed] [Google Scholar]
- MR enterography findings in abdominopelvic extraintestinal complications of Crohn's disease. J Magn Reson Imaging. 2013;37:1055-63.
- [CrossRef] [PubMed] [Google Scholar]
- Guide to magnetic resonance in Crohn's disease: From common findings to the more rare complicances. Inflamm Bowel Dis. 2011;17:1209-22.
- [CrossRef] [PubMed] [Google Scholar]
- Superior mesenteric artery syndrome in patients with Crohn's disease: A description of 2 cases studied with a novel magnetic resonance enterography (MRE) procedure. Am J Case Rep. 2018;19:431-7.
- [CrossRef] [PubMed] [Google Scholar]
- Detecting active inflammation and fibrosis in pediatric Crohn's disease: Prospective evaluation of MR-E and CT-E. Abdom Imaging. 2013;38:705-13.
- [CrossRef] [PubMed] [Google Scholar]
- Imaging techniques for assessment of inflammatory bowel disease: Joint ECCO and ESGAR evidence-based consensus guidelines. J Crohns Colitis. 2013;7:556-85.
- [CrossRef] [PubMed] [Google Scholar]
- The first joint ESGAR/ESPR consensus statement on the technical performance of cross-sectional small bowel and colonic imaging. Eur Radiol. 2017;27:2570-82.
- [CrossRef] [PubMed] [Google Scholar]
- Inter rater agreement In: Practical Statistics for Medical Research. London: Chapman & Hall, CRC; 1990. p. :403-8.
- [CrossRef] [Google Scholar]
- Methods for evaluating the agreement between diagnostic tests. J Nucl Cardiol. 2016;23:511-3.
- [CrossRef] [PubMed] [Google Scholar]
- A Proposal for Strength-of- Agreement Criteria for Lin's Concordance Correlation Coefficient NIWA Client Report HAM2005062. Atmospheric Research. Tamil Nadu: National Institute of Water & amp; 2005.
- [Google Scholar]
- Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307-10.
- [CrossRef] [Google Scholar]
- Magnetic resonance enterography Findings of intestinal behçet disease in a child. Case Rep Radiol. 2017;2017:8061-648.
- [CrossRef] [PubMed] [Google Scholar]
- Magnetic resonance enterography appraisal of lupus enteritis: A case report. Radiol Case Rep. 2018;13:915-19.
- [CrossRef] [PubMed] [Google Scholar]
- MR enterography: What is next after Crohn's disease? Jpn J Radiol. 2019;37:511-7.
- [CrossRef] [PubMed] [Google Scholar]
- Diffusion-weighted MRI in Crohn's disease: Current status and recommendations. J Magn Reson Imaging. 2016;44:1381-96.
- [CrossRef] [PubMed] [Google Scholar]
- Magnetic resonance enterography: A stepwise interpretation approach and role of imaging in management of adult Crohn's disease. Indian J Radiol Imaging. 2016;26:173-84.
- [CrossRef] [PubMed] [Google Scholar]
- MR enterography of inflammatory bowel disease with endoscopic correlation. Radiographics. 2017;37:116-31.
- [CrossRef] [PubMed] [Google Scholar]
- Small bowel imaging: An update. Curr Gastroenterol Rep. 2016;18:39.
- [CrossRef] [PubMed] [Google Scholar]
- Diffusion-weighted magnetic resonance without bowel preparation for detecting colonic inflammation in inflammatory bowel disease. Gut. 2010;59:1056-65.
- [CrossRef] [PubMed] [Google Scholar]
- Diffusion-weighted magnetic resonance imaging for detecting and assessing ileal inflammation in Crohn's disease. Aliment Pharmacol Ther. 2013;37:537-45.
- [CrossRef] [Google Scholar]
- MR enterography for the evaluation of small-bowel inflammation in Crohn disease by using diffusion weighted imaging without intravenous contrast material: A prospective noninferiority study. Radiology. 2016;278:585-94.
- [CrossRef] [PubMed] [Google Scholar]
- Diffusion-weighted magnetic resonance enterography for evaluating bowel inflammation in Crohn's disease: A systematic review and meta-analysis. Inflamm Bowel Dis. 2016;22:669-79.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of gadolinium-based contrast agent in the assessment of Crohn's disease activity: Is contrast agent injection necessary? J Magn Reson Imaging. 2016;43:688-97.
- [CrossRef] [PubMed] [Google Scholar]
- Diffusion-weighted MRI: Role in detecting abdominopelvic internal fistulas and sinus tracts. J Magn Reson Imaging. 2012;35:125-31.
- [CrossRef] [PubMed] [Google Scholar]
- Increasing efficiency of MRE for diagnosis of Crohn's disease activity through proper sequence selection: A practical approach for clinical trials. Abdom Radiol (NY). 2017;42:2783-91.
- [CrossRef] [PubMed] [Google Scholar]






