Translate this page into:
Extramedullary Plasmacytoma of the Gallbladder Detected on Fluorine 18-fluorodeoxyglucose Positron Emission Tomography/Computed Tomography
Address for correspondence: Dr. Asif Ali Fakhri, Department of Nuclear Medicine and Molecular Imaging, Jacobs School of Medicine and Biomedical Sciences, University at Buffalo, Buffalo, New York, USA. E-mail: asiffakh@buffalo.edu
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Extramedullary plasmacytoma is rare in patients with diagnosed multiple myeloma. Soft tissue plasmacytoma of the gallbladder is particularly uncommon and has been described in only a handful of cases. Diagnosis of gallbladder plasmacytoma with fluorine 18-fluorodeoxyglucose (F18-FDG) positron emission tomography/computed tomography (PET/CT) has not previously been reported. We present a 65-year-old female with a history of multiple myeloma who underwent a restaging F18-FDG-PET/CT which showed a focal area of hypermetabolic activity, corresponding to a nodular lesion within the posterior gallbladder wall. The patient underwent successful cholecystectomy, with surgical pathology revealing gallbladder plasmacytoma. A follow-up scan was negative for active malignancy. This is a novel case of gallbladder plasmacytoma diagnosed on whole-body F18-FDG PET/CT – thus demonstrating the clinical value of this imaging modality in staging, restaging, and surveillance for patients with multiple myeloma.
Keywords
Gallbladder
fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography
plasmacytoma

INTRODUCTION
Multiple myeloma is a common hematologic cancer diagnosed by the presence of paraproteinemia, plasma cell infiltration of the bone marrow, and osteolytic bone destruction.[1] Extramedullary plasmacytoma is involvement of any tissue outside the skeleton, which is seen in approximately 3% of patients with plasma cell myeloma.[2] The most common areas of soft tissue involvement are reported to be kidney, spleen, and liver.[3] However, extraosseous involvement of multiple myeloma is becoming increasingly prevalent as diagnostic imaging at diagnosis, and for treatment, follow-up is rapidly becoming the standard of care.[2]
Among the diagnostic imaging modalities, fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography (F18-FDG-PET/CT) has been shown to be a valuable noninvasive imaging technique for staging multiple myeloma.[2] With its use in staging and treatment monitoring becoming more common place, and with patients diagnosed with the disease surviving longer, there are now more described cases of extraosseous involvement. We present a unique case of gallbladder plasmacytoma that was detected on F18-FDG-PET/CT.
CASE REPORT
A 65-year-old female with a history of IgG-kappa chain multiple myeloma status postseveral rounds of chemotherapy with cyclophosphamide, bortezomib, and dexamethasone presented for a restaging F18-FDG PET/CT, as part of posttreatment monitoring [Figure 1]. This demonstrated a focal area of intense hypermetabolic activity corresponding to an abnormal nodular thickening of the posterior gallbladder wall.
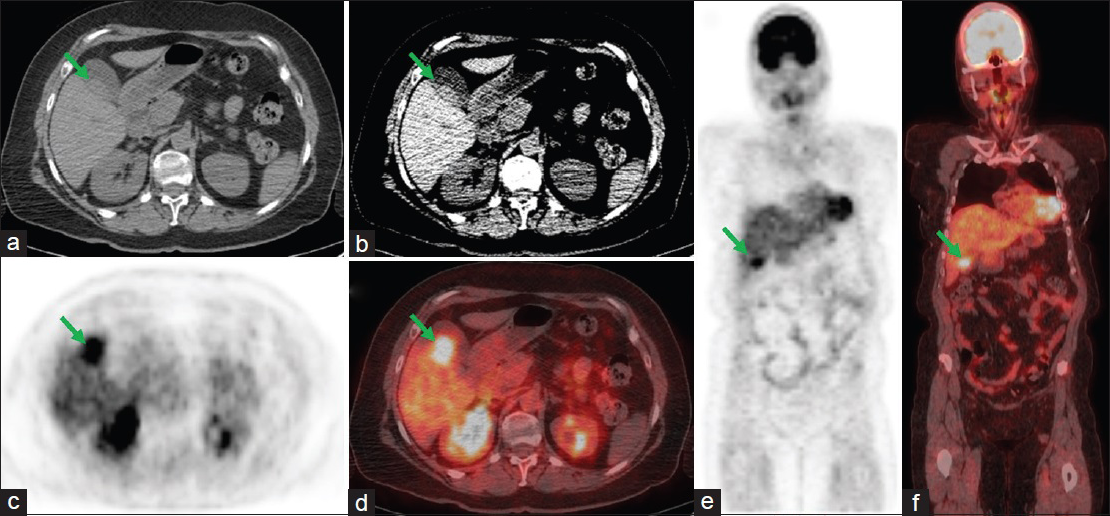
- A 65-year-old female with a history of multiple myeloma status postchemotherapy, presenting for a restaging whole-body fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography scan. (a and b) Transverse computed tomography sections of the abdomen showing nodular thickening (arrows) within the posterior gallbladder wall. (c and e) Transverse and coronal fluorine 18-fluorodeoxyglucose positron emission tomography images showing intense focal hypermetabolic activity (arrows) in the region of the gallbladder. (d and f) Fused transverse and coronal positron emission tomography/computed tomography images showing the focal area of hypermetabolic activity corresponding to the nodular thickening within the posterior gallbladder wall (arrows).
Due to suspicion for malignancy, the patient underwent elective cholecystectomy. Intraoperatively, an exophytic mass with some vascularity was noted in the fundus and body of the gallbladder. The mass was removed along with the gallbladder. Histopathology was negative for carcinoma but revealed kappa light chain restricted plasmacytoma. Her M-spike counts remained stable on postcholecystectomy follow-up. A restaging F18-FDG-PET/CT 4 months postoperatively revealed no abnormal hypermetabolism within the postsurgical gallbladder fossa [Figure 2].
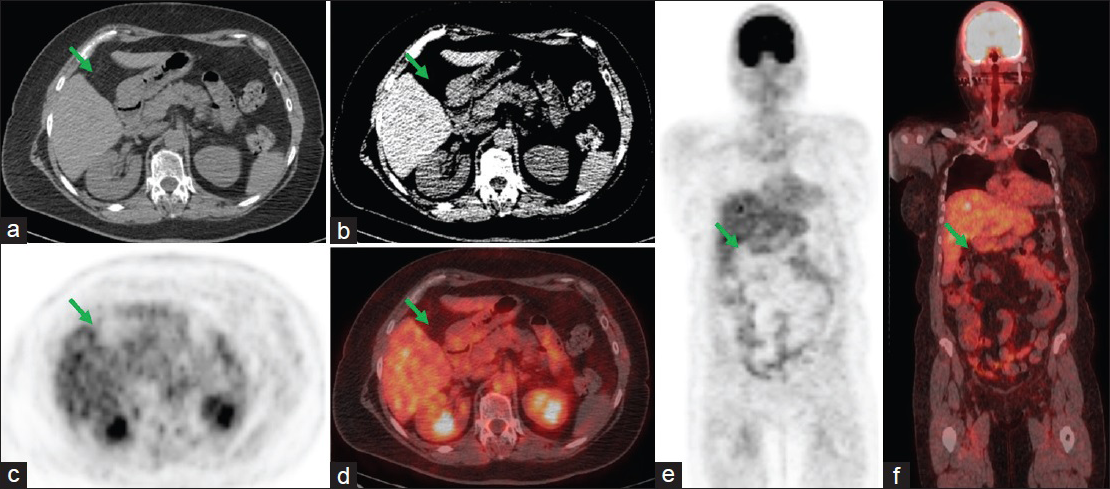
- Postcholecystectomy surveillance whole-body fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography scan. (a and b) Transverse computed tomography sections of the abdomen showing postsurgical changes from cholecystectomy (arrows). (c and e) Transverse and coronal fluorine 18-fluorodeoxyglucose positron emission tomography images showing no metabolic evidence of active malignancy in the gallbladder fossa (arrows). (d and f) Fused transverse and coronal positron emission tomography/computed tomography images confirming that there is no metabolic evidence of active malignancy in the gallbladder fossa (arrows).
DISCUSSION
Various findings can explain the elevated glucose metabolism involving the gallbladder on F18-FDG PET/CT imaging. In patients with hypermetabolic activity involving the gallbladder on F-18-FDG PET/CT, inflammatory or infectious etiologies, such as acute or chronic cholecystitis, can easily be mistaken for malignancy.[4] The focal and nodular gallbladder wall thickening, with corresponding focally elevated metabolism, in this patient, made the differential consideration of cholecystitis much less likely.
Primary malignancy of the gallbladder, such as primary gallbladder carcinoma, could also explain the findings in this patient. The most common malignant gallbladder polyp is primary adenocarcinoma of the gallbladder, which is FDG avid on F18-FDG-PET/CT.[5] Without histopathology from the surgical sample after cholecystectomy, primary carcinoma would have been difficult to exclude. In fact, given the overall rarity of metastatic disease involving the gallbladder, particularly metastatic multiple myeloma, primary gallbladder carcinoma would have been a reasonable differential consideration.[5]
Benign hyperplastic cholecystosis of the gallbladder wall, otherwise known as gallbladder adenomyomatosis, would have been another potential differential consideration in this patient. Adenomyomatosis of the gallbladder has also been shown to cause hypermetabolic activity on F18-FDG-PET/CT.[5] Furthermore, the patient's demographics are congruent with higher risk of gallbladder adenomyomatosis as it has a greater incidence in female patients past the fifth decade of life.[5]
Extramedullary plasmacytomas have reported in the literature as being present in various soft tissues.[6] Metastatic deposit via hematogenous seeding certainly makes liver, spleen, and kidney vulnerable targets for plasmacytoma, given their dominant vascularity. This is a possible explanation for the relative rarity of metastatic disease to the gallbladder. There are only 6 other cases which have been reported; however, characterization on F18-FDG PET/CT has not previously been described.[7] Timely detection and appropriate diagnosis of extramedullary plasmacytoma in patients with diagnosed multiple myeloma are of particular importance because this finding carries a worse prognosis.[1]
FDG-PET/CT offers a distinct advantage over traditional anatomic imaging modalities in surveillance and treatment monitoring of multiple myeloma – by providing a whole-body survey of active disease. In this way, it is particularly helpful in detecting not only new osseous lesions but also extraosseous lesions.[2] Gallbladder plasmacytoma represents an unlikely extraosseous manifestation of multiple myeloma that would lend itself to more accurate detection through metabolic imaging such as FDG-PET/CT.
CONCLUSION
This case illustrates the overall value of whole-body F18-FDG-PET/CT in both stagings of skeletal involvement in patients with multiple myeloma and in detection of extraosseous plasmacytoma. Furthermore, it demonstrates the utility of metabolic imaging in diagnosing unlikely soft tissue plasmacytoma, such as within the gallbladder.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/40/191127
REFERENCES
- Value of FDG PET in the assessment of patients with multiple myeloma. AJR Am J Roentgenol. 2005;184:1199-204.
- [Google Scholar]
- FDG PET or PET/CT for detecting intramedullary and extramedullary lesions in multiple myeloma: A systematic review and meta-analysis. Clin Nucl Med. 2012;37:833-7.
- [Google Scholar]
- Clinical and pathologic findings in 52 consecutively autopsied cases with multiple myeloma. Am J Hematol. 2001;67:1-5.
- [Google Scholar]
- Chronic cholecystitis with hilar bile duct stricture mimicking gallbladder carcinoma on positron emission tomography: A case report. Mol Clin Oncol. 2013;1:517-20.
- [Google Scholar]
- Polypoid lesions of the gallbladder: Disease spectrum with pathologic correlation. Radiographics. 2015;35:387-99.
- [Google Scholar]
- Extramedullary lesions in multiple myeloma; review of literature and pathologic studies. AMA Arch Pathol. 1952;53:262-72.
- [Google Scholar]
- Extramedullary plasmacytoma of the gallbladder diagnosed by endoscopic ultrasound fine needle aspiration (EUS-FNA) J Gastrointest Oncol. 2015;6:E7-9.
- [Google Scholar]






