Translate this page into:
Diffusion-weighted Magnetic Resonance Imaging in the Early Diagnosis of Neonatal Adrenoleukodystrophy
Address for correspondence: Dr. Mehmet H Atalar, Cumhuriyet University School of Medicine, Department of Radiology, Sivas, TR 58140, Turkey. E-mail: mhatalar@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
A newborn baby girl developed seizures right after birth. On the fourth day, the baby was examined using diffusion sequence magnetic resonance imaging (MRI) and diagnosed to have neonatal adrenoleukodystrophy. Laboratory findings confirmed the diagnosis. This is the first case of neonatal adrenoleukodystrophy (NALD) where diffusion MRI sequence helped in the diagnosis. We find association of NALD with seizures at birth is an extremely rare occurrence, and so far, only one case has been mentioned in the literature.
Keywords
Adrenoleukodystrophy
diffusion tensor imaging
newborn
MRI
brain
INTRODUCTION

Neonatal adrenoleukodystrophy (NALD) belongs to a family of disorders that result from defects in the biogenesis and/or functioning of the peroxisomes and are referred to as peroxisome biogenesis disorders (PBDs). NALD bears many clinical and biochemical similarities to X-linked adrenoleukodystrophy (X-ALD).[1] The lesions are usually seen in the occipital lobes and corpus callosum,[2–8] although atypical cases with predominantly frontal lobe involvement can be seen at times.[9] In the literature, there are several diffusion magnetic resonance imaging (MRI) reports on adrenoleukodystrophy and adrenomyeloneuropathy;[2–9] however, no diffusion MRI report exists on the neonatal form. There is only a single report in literature, of a case of NALD presenting with seizures at birth.[10] Here we report a case of a neonate who had seizures, and adrenoleukodystrophy was diagnosed by a diffusion MRI sequence.
CASE REPORT
Our patient was a 4-day-old girl born after a normal pregnancy and spontaneous vaginal delivery. Myoclonic seizures were observed in the baby right after the delivery. She had generalized hypotonia. Routine laboratory blood and urine tests were unremarkable. Seizures were controlled by phenobarbital. Computed tomography (CT) and MRI examinations were performed on the fourth day of life. A CT scan was unremarkable. T1- and T2-weighted sequences of the MRI examination were also normal [Figure 1a]. A diffusion MRI sequence was added to the imaging protocol. A maximum gradient strength of 30 mT/m was administered. The diffusion MRI sequence used was the so-called trace sequence, a single-shot, spin-echo, echo-planar imaging sequence, which averages the data from the three (x, y, and z) gradients, and provides automatically generated ADC (apparent diffusion coefficient) maps. The acquisition time was 22 sec with TR = 3900 msec, and TE = 116 msec; the slice thickness was 5 mm. The diffusion-weighted images, obtained with a b value of 1000 sec/mm2, revealed apparent high-signal changes in the splenium of the corpus callosum, and occipital lobes [Figure 1b]. The ADC maps (which are free from T2 shine-through effects) revealed that the ADC values of the involved regions (0.69 × 10-3 mm2/sec) were low, compared to other parenchyma regions with value of 1.24 × 10-3 mm2/sec [Figure 1c].
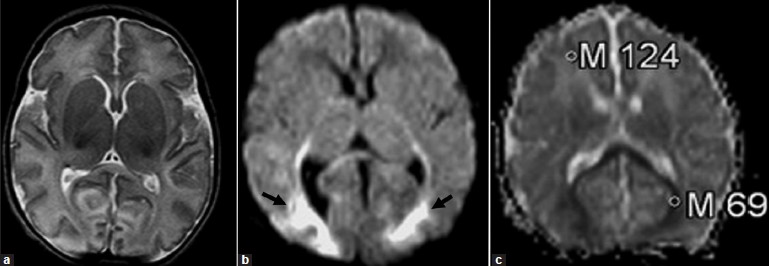
- Neonatal adrenoleukodystrophy: (a) the axial T2-weighted MR image is normal; (b) the diffusion-weighted image (b = 1000 sec/mm2) reveals high-signal changes in the splenium of the corpus callosum, and occipital lobes (black arrows); (c) the ADC map reveals that the ADC values of the involved regions is low (0.69 × 10–3 mm2/sec), compared to the frontal regions (1.24 × 10–3 mm2/s).
A radiological diagnosis of NALD was suggested to the clinicians. The diagnosis was confirmed by the demonstration of abnormaly high levels of saturated, very long chain of fatty acids (VLCFA) (C24/C22, C26/C22, and pipecolic acid) in a plasma assay.
DISCUSSION
According to the previous reports dealing with diffusion MRI changes in adrenoleukodystrophy, alterations in diffusion coefficients were noted in the brain regions affected by this disorder. Elevated isotropic diffusion and reduced fractional anisotropy were the main findings. Zonal differences within the lesions were noted, reflecting zonal histopathologic changes. Also, diffusion-weighted images, (b = 1000 sec/mm2), and ADC values revealed significant changes at the lesion sites in comparison to the normal-appearing brain parenchyma.[2–9] A number of recent diffusion MRI studies have provided normal ADC values of the cerebral white matter and basal ganglia [Figure 2]. It appears that there is a consensus that the mean ADC value of the normal white matter in newborns is about 1.23 × 10–3 mm2/sec. In Sener's study, the mean ADC values (1.23 × 10–3 mm2/sec) in the normal brain of infants up to 2 years of age are as follows: unmyelinated white matter, 1.64 ± 0.17; myelinated white matter, 0.90 ± 0.12; paracentral cortices, 0.83 ± 0.14; basal ganglia and thalami, 0.98 ± 0.11; brainstem, 1.00 ± 0.10; and cerebellar parenchyma, 0.97 ± 0.13.[10] Thus, the ADC values in the occipital lobes of the present patient with NALD (e.g., 0.69 × 10–3 mm2/sec) were apparently low. In this case, although a CT scan and T1- and T2-weighted MR images of the newborn appeared entirely normal, the diffusion-weighted images, (b = 1000 sec/mm2), revealed apparent high-signal changes in the splenium of the corpus callosum and occipital lobes, enabling the correct diagnosis of NALD. The ADC maps, which are free from T2 shine-through effects, revealed that the ADC values of the involved regions were low, compared to other parenchyma regions. These diffusion MRI changes were equivalent to a restricted diffusion pattern in the regions involved with NALD. This pattern is likely to change according to the evolution of the disease process as the child gets older.
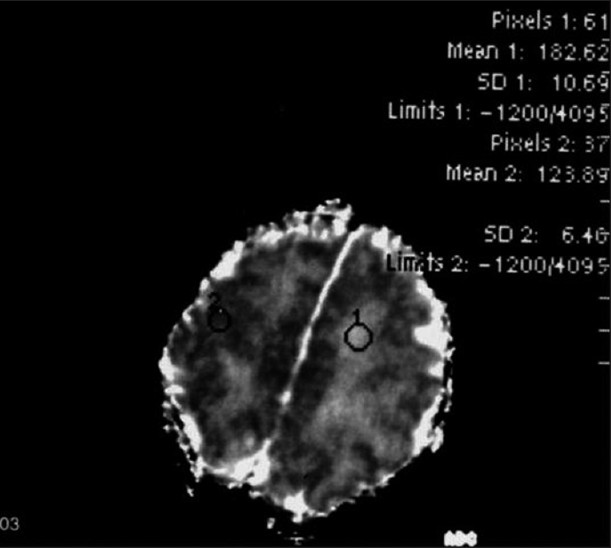
- A healthy 4-day-old boy. ADC map: normal parechymal values. ADC measurements are shown by region of interest (ROI) evaluations. Large ROI evaluations from different regions in the parenchyma reveal normal ADC values: 1.82 and 1.23 × 10–3 mm2/sec.
On the other hand, a recent report by Chang et al.[11] dealt with a newborn with seizures, who was shown to have NALD by laboratory tests. The authors cited that antepartum and peripartum seizures are rare, and an onset of seizures in the first 3 days of life occurs in hypoxic–ischemic injuries, intracranial hemorrhage, congenital brain malformations, drug-related conditions, and in various metabolic encephalopathies. A later onset of seizures occurs with intracranial infection, metabolic disorder, and drug withdrawal.[11] The authors also noted that their case of NALD with seizures at birth was a unique case. Therefore, our patient with NALD and myoclonic seizures at birth becomes the second example of this condition.
In conclusion, we found diffusion MRI very useful in diagnosing NALD by revealing apparent changes in the occipital lobes and corpus callosum in the absence of any detectable change in CT and conventional MRI. Besides the recently reported case,[11] this study on our patient suggests that NALD should be added as an imaging protocol in the differential diagnosis of very early onset of seizures that occur in the first 3 days of life.
Source of Support: Nil
Conflict of Interest: None declared.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp? 2011/1/20/78530
REFERENCES
- Pharmacological induction of peroxisomes in peroxisome biogenesis disorders. Ann Neurol. 2000;47:286-96.
- [Google Scholar]
- Diffusion tensor brain MR imaging in X-linked cerebral adrenoleukodystrophy. Neurology. 2001;56:544-7.
- [Google Scholar]
- Diffusion anisotropy in cerebral white matter lesion. No To Shinkei. 1994;46:765-70.
- [Google Scholar]
- Peroxisomal biogenesis disorder: Comparison of conventional MR imaging with diffusion-weighted and diffusion-tensor imaging findings. AJNR Am J Neuroradiol. 2004;25:1022-7.
- [Google Scholar]
- Diffusion tensor imaging in cases of adrenoleukodystrophy: Preliminary experience as a marker for early demyelination? AJNR Am J Neuroradiol. 2003;24:819-24.
- [Google Scholar]
- Proton MR spectroscopic and diffusion tensor brain MR imaging in X-linked adrenoleukodystrophy: Initial experience. Radiology. 2002;225:245-52.
- [Google Scholar]
- Concepts of myelin and myelination in neuroradiology. AJNR Am J Neuroradiol. 2000;21:1099-109.
- [Google Scholar]
- Diffusion tensor-based imaging reveals occult abnormalities in adrenomyeloneuropathy. Ann Neurol. 2005;58:758-66.
- [Google Scholar]
- Atypical X-linked adrenoleukodystrophy: New MRI observations with FLAIR, magnetization transfer contrast, diffusion MRI, and proton spectroscopy. Magn Reson Imaging. 2002;20:215-9.
- [Google Scholar]
- Diffusion MRI: Apparent diffusion coefficient (ADC) values in the normal brain and a classification of brain disorders based ADC values. Comput Med Imaging Graph. 2001;25:299-326.
- [Google Scholar]
- Neonatal adrenoleukodystrophy presenting with seizure at birth: A case report and review of the literature. Pediatr Neurol. 2008;38:137-9.
- [Google Scholar]






