Translate this page into:
Dermatofibrosarcoma Protuberans: Insights into a Rare Soft Tissue Tumor
Address for correspondence: Dr. Sawsan Taif, Department of Radiology, Khoula Hospital, PO Box 90, 116 Muscat, Sultanate of Oman. E-mail: sawsantaif@gmail.com
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Dermatofibrosarcoma protuberans (DFSP) is a rare soft tumor which originally represents a cutaneous sarcoma. It grows slowly and presents usually as nodular superficial lesion on the trunk or the extremities. Although these tumors are locally aggressive with high rate of recurrence following surgery; the prognosis is considered excellent when it is effectively treated. The radiological appearance of this tumor has rarely been studied and findings infrequently discussed in the literature probably because many lesions underwent resection before imaging. Although imaging is infrequently performed for this lesion; it can show characteristic features and demonstrate the full extent. Imaging may also play a role in the differentiation of this tumor from more serious soft tissue lesions such as more aggressive sarcomas and hemangioma. In this article, we discuss the imaging findings of DFSP that can aid in its diagnosis and its variable appearances. In addition; the clinical presentation and treatment options are also described with review of the previous literature.
Keywords
Dermatofibrosarcoma protuberans
literature review
low-grade tumor
soft tissue sarcoma

- Aliya Sarhan Al Barwani
INTRODUCTION
Dermatofibrosarcoma protuberans (DFSP) represents a low-grade soft tissue sarcoma that originates from the skin dermis layer. It is a rare tumor which accounts for about 5% of soft tissue sarcoma. They can occur in all age groups and both sexes with young adults most frequently affected. The incidence is also slightly higher in men than women.
It usually presents as a slowly growing superficial soft tissue mass, which when effectively treated, has a very good prognosis. Occasionally, these lesions become locally aggressive and invade the deep soft tissues. In addition, these lesions may show recurrence particularly if the treatment is not appropriate or the removal is incomplete.
This tumor was first described by Darier and Ferrand in 1924 and named later by Hoffmann in 1925.[12]
At presentation, the size of these lesions also ranges; they can be as small as one centimeter. However, some lesions can be large.
The trunk and extremities are considered the most common sites for DFSP; nevertheless, it has been described in various parts of the body including the neck, head, breast, and even vulva.
As patients present usually with slowly growing long-standing superficial mass which eventually require surgical removal, imaging studies are infrequently performed and play a small role in the management of this condition. However, computed tomography (CT) or magnetic resonance imaging (MRI), when readily available, may be offered in some centers for further characterization of a solid soft tissue mass. Imaging may also be requested to see the full extent of the mass in the rare occasions of deeply seated, infiltrative lesions described by some authors.[345]
In this review, we describe the imaging features of DFSP to highlight this lesion and show the importance to keep it as one of the differentials in superficial soft tissue masses. We discuss the clinical manifestations and variable treatment options as well.
CLINICAL PRESENTATION
Typically, patients present with slow-growing mass or skin lesion which is not usually causing much pain or other symptoms. It is superficial in most of the cases, red-brown to bluish nodular in appearance and can show irregular margins. Consequently, patients frequently tend to neglect their problem and presentation can be late. Most patients have this tumor either as a stable lesion or growing over years. However, a few may have accelerated growth, painful swelling or even in rare cases ulceration, and bleeding in larger lesions.
The size at presentation is also very variable ranging from several centimeters to over 20 cm.[34]
These lesions are often located on the trunk and extremities; however, dermatofibrosarcoma has been reported in many other areas such as scalp, breast, pubic area, and vulva.[67891011] Therefore, the presentation also depends on area involved. It can present as mass in the scalp, breast or vulva, and extremities.
Most of the patients are young or middle age with tumor occurring in the third to seventh decades of life. However, this lesion has also been detected in children. A study that performed by Tsai et al., on pediatric DFSP has described this issue and showed the importance of differentiating this lesion from other soft tissues lesions such as hemangioma.[9]
IMAGING FINDINGS AND DIAGNOSIS
Owing to its rarity, literature has shown only a few studies which discussed imaging findings and treatment options of DFSP. By reviewing the literature published in the last 40 years, we found only two studies that reviewed imaging appearance of this lesion. Another factor for the little information obtained about the imaging findings of DFSP is its superficial location in most of the cases. Consequently, patients undergo excision and biopsy of the mass without the need for CT or MRI.
In the study done by Kransdorf and Meis-Kindblom, the imaging investigations of 11 patients with a histologically proven DFSP were reviewed which showed superficial well-defined nodular masses involving the skin and subcutaneous fatty tissue. On CT, these lesions showed attenuation values approaching that of skeletal muscle. Moderate degree of contrast enhancement was noted in these lesions. MR, which was done in only four of the cases, demonstrated prolonged T1 and T2 relaxation times (hyperintense on T2 and hypointense on T1-weighted images); signal characteristic which is considered nonspecific and demonstrated by many other benign and malignant soft tissue conditions.[12]
Another study by Torreggiani et al., reviewed imaging studies of 10 cases of histologically confirmed DFSP. In this study, three cases out of 10 were found to be deep in contrast to the usual superficial location. In two of them, the tumor seems not even related to the skin. They also found one patient showing an ill-defined margin rather than the more common well-defined tumors. Similar to the observation made by Kransdorf and Meis-Kindblom, they noticed predominantly high signal on T2 and STIR and low or intermediate signal on T1 and either uniform or patchy pattern of enhancement. Consequently, Torreggiani concluded that although most of the times, DFSP is superficial and well defined, ill-defined, and deeply seated lesions do occur and knowledge of these variable MRI characteristics may aid in the diagnosis of difficult or atypical cases.[5] A typical case of DFSP in the shoulder shown in Figure 1 demonstrates its superficial location, relation to the skin, and the typical MRI signal [Figure 1].
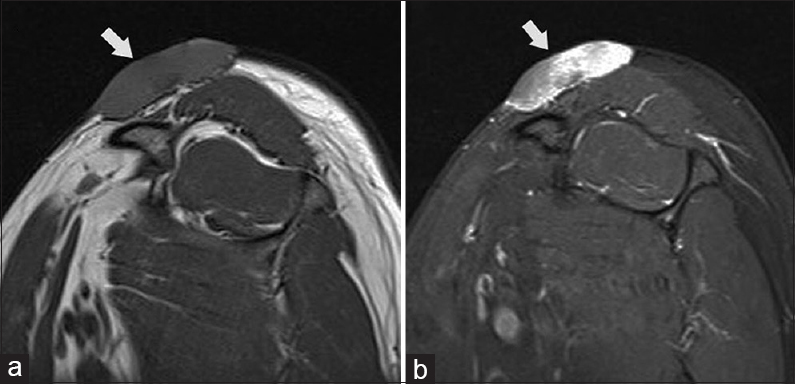
- Dermatofibrosarcoma protuberans of the shoulder in a young female. Sagittal T1 (a) and PD FS (b) MR images of the shoulder showing the tumour (arrows) as well defined solid mass lying in the subcutaneous fat and in close relation to the skin; showing isointense signal on T1 and hyperintense signal on PD.
Owing to the previously mentioned imaging findings and the rarity of this tumor, its presence can be confused with common lesions such as hemangioma.[9]
Moreover, cases of DFSP with atypical locations show not much radiological difference from that found in the typical common locations such as trunk and extremities. Similar MRI appearance was observed by Ozmen et al., who reported a case of vulvar DFSP which appeared hyperintense on T2 and STIR and hypointense on T1-weighted images and demonstrated heterogeneous enhancement.[11]
Lee et al., have done a study on cases of DFSP arising within the breasts and reviewed their imaging characteristics. Lesions presented as skin-based circumscribed dense mass on mammography in most of the cases. On US, most of their patients demonstrated heterogeneous circumscribed lesions and some showed few lobulations. In the study by Lee et al., only one patient underwent MRI that demonstrated characteristics similar to rest of the reported cases showing skin-based, spindle-shaped tumor with smooth margin and appeared hyperintense on T2 and isointense on T1-weighted images as compared to the breast parenchyma and showed homogenous postcontrast enhancement.[7] From the previously reported cases, we can notice that breast's DFSP appear as solid masses and usually demonstrate benign imaging appearance. However, in the case reported by Jiang et al., they described a case of breast dermatofibrosarcoma which appeared irregular hypoechoic on US with evidence of invasion into the surrounding tissues making it suspicious for malignancy; therefore, it was given a score of 4 on Breast Imaging Reporting and Data System.[8] Subsequent biopsy and histological examination confirmed DFSP.
In the suprapubic dermatofibrosarcoma reported by Lim et al., patient underwent CT which revealed solid isodense subcutaneous tumor showing enhancement in postcontrast images. However, MRI was not done in this case.[10]
Although considered benign, extremely rare, and unusual manifestations have also been observed like the case reported by Dhir et al., who described a patient with chest wall mass that proved to be DFSP with fibrosarcomatous transformation. Two years following resection, the patient presented again with abdominal mass involving the pancreas, spleen, and stomach that appeared on CT as a huge retroperitoneal heterogeneous density mass.[13]
HISTOPATHOLOGY
DFSP is a spindle cell tumor, which was first described in 1924 by Darier and Ferrand.[1] This lesion usually arises in the dermis forming a multinodular mass which can also extend into the subcutaneous tissues and muscles [Figure 2].
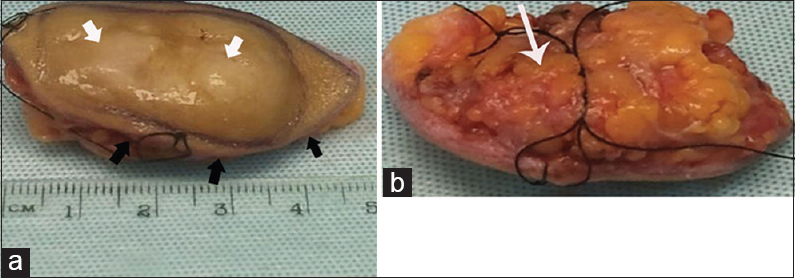
- Macroscopic appearance of an excised dermatofibrosarcoma protuberans. (a) Gross examination shows oval-shaped mass with skin on one surface (black arrow) and fat on the opposite (white arrow). (b) Sectioning reveals the yellow-white solid nodular tumor (arrow).
Microscopically, it demonstrates a uniform population of monomorphic spindle cells arranged in a characteristic storiform pattern over a background of fibrous stroma [Figure 3]. These cells may show infiltration into the surrounding subcutaneous fat. The lesions show little nuclear pleomorphism and have low to moderate mitotic activity. They occasionally show myxoid or densely collagenous areas or areas of hemorrhage.[13514]
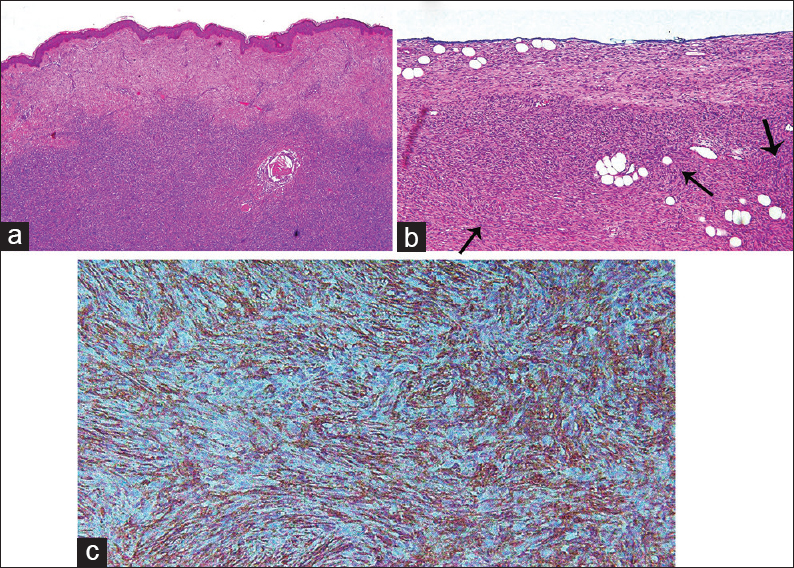
- Typical histological findings of dermatofibrosarcoma protuberans. Hematoxylin- and eosin-stained tissue (a) low power microscope and (b) high power field show fibroblastic neoplasm with uniform appearance of storiform pattern (arrow) with minimal cellular pleomorphism and low mitotic activity. (c) Immunohistochemical staining for CD34 reveals positive result.
Immunohistochemical studies reveal strong staining with human progenitor cell antigen CD34, but negative for factor XIIIa [Figure 3].[1415]
Variants of this lesion have been identified such as myxoid, myoid, granular cell, sclerotic, atrophic DFSP, and giant cell fibroblastoma. A special form of DFSP which is characterized by pigmented appearance is called Bednar's tumor. This type constitutes <5% of all lesions.
Although these lesions are benign in most of the cases, higher-grade sarcomas, usually fibrosarcoma, may superimpose on conventional forms of dermatofibrosarcoma.[7141516]
TREATMENT AND PROGNOSIS
Although authors label the prognosis as excellent; DFSP are also reported to have a high recurrence rate. This tumor can be locally aggressive and infiltrative, especially when incompletely resected. Sometimes, it shows deep subcutaneous tissue extension or more rarely deeper extension inside the muscle compartment.[3516]
Basically, the optimal treatment of these tumors whether primary or metastatic is complete surgical resection with negative margins which can be achieved using two different techniques.
Although there was some debate regarding its role, Mohs micrographic surgery (MMS) is considered by many as the ideal treatment for DFSP. Mohs surgery is a method of obtaining complete margin during removal of a skin cancer as it involves microscopical examination of each removed tissue for cancer cells. It differs from the conventional surgical technique by the way of pathology processing which can be very time-consuming.
Serra-Guillén et al., studied the results obtained from Mohs surgery and compared them to those obtained by conventional surgical procedures. They have concluded that MMS can achieve tumor clearance with smaller margins and greater preservation of healthy tissue.[17] However, this procedure has the drawbacks of limited availability to only some centers and long procedure time.
An alternative option to Mohs surgery is wide excision which used to be the standard treatment. This procedure involves proper identification of the tumor periphery and is followed by 1–2 cm resection margin. It is combined with frozen section for histologic analysis and if the margin is positive re-resection will be needed.
Some authors have suggested a higher cure rate with MMS due to better control of the margins. Gloster et al., studied cases of DF treated by Mohs surgery and compared their results to that of wide excision present in the literature. They have reported higher cure rate using Mohs surgery and suggested that this procedure may be the treatment of choice for DFSP.[18]
Meguerditchian et al., thought that the local control of the tumor will eventually be similar in both MMS and wide excision although the positive margin is more common with wide excision.[19]
Snow et al., studied the data of 136 patients with DFSP, who underwent MMS and reported 7% local recurrence rate after 5 years which can be controlled by another Mohs surgery. He also reported cure rate of 98.5% after second Mohs procedure. He advised to keep the patients on long follow-up as late local recurrence can occur even after 10 years.[20]
DuBay et al., have done a study that confirmed efficacy of wide excision which is usually used for the treatment of DFSP in their center.[21]
In general, the choice between MMS and wide excision should depend on tumor location and personal experience in that particular center. MMS is preferred in regions such as the head and neck regions, where tissue conservation is an important issue.
Moreover, proper management of DFSP may involve radiotherapy. Williams et al., assessed the effectiveness of postoperative radiotherapy in treating patients with this condition. They have shown that radiotherapy may improve local control and reduce the risk of recurrence postoperatively in patients with DFSP.[22]
Authors have also found that adjuvant radiotherapy improves local control in patients with close or positive margins following surgery. In their studies, Dagan and Sun et al., concluded that postoperative radiotherapy may reduce the risk of local recurrence in patients with DFSP, who have a high risk of residual disease after surgery.[2324]
Regarding the role of chemotherapy, reports showed that patients with inoperable, recurrent, or metastatic disease may benefit from imatinib which is a tyrosine kinase inhibitor. It acts by inhibiting the platelet-derived growth factor receptor tyrosine kinase. Neoadjuvant imatinib was also tried for locally advanced primary tumors, whereas adjuvant imatinib was used for resected tumors with positive margins. Dhir et al., reported a case of metastatic DF in the pancreas which was treated by imatinib.[13]
CONCLUSION
DFSP is rare, typically superficial soft tissue tumor which sometimes does not require imaging before excision and histological diagnosis. However, knowledge of the radiological appearance is essential to diagnose typical cases and differentiate them from other soft tissue lesions. Imaging is also helpful for assessment of the full extent of atypical aggressive lesions. The usual imaging appearance is a noncalcified superficial nodular mass frequently related to the skin which is isodense to muscles on CT and showing T2 high and T1 low MR signal. This tumor should be kept in mind as one of the differential diagnoses particularly in predominantly superficial lesions.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2016/6/1/16/181492
REFERENCES
- Dermatofi brosarcomes prograssifs at ricidivantes on fibrosarcomes de la peau (Dermatofibrosarcoma: A progressive and recurring rigid fibrosarcoma of the skin) Ann Dermatol Venereol. 1924;5:545-62.
- [Google Scholar]
- Über das knollentreibende fibrosarkom der haut (About the dermatofibrosarcoma protuberans of the skin) Acta Dermatol Venereol Suppl (Stockh). 1925;43:1-28.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: MR imaging features. AJR Am J Roentgenol. 2002;178:989-93.
- [Google Scholar]
- Dermatofibrosarcoma protuberans of the breast: Imaging features and review of the literature. AJR Am J Roentgenol. 2009;193:W64-9.
- [Google Scholar]
- Dermatofibrosarcoma protuberans of the breast: A case report. Oncol Lett. 2014;8:1202-4.
- [Google Scholar]
- Dermatofibrosarcoma protuberans in children and adolescents: Clinical presentation, histology, treatment, and review of the literature. J Plast Reconstr Aesthet Surg. 2014;67:1222-9.
- [Google Scholar]
- Suprapubic dermatofibrosarcoma protuberans: Case report and literature review. JTUA. 2008;19:232-4.
- [Google Scholar]
- Magnetic resonance imaging of vulvar dermatofibrosarcoma protuberans – Report of a case. Radiol Oncol. 2013;47:244-6.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: Radiologic appearance. AJR Am J Roentgenol. 1994;163:391-4.
- [Google Scholar]
- Neoadjuvant treatment of dermatofibrosarcoma protuberans of pancreas with imatinib: Case report and systematic review of literature. Clin Sarcoma Res. 2014;4:8.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: A comprehensive review and update on diagnosis and management. Semin Diagn Pathol. 2013;30:13-28.
- [Google Scholar]
- Expression of the human progenitor cell antigen CD34 (HPCA-1) distinguishes dermatofibrosarcoma protuberans from fibrous histiocytoma in formalin-fixed, paraffin-embedded tissue. J Am Acad Dermatol. 1993;28:613-7.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: Review of 20-years experience. Clin Transl Oncol. 2006;8:606-10.
- [Google Scholar]
- Mohs micrographic surgery in dermatofibrosarcoma protuberans allows tumour clearance with smaller margins and greater preservation of healthy tissue compared with conventional surgery: A study of 74 primary cases. Br J Dermatol. 2015;172:1303-7.
- [Google Scholar]
- A comparison between Mohs micrographic surgery and wide surgical excision for the treatment of dermatofibrosarcoma protuberans. J Am Acad Dermatol. 1996;35:82-7.
- [Google Scholar]
- Wide excision or Mohs micrographic surgery for the treatment of primary dermatofibrosarcoma protuberans. Am J Clin Oncol. 2010;33:300-3.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: A report on 29 patients treated by Mohs micrographic surgery with long-term follow-up and review of the literature. Cancer. 2004;101:28-38.
- [Google Scholar]
- Low recurrence rate after surgery for dermatofibrosarcoma protuberans: A multidisciplinary approach from a single institution. Cancer. 2004;100:1008-16.
- [Google Scholar]
- Dermatofibrosarcoma protuberans: Treatment results of 35 cases. Radiother Oncol. 2000;57:175-81.
- [Google Scholar]
- Radiotherapy in the treatment of dermatofibrosarcoma protuberans. Am J Clin Oncol. 2005;28:537-9.
- [Google Scholar]






